Evaluation of Several Columns and Solvents for Gel Permeation Chromatography (GPC) Clean-Up of Fish Tissue Prior to PCB Analysis
The determination of PCBs, as well as other environmental contaminants, in fish tissue requires extensive sample cleanup prior to analysis by gas chromatography with an electron capture detector (GC-ECD).
Michael Halvorson1, Curtis Hedman2, and Thomas Gibson2, 1Gilson, Inc. and 2Wisconsin State Lab of Hygiene
The determination of PCBs, as well as other environmental contaminants, in fish tissue requires extensive sample cleanup prior to analysis by gas chromatography with an electron capture detector (GC-ECD). GPC is a size-exclusion clean-up procedure that uses organic solvents and a hydrophobic gel to separate macromolecules. The most common GPC clean-up procedures for fish extract cleanup utilizes a glass column containing 60 g of Envirobeads™ SX-3 resin with a mobile phase of 1:1 dichloromethane/cyclohexane. Although this column is highly efficient in removing lipids, sample throughput is limited and utilizes large amounts of solvent per sample. The purpose of this study was to use a Gilson GX-271 Automated GPC Clean-up System to evaluate alternatives to the traditional column.
Experimental Conditions
10 g of ground fish tissue spiked with PCB congeners was extracted using dichloromethane and further purified with a Florisil® column prior to GPC clean-up.
Flow rates were at 5 mL/min except for the Jordi column (1 mL/min). Injection volumes were 5 mL of extract or 1 mL of extract. The following columns were used:
- OI Analytical glass column (60 g Envirobeads SX-3) and mobile phase of 1:1 dichloromethane (DCM)/cyclohexane (CYX)
- Phenomenex EnviroSep-ABC column (21 × 350mm) and mobile phase of 1:1 DCM/CYX or mobile phase of 100% DCM
- Jordi Flash Gel Fluorinated DVB (10 × 250mm); 100% DCM
- OI Analytical Optima column (21 g Envirobeads SX-3), 1:1 ethyl acetate/CYX
PCBs were analyzed with an Agilent HP5890-II GC with a DB5 column (60 m × 0.25 mm ID, 0.1 μM phase) configured with ECD.
Results
See Table I and Table II.
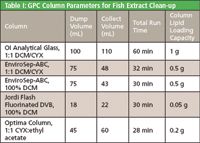
Table I: GPC Column Parameters for Fish Extract Clean-up
Conclusion
All tested columns were able to separate the lipids in fish extracts from the PCBs. The traditional glass column achieved excellent separation and had the highest lipid loading capacity. However, it utilized the most solvent and had the lowest throughput.
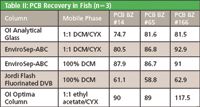
Table II: PCB Recovery in Fish (n=3)
The EnviroSep-ABC column, using either of the mobile phases tested, had twice the throughput of the traditional column and used half the amount of solvent. It provided very good recoveries. Lipid loading capacity was one half that of the traditional column.
GPC clean-up readily lends itself to automation and helps reduce GC maintenance costs by preventing buildup on GC injection ports and columns.
Envirobeads is a registered trademark of Bio-Rad Laboratories, Inc.
EnviroSep is a trademark of Phenomenex Corporation.
Florisil is a registered trademark of U.S. Silica Company.
Optima is a trademark of OI Analytical Corporation.

Gilson, Inc. World Headquarters
3000 Parmenter Street, P.O. Box 620027 Middleton, WI 53562
tel. (608)836-1551; fax: (608)831-4451
Website: www.gilson.com.

Separation of Ultra-Short and Long Chain PFAS Compounds Using a Positive Charge Surface Column
December 11th 2024A separation of ultra-short and long chain PFAS (C1-C18) is performed on a HALO®PCS Phenyl-Hexyl column along with a HALO®PFAS Delay column which demonstrates excellent retention for both hydrophilic and hydrophobic analytes.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)














