Optimizing the Analysis of Sugar Alcohol Excipients in Pharmaceutical Tablet Formulations Using Rezex Ion Exclusion HPLC Columns
Tablet formulations of most major pharmaceutical drug products contain significant amounts of inactive ingredients (excipients) in their formulations.
Michael McGinley, Phenomenex, Inc.,
Tablet formulations of most major pharmaceutical drug products contain significant amounts of inactive ingredients (excipients) in their formulations. Such excipients are often used as binders to hold a tablet together or as a filler to increase the bulk volume of a tablet (especially for highly potent active pharmaceutical ingredients). Sugar alcohols, such as mannitol, sorbitol, and xylitol, are often used as fillers because of their inert properties and sweet taste (1).
While inexpensive and convenient, such sugar alcohols require unique methods for analysis and quantitation by HPLC due to their high polarity and lack of a UV absorbing chromophore. For such separations, ion exclusion chromatography is often used to detect and quantitate sugar alcohols. The method uses a combination of separation modes including gel filtration, ion-exchange, and affinity to resolve minor differences between the sugar alcohols.
Several different USP methods have been developed that take advantage of the unique selectivity provided by ion exclusion HPLC. In this application note, several different separations of sugars and sugar alcohols were performed that mimic the USP methods using Rezex RPM and RCU HPLC columns (2).
Material and Methods
Analyses were performed using an HP 1100 LC system (Agilent Technologies, Palo Alto, California) equipped with an autosampler. Analytes were detected using either a Shimadzu RID10A RI detector (Shimadzu Scientific, Columbia, Maryland) or a Polymer Labs ELS-2100 ELSD detector (Polymer Labs, Amherst, Massachusetts). HPLC columns used for analysis include Rezex RPM 100 × 7.8 mm and 300 × 7.8 mm columns (USP L34). In addition, the Rezex RCM 300 × 7.8 mm and Rezex RCU 250 × 4.0 columns (USP L19) (Phenomenex, Torrance, California) were used to provide alternate selectivity. Sugars and sugar alcohols were purchased from Sigma Chemicals (St. Louis, Missouri) and solvents were purchased from Thermo Fisher Scientific (Fairlawn, New Jersey).
The HPLC conditions used were dependent on the columns being used. For applications using the Rezex RPM and Rezex RCM columns, a flow rate of 0.6 mL/min was used. The applications using the Rezex RCU column used a flow rate of 0.2 mL/min. An isocratic method was used with water as the mobile phase and the column temperature was maintained constant between 75 °C and 85 °C, depending on the application run.
Results and Discussion
Efforts were undertaken to perform separations on different Rezex columns mimicking the different USP methods that are used to analyze excipients. Ion exclusion chromatography offers advantages over HILIC based methods in that they are isocratic, use a simple mobile phase (water), and can resolve chemically similar sugars. Depending on the metal salt used in the column (Lead {Pb2+ } or Calcium {Ca2+ }), one can achieve significantly different selectivities that can be used to optimize the desired separation.
An example of a sugar and sugar alcohol mixture run on the Rezex RPM 300 × 7.8 mm column is shown in Figure 1. In this application, the Rezex RPM separates 11 different sugars and sugar alcohols. Note how the column separates the isomers mannitol and sorbitol (peaks 8 and 11) with over 10 min of separation between the peaks. This type of separation is useful when a complex mixture of sugars is used in a formulation and one wishes to elucidate all the potential components. However, the long run time may be an issue when a large number of samples must be analyzed.
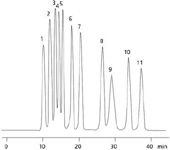
Figure 1: A mixture of sugars and sugar alcohols run on a Rezex RPM-Monosaccharide 300 Ã 7.8 mm column. The order of analytes is: (1) Stachyose, (2) Maltose, (3) Glucose, (4) Xylose, (5) Galactose, (6) Fructose, (7) Meso-erythritol, (8) Mannitol, (9) Salicin, (10) Xylitol, (11) Sorbitol. Note the wide separation between Mannitol (8), Xylitol (10) and Sorbitol (11).
