Identification of Metabolites of Doxylamine and Dextromethorphan Using UHPLC with High-Performance Time-of-flight Mass Spectrometry
Ultrahigh-performance TOF–MS is utilized for the identification of a common pharmaceutical compound's metabolites. Mass spectral analysis is combined with UHPLC and post-source fragment ion generation to facilitate analysis.
Ultrahigh-performance TOF–MS is utilized for the identification of a common pharmaceutical compound's metabolites. Mass spectral analysis is combined with UHPLC and post-source fragment ion generation to facilitate analysis.
The detection and identification of metabolites of pharmaceutical significance by LC–MS remains a substantial challenge. Characterization is often achieved using data-dependent MSn or convoluted MS-MS experiments at a cost of analysis and processing time. Here, accurate mass measurement, rapid spectral acquisition, and high sensitivity available in ultrahigh-resolution time-of-flight mass spectrometry are used to identify metabolites of doxylamine and dextromethorphan in human urine in less than 2 min with minimal cleanup.
Experimental
Urine samples were collected immediately prior to dosing (6.5 mg doxylamine, dextromethorphan, and polyethylene glycol [PEG]) and approximately 8 h post-dosing. Salt and protein were removed and a portion of urine (ca. 5 μL) was analyzed as follows:
Column: 1.9 μm Hypersil phenyl 50 × 1.0 mm (45 °C)
Mobile phase: 0.1% formic acid in water (A), metha nol (B)
Gradient: 0% to 97.5% MeOH in 2.6 min at 0.375 mL/min
Mass spectrometer: CITIUS™ LC-HRT (LECO ESI source; positive mode) using R = 50,000 (FWHH), 15 eV collision offset for fragmentation, 5 spectra/s precursor and 5 spectra/s fragment (in an alternating fashion) over 50-2250 m/z. Processing was achieved using ChromaTOF-HRT software.
Results
A typical set of extracted chromatograms from the dosed urine sample is shown in Figure 1. The sample data were integrated and processed and ions whose signals are shown in Figure 1 exist only in the dosed sample. The analytes deconvoluted and identified include doxylamine, doxylamine oxide, desmethyl and didesmethyldoxylamine, dextromethorphan, dextrorphan, and dextromethorphan glucuronide. Identification is based on accurate m/z values of parent and fragment ions (±1 ppm and 2 ppm, respectively) and relative isotope abundance comparison (errors of typically less than 5%). The high abundance of PEGs in the urine samples did not interfere with the detection of the metabolites or parent compounds.
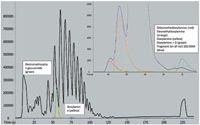
Figure 1: Total ion chromatogram (TIC; black) and select extracted precursor ion chromatograms (±2 ppm mass window) from the dosed sample. The colored traces are identified in the captions. The extracted fragment ion at 182.0946 (doxylamine core) is ±3 ppm. (PEGs are seen between 50 and 100 s in the TIC.)
Discussion
Metabolites of doxylamine and dextromethorphan have been readily detected and identified using precursor and fragment ions generated in an ultrahigh-resolution time-of-flight mass spectrometer (R = ca. 50,000 at <2 ppm mass error). Minimal preparation was required and limited separation achieved using fast HPLC. The instrument's gridless mirrors and periodic ion beam refocusing affords these capabilities at no cost to sensitivity. These analyses were achieved without need for pre-information required with analyses undertaken using other MS-MS systems.
LECO Corporation
3000 Lakeview Avenue, St. Joseph, MI 49085
tel. (269) 985-5496, fax (269) 982-8977
Website: www.leco.com

SEC-MALS of Antibody Therapeutics—A Robust Method for In-Depth Sample Characterization
June 1st 2022Monoclonal antibodies (mAbs) are effective therapeutics for cancers, auto-immune diseases, viral infections, and other diseases. Recent developments in antibody therapeutics aim to add more specific binding regions (bi- and multi-specificity) to increase their effectiveness and/or to downsize the molecule to the specific binding regions (for example, scFv or Fab fragment) to achieve better penetration of the tissue. As the molecule gets more complex, the possible high and low molecular weight (H/LMW) impurities become more complex, too. In order to accurately analyze the various species, more advanced detection than ultraviolet (UV) is required to characterize a mAb sample.















