Improving Mass Load Capacity for Basic Compounds Using Charged Surface Hybrid (CSH) Technology Columns
Many modern stationary phases suffer from poor chromatographic performance for basic compounds at low pH, especially when using low-ionic strength mobile phases such as formic acid.
Kenneth J. Fountain and Hillary B. Hewitson, Waters Corporation
Many modern stationary phases suffer from poor chromatographic performance for basic compounds at low pH, especially when using low-ionic strength mobile phases such as formic acid. While the exact cause for this behavior is not completely known, it is thought that variations in the surface charge of high purity chromatographic materials can have a large impact on the chromatography, even at fairly low analytical loads. The advantages of using Charged Surface Hybrid (CSH) technology columns for impurity profiling in stability indicating methods and/or forced degradation studies of commercial drug substances will be demonstrated using typical LC–MS conditions.
Experimental
UPLC System: Waters ACQUITY UPLC® H-Class System with a PDA detector and an SQ mass detector
Columns: Waters ACQUITY UPLC CSH C18, 2.1 × 50 mm, 1.7 µm
Fully porous silica C18, 2.1 × 50 mm, 1.8 µm
Superficially porous C18, 2.1 × 50 mm, 1.7 µm
Flow rate: 0.6 mL/min
Detection: 254 nm
All other conditions are specified in figure captions.
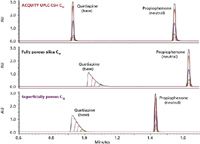
Figure 1: Loading capacity for quetiapine (base) and propiophenone (neutral) on three different columns in formic acid conditions. Mixtures containing 2 to 200 µg/mL each of quetiapine and propiophenone were prepared in 50/50 CH3CN/H2O. Mobile phase A was 0.1% formic acid in water, and mobile phase B was 100% acetonitrile. The gradient was from 5 to 95% B in 2.5 min. Injection volume was 5 µL. Column temperature was 30 °C.
Results and Discussion
In order to compare the loadability of different stationary phases at low pH, a mixture of a neutral and a basic compound were analyzed at different mass loads on an ACQUITY UPLC CSH C18 column, a fully porous silica C18 column, and a superficially porous C18 column using formic acid mobile phases (Figure 1). As the mass load increases for the neutral compound (propiophenone), all three columns exhibit symmetrical peak shape and no mass overloading is observed. However, when the mass load of quetiapine (a weak base) is increased on the three columns, differences in performance can be observed. The fully porous silica C18 and superficially porous C18 columns quickly become overloaded under the same gradient conditions for this compound. This occurs with a mass load as low as 50 ng on-column (10 µg/mL). The ACQUITY UPLC CSH C18 column, however, does not show mass overload for any of the concentrations of quetiapine, indicating that the performance of this column is identical for all analytes including basic compounds, especially in low-ionic strength conditions.

Figure 2: UV chromatograms for the analysis of trace impurities on ACQUITY UPLC CSH C18 and superficially porous C18 columns. Mobile phase A was water, mobile phase B was 100% acetonitrile, mobile phase C was 2% formic acid in water, and mobile phase D was 2% TFA in water. The gradient was from 25 to 35% B in 2 min, followed by an increase to 95% B in 1 min. Different percentages of mobile phase C and D were programmed to get the concentration of acidic modifier shown. Injection volume was 5 µL. Column temperature was 40 °C.
The ability to load large amounts of basic compounds in low-ionic strength mobile phases has many practical implications, especially for impurity profiling and identifying unknown impurities in stability indicating methods and/or forced degradation studies using LC–MS. For this type of work, a large amount of the active pharmaceutical ingredient (API) must be injected in order to see low-level impurities in the baseline. To demonstrate this, a 0.5 mg/mL solution of imipramine spiked with 0.1% of amitriptyline was injected onto ACQUITY UPLC CSH C18 and superficially porous C18 columns using formic acid mobile phases (Figure 2). Since amitriptyline elutes on the tail end of the imipramine peak, this example represents the challenges faced by LC–MS scientists who are routinely trying to identify unknown low-level impurities in a new synthesis pathway or forced degradation sample. Since the imipramine is massively overloaded on the superficially porous C18 column, the ability to see the 0.1% impurity on the tail is lost with formic acid (Figure 2B). Resolution can be improved on this column by substituting formic acid with TFA (Figure 2C). However, when trying to get mass spectral information on unknown impurities in the baseline, TFA is not a desirable mobile phase additive, since it severely suppresses ionization. In the case of the superficially porous C18 column, the impurity peak cannot be seen in the total ion chromatogram, and thus, no molecular weight information can be obtained (Figure 3B). Since the imipramine peak is not heavily overloaded on the ACQUITY UPLC CSH C18 column under formic acid conditions (Figure 2A), one can obtain both full resolution of the impurity and molecular weight information in the same run (Figure 3A).
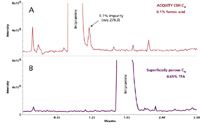
Figure 3: Total ion chromatograms (TICs) for the trace impurity analysis shown in Figure 2.
Conclusions
CSH Technology columns have excellent loadability for basic compounds, and can be routinely used in LC–MS applications where low-ionic strength mobile phases (e.g., formic acid) are typically used. This allows separations of basic compounds to be performed at high mass loads without the need for ion-pairing agents such as TFA to improve peak shape, which is useful for applications where low-level baseline impurities must be detected in the presence of a large amount of API.
© 2011 Waters Corporation. Waters, ACQUITY UPLC, The Science of What's Possible and CSH Technology are trademarks of Waters Corporation.
Waters Corporation
34 Maple Street, Milford, MA 01757
tel. (508) 478-2000, fax (508) 478-1990
Website: www.waters.com

The Benefits of Custom Bonded Silica
April 1st 2025Not all chromatography resins are created equal. Off-the-shelf chromatography resins might not always meet the rigorous purification requirements of biopharmaceutical manufacturing. Custom bonded silica from Grace can address a wide range of separation challenges, leading to real performance improvements. Discover more about the latest innovations in chromatography silica from Grace, including VYDAC® and DAVISIL®.
5 Things to Consider When Selecting a Chromatography Silica
April 1st 2025Particularly in the pharmaceutical industry, drug purity isn’t just a goal – it’s essential for achieving safety, stability and efficacy. However, purification is easier said than done, especially with challenging molecules like DNA and RNA “oligonucleotides,” due in large part to their diversity and the range of impurities that can be generated during production. Enter DAVISIL® chromatographic silica, with a wide range of pore diameters and particle sizes to meet your specific application, performance and sustainability requirements. Before you choose the chromatography resin for your next purification application, take a look at these 5 considerations.
Automating Protein Purification: Efficiency, Yield, and Reproducibility
March 27th 2025Recent advancements in automated protein purification stress the importance of efficiency, scalability, and yield consistency. This eBook compares different purification platforms, highlighting their impact on downstream applications and demonstrating how automation enhances throughput and process control.
MilliporeSigma: Ultrapure Water for Sensitive LC-MS Analysis of Pesticides
March 25th 2025The aim of the study was to illustrate the efficiency of Milli-Q® water purification systems in eliminating pesticides from tap water, thereby producing and delivering reliable and consistent-quality ultrapure water suitable for pesticides analysis















