The Analysis of Melamine in Milk Powder Using HILIC with UV Detection
The analysis of melamine in milk and other food products became urgent when first the pet food, and later the milk scandal were discovered in the U.S. and China.
Wen Jiang and Felicia Nkem Ihunegbo, Merck SeQuant AB
The analysis of melamine in milk and other food products became urgent when first the pet food, and later the milk scandal were discovered in the U.S. and China. Analytical techniques, e.g. ion-pair reversed-phase chromatography, hydrophilic interaction liquid chromatography (HILIC), ELISA, and GC–MS have been applied to separate and determine melamine (1).
HILIC is a straightforward chromatographic separation mode, which is suitable for separating polar and hydrophilic compounds, such as melamine without any sample derivatisation or ion-pair additive in the mobile phase. Several studies have already shown that the combination of HILIC with mass spectrometry (MS) can be used to determine melamine at trace level (2–7). Zwitterionic HILIC stationary phase was also used to simultaneously determine both melamine and cyanuric acid in one single chromatographic run, which provides more crucial information on the melamine fraud (3,4,6).
However, due to limited availability of MS instrument and the following analysis cost, it is still important to develop a method which can combine HILIC and UV detection for screening of melamine in milk products. This study describes the sample preparation and application of HILIC-UV for this.

Sample Preparation
Standard Samples
Standard solutions were prepared from the stock solution of 100 mg/mL melamine stock solution in water and diluted with 85/15 (v/v) acetonitrile/water.
Preparation of pre-spiked melamine in milk powder extract
2 g (±0.1 g) commerical milk powder was weighted in a 50 mL plastic Falcon tube, thereafter 7.125 mL aqueous solution containing either 2.5% ammonia, perchloric acid, or formic acid, respectively, and finally 0.375 μL of 100 μg/mL melamine standard stock solution was added. The mixture was mixed on a Vortex shaker till it was uniform (~1 min). 7.5 mL of acetonitrile were added into tube and mixed on a Vortex shaker for 30 s. The tube was then shaken for 30 min on an orbital shaker, and was sonicated for 20 min in an ultrasonic bath.
An aliquot of 2 ml of milky supernatant was transferred into a 2 mL centrifuge tube, and centrifuged at 14000 rpm for 10 min. Injection samples were then prepared by mixing 0.35 mL of supernatant with 1.4 mL acetonitrile in a 2 mL centrifuge tube, and centrifuged for 10 min at 14000 rpm. The total melamine concentration dilution was about 38 times.
Preparation of post-spiked melamine in milk powder extract
The first step for preparing post-spiked samples was the same as that for pre-spiked extracts with the exception of no melamine addition. The total volume of aqueous solution was 7.5 mL.
An aliquot of 2 ml of milky supernatant was transferred into 2 mL centrifuge tube, and centrifuged at 14000 rpm for 10 min. Blank and post-spiked extracts were prepared by mixing 0.35 mL of supernatant and an aliquot of 0, 17.5, 35, 87.5, 175, and 350 μL of 5 μg/mL melamine standard solution (in 85/15 acetonitrile/water), and added with acetonitrile to a total volume of 1.75 mL. Thus the injection sample solutions correspondingly containing 0, 0.05, 0.1, 0.25, 0.5, 1 μg/mL melamine were prepared. The total dilution of melamine concentration was about 38 times.
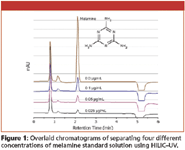
Figure 1
Results and Discussion
Figure 1 illustrates the separation of melamine standard at various concentration levels. The limit of quantification (LOQ) was 0.015 μg/mL. Calibration curves (peak area and height) showed excellent linearity (R2 > 0.9999) at concentrations between 0.025 and 1 μg/mL.
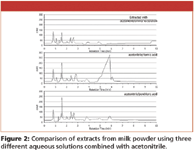
Figure 2
Three different extraction solutions were critically investigated for the optimum extraction of melamine from milk powder. Figure 2 shows the HILIC-UV analysis of extracts from milk powder using the mixture of 50/50 (v/v) acetonitrile/2.5% ammonia, perchloric acid, and formic acid, respectively. It was concluded that perchloric acid is the most suitable extraction solution according to its lower interference on the separation and better recovery, which was 78% compared to 52 and 41% for ammonia and formic acid, respectively.
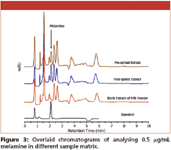
Figure 3
The analyses of melamine in standard solution, the blank, pre- and post-spiked milk powder extracts, extracted with 50/50 (v/v) acetonitrile/2.5% perchloric acid, is presented in Figure 3. It was found the peak heights of post-spiked milk extracts were very similar to those of standard solution with the same concentration of melamine. However, they were lower for the pre-spiked sample. The calibration curves (peak area and height) prepared from the extracts spiked with melamine showed the good linearity (R2 > 0.9954) in the concentration range of 0.05 to 1 μg/mL. Moreover, Figure 4 shows that it is possible to use HILIC-UV method to analyse 0.1 μg/mL melamine in extracts from milk powder, which corresponds to 3.8 μg/g in milk powder.
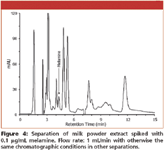
Figure 4
Conclusion
Melamine has good retention and separation in HILIC separation mode on a zwitterionic stationary phase. It is possible to use UV detection to detect the melamine both in standard and spiked milk extracts. Using a mixture of acetonitrile and perchloric acid as extraction solution, melamine can be efficiently extracted from milk powder with relatively good recovery. The described sample preparation for chromatographic injections is simple and reproducible. By comparing the chromatogram from both blank and melamine tainted sample, HILIC-UV method is useful for screening melamine contaminated milk and other food products.
References
(1) M. Lin, Front. Chem. Eng. China, 2009, 3, 427–435.
(2) W.C. Andersen, et al. Laboratory Information Bulletin, LIB No. 4396, http://www.fda.gov/Food/ScienceResearch/LaboratoryMethods/DrugChemicalResiduesMethodology/ucm071796.htm.
(3) D.N. Heller, C.B. Nochetto, Rapid Commun. Mass Spectrom. 2008 (22):3624–32.
(4) S. Turnipseed et al. Laboratory Information Bulletin, LIB No. 4421, http://www.fda.gov/Food/ScienceResearch/LaboratoryMethods/DrugChemicalResiduesMethodology/ucm071637.htm.
(5) M. Smoker et al. Laboratory Information Bulletin, LIB No. 4422, http://www.fda.gov/Food/ScienceResearch/LaboratoryMethods/DrugChemicalResiduesMethodology/ucm071673.htm.
(6) Merck SeQuant Technical Note TN-008, http://www.sequant.com/technicalnotes.
(7) W.C. Andersen, et al. J. Agric. Food Chem. 2008, (56):4340–7.

EMD Chemicals, Inc. An affiliate of Merck KGaA, Darmstadt, Germany
480 South Democrat Road, Gibbstown, NJ 08027
tel. (800)222-0342; Website: www.emdchemicals.com

Assessing Thorium-Peptide Interactions Using Hydrophilic Interaction Liquid Chromatography
February 4th 2025Paris-Saclay University scientists used hydrophilic interaction liquid chromatography (HILIC) coupled to electrospray ionization mass spectrometry (ESI-MS) and inductively coupled plasma mass spectrometry (ICP-MS) to assess thorium’s interaction with peptides.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)















