Cleaning Up of α-Methylstyrene by Flash Chromatography
The isolation of α-Methylstyrene from the crude reaction mixture seems easy. However the reaction mixture contains some dimethyl sulfoxide and the oily wetting agent from the sodiumhydride.
A. Talamona, BÜCHI Labortechnik AG
The isolation of α-Methylstyrene from the crude reaction mixture seems easy. However the reaction mixture contains some dimethyl sulfoxide and the oily wetting agent from the sodiumhydride. Both of them are not UV-active and therefore invisible in the TLC.
TLC of the crude reaction mixture
TLC on silica gel Si60F254, developed in n-hexane/ethyl acetate 90:10, detection UV 254 nm.
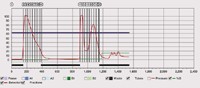
Figure 1: Chromatogram of α-Methylstyrene crude reaction mixture.
TLC of the the separated fractions
TLC on silica gel 60F254, developed in n-hexane-ethyl acetate 9:1, detection UV 254 nm.
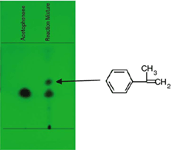
Figure 2: TLC of crude reaction mixture and pooled fractions 14â18.
Recovery
Fraction 11–12: 0.4 g colorless oily liquid.

Figure 3: TLC of crude reaction mixture and pooled fractions 11â12.
Sepacore configuration
Cartridge 40 × 150 mm, prepacked with silica gel 60, 40–63 µm
2 Pump Modules C-605
Fraction Collector C-660
Control-Unit C-620 with Sepacore Control software
UV Photometer C-635
Separation conditions
Eluent: n-hexane with 0%, 2%, and 15% ethyl acetate, step gradient
Flow rate: 100 mL/min
Sample: 3 g crude mixture, dissolved in toluene
Injection volume: 4.5 mL (although the mixture is liquid, the sample is not fully miscible with n-hexane and must be dissolved in toluene and injected directly onto the cartridge).
Fractions
2–10: mainly toluene and wetting agent
11–12: α-Methylstyrene
14–18: Acetophenone (reactant)
Buchi Corporation
19 Lukens Drive, Suite 400, New Castle, DE 19720
tel. (302) 652-3000, fax: (302) 652-8777
Website: www.mybuchi.com

SEC-MALS of Antibody Therapeutics—A Robust Method for In-Depth Sample Characterization
June 1st 2022Monoclonal antibodies (mAbs) are effective therapeutics for cancers, auto-immune diseases, viral infections, and other diseases. Recent developments in antibody therapeutics aim to add more specific binding regions (bi- and multi-specificity) to increase their effectiveness and/or to downsize the molecule to the specific binding regions (for example, scFv or Fab fragment) to achieve better penetration of the tissue. As the molecule gets more complex, the possible high and low molecular weight (H/LMW) impurities become more complex, too. In order to accurately analyze the various species, more advanced detection than ultraviolet (UV) is required to characterize a mAb sample.