For separations where less resolution is needed and better sample throughput is required, a shorter column can be used. In Figure 2, a shorter Rezex™ RPM 100 × 7.8 column is used to separate a mannitol and sorbitol mixture. This separation is similar to the published USP method for sorbitol and provides significant baseline separation of the two isomeric sugar alcohols (mannitol and sorbitol) in less than 15 min. These two chromatograms show that the Rezex RPM-Monosaccharide is useful for analyzing sugar alcohols that are often used as excipients in drug formulations.
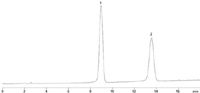
Figure 2: A mixture of mannitol (1) and sorbitol (2) run on a Rezex RPM-Monosaccharide 100 Ã 7.8 mm column using similar conditions to the USP method. Note the reduced run time of 15 min for separating the two excipients.
For situations where the selectivity of the Rezex RPM does not satisfy the separation requirements for a particular application, the Rezex RCM column can be used. The different metal impregnated Rezex phases take advantage of the different affinities that sugars have to various metals. This sugar-specific affinity allows one to investigate several different Rezex phases to determine which one provides the proper selectivity for a specific separation. However, for monosaccharide and sugar alcohols, the Rezex RPM and RCM/RCU phases are most commonly used.
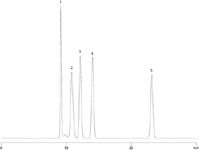
Figure 3: A mixture of sugars and sugar alcohols run on an alternative selectivity Rezex RCM-Monosaccharide 300 Ã 7.8 mm column for method development flexibility. The order of analytes is: (1) Sucrose, (2) Glucose, (3) Galactose, (4) Fructose, (5) Sorbitol. Note the different retention times for compounds compared to Figure 1, despite using similar column dimensions and flow rates. Rezex RCM offers different selectivity versus the Rezex RPM column.
Figure 3 shows the use of a Rezex RCM 300 × 7.8 mm column for separating a different mixture of sugars and sugar alcohols. Note the retention differences compared to the RPM column. The different selectivity of the calcium based phase is further shown in Figure 4, which is a different mannitol and sorbitol separation. This separation on the Rezex RCU 250 × 4.0 mm column mimics the USP method for mannitol and is an alternative for analyzing excipients in a sample.

Figure 4: A mixture of (1) mannitol and (2) sorbitol run on a Rezex⢠RCU-USP Sugar Alcohols 250 à 4.0 mm column using a method similar to the published USP method. The Rezex RCU offers a different option for the Mannitol and Sorbitol application.
Conclusion
The Rezex line of ion exclusion HPLC columns offers several different selectivity choices for analyzing sugars and sugar alcohols commonly found in pharmaceutical formulations. Rezex RPM, RCM, and RCU phases are well suited for use in USP methodologies for the analysis of mannitol, sorbitol, and other excipients. Shorter Rezex 100 × 7.8 mm columns may be used to reduce analysis times and increase sample throughput.
References
(1) K. Nuguru, C. Dolge, J. Easson, and E. Schwarz. AAPS PharmSci 11 (S1) (1999).
(2) R. Forget and S. Spagnoli. Journal of Pharmaceutical and Bio-medical Analysis 41(3) 1051–1055 (2006).
Phenomenex, Inc.,
411 Madrid Ave., Torrance, CA 90501
tel. (310) 212-0555, fax (310) 328-7768
Website: www.phenomenex.com

SEC-MALS of Antibody Therapeutics—A Robust Method for In-Depth Sample Characterization
June 1st 2022Monoclonal antibodies (mAbs) are effective therapeutics for cancers, auto-immune diseases, viral infections, and other diseases. Recent developments in antibody therapeutics aim to add more specific binding regions (bi- and multi-specificity) to increase their effectiveness and/or to downsize the molecule to the specific binding regions (for example, scFv or Fab fragment) to achieve better penetration of the tissue. As the molecule gets more complex, the possible high and low molecular weight (H/LMW) impurities become more complex, too. In order to accurately analyze the various species, more advanced detection than ultraviolet (UV) is required to characterize a mAb sample.















