Ion Chromatography Method for Determination of Risedronate in Pharmaceutical Preparations
Risedronate, a pyridinyl bisphosphonate, is one of the most popular first-line drugs in the treatment of osteoporosis, Paget's disease, and other bone fragility conditions. In October 2010, the United States FDA approved a second-generation risedronate in a delayed-release formulation.
Risedronate, a pyridinyl bisphosphonate, is one of the most popular first-line drugs in the treatment of osteoporosis, Paget's disease, and other bone fragility conditions. In October 2010, the United States FDA approved a second-generation risedronate in a delayed-release formulation.
An assay of the active pharmaceutical ingredient (API) and determining the presence of impurities and other related compounds is critical to ensure the formulation of the drug is safe and effective. The U.S. Pharmacopeia (USP) monograph describes an ion chromatography (IC) method to assay risedronate in the drug substance and product. The method specifies the Thermo Fisher Scientific Dionex IonPac™ AS7 column with an EDTA eluent at pH 9.5 and UV detection at 263 nm (1). The method was determined to be simple, rugged, and accurate.
Experimental
Analyses were performed using a Thermo Fisher Scientific Dionex ICS-3000 system, autosampler, and Thermo Fisher Scientific Dionex Chromeleon™ Chromatography Data System (CDS) software. All samples and standards were prepared according to the procedures described in the USP monograph for risedronate sodium.
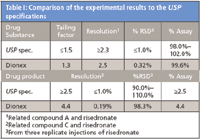
Table I: Comparison of the experimental results to the USP specifications
Results and Discussion
The USP monograph for risedronate sodium was used to assay the API in the drug substance and product, available in 5–75 mg strength tablets. This method is based on an anion-exchange separation with a 4 × 250 mm Dionex IonPac AS7 (USP L48) column using an EDTA eluent to resolve risedronate from related compounds. As shown in Table I, the results exceeded the USP monograph specifications. The method separated risedronate in <15 min as shown in Figure 1, and the resolution between related compounds A and C exceeded the USP specifications. The method ruggedness was also evaluated by varying the column temperature, eluent pH, and different Dionex IonPac AS7 columns from the same lot. For each variable, the results met or exceeded the USP specifications to demonstrate that the method is rugged. The results from this study demonstrated a simple, rugged, and accurate method to assay risedronate sodium in the drug substance and product. Therefore, the method is suitable for QC labs to ensure that the drug is safe and effective for consumers.

Figure 1: Chromatogram showing separation between related compound A, related compound C, and risedronate. Peaks 1: compound A, peak 2: compound C, peak 3: risedronate. Column: Dionex IonPac AG7/AS7 column set (USP L48); eluent: 4.8 mM EDTA, pH adjusted to 9.5 ± 0.1 with 5 M NaOH; flow rate: 0.8 mL/min; temp: 25 °C; inj. vol.: 20 μL; detection: UV at 263 nm.
Reference
(1) USP 34 – NF 29, 4166 (2011).
Chromeleon and IonPac are registered trademarks of Thermo Fisher Scientific and its subsidiaries.
Dionex, now part of Thermo Fisher Scientific
1228 Titan Way, P.O. Box 3603, Sunnyvale, CA 94088
tel. (408) 737-0700, fax (408) 730-9403
Website: www.thermoscientific.com/dionex

Reversed-Phases for LC Deliberately Doped with Positive Charge: Tips and Tricks for Effective Use
May 13th 2025In this month's edition of LC Troubleshooting, Dwight Stoll and his fellow researchers discuss both the benefits (improved peak shape/loading) and challenges (excessive interaction) associated with charge-doped reversed-phase (RP) columns for both analytical and preparative separations.
Evaluating the Accuracy of Mass Spectrometry Spectral Databases
May 12th 2025Mass spectrometry (MS) can be effective in identifying unknown compounds, though this can be complicated if spectra is outside of known databases. Researchers aimed to test MS databases using electron–ionization (EI)–MS.
Detecting Hyper-Fast Chromatographic Peaks Using Ion Mobility Spectrometry
May 6th 2025Ion mobility spectrometers can detect trace compounds quickly, though they can face various issues with detecting certain peaks. University of Hannover scientists created a new system for resolving hyper-fast gas chromatography (GC) peaks.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)



















