GC Columns for the Analysis of Polynuclear Aromatic Hydrocarbons
SGE Analytical Science Application Note
This application note outlines the use of 5% phenyl and 50% phenyl polysilphenylene siloxane stationary phases to resolve isomer separations for PAH analysis.
Polynuclear Aromatic Hydrocarbons (PAH) are prevalent in the environment following their use in manufacturing processes or as byproducts formed through the incomplete combustion of organic materials. The larger PAHs (containing ≥6 benzene rings) tend to be extremely persistent in soil as their water solubility and mobility decreases substantially with their increasing molecular weight. Of the 24 PAHs that are frequently monitored, the European Union (EU) and the Scientific Committee on Food (SCF) recommend a set of 15 standard PAHs plus 1 priority PAH. The US EPA mandates the determination of 16 priority PAH compounds including benzo(j)fluoranthene, dibenz(a,h)acridine and dibenzo(a,e)pyrene, which are also included in the SCF regulatory requirements, but these are difficult to analyse via conventional test conditions. In GC analyses employing a 5% phenyl stationary phase, benzo(j)fluoranthene co-elutes with benzo(b)fluoranthene, therefore its determination must be reported as a combined sum of the two isomers. Where regulations mandate uniquely reported concentrations for individual analytes, this is an ineffective technique. This application note describes how to achieve greater separation of these isomers by GC analysis using polysilphenylene siloxane stationary phases.
Experimental Conditions
Routine PAH Analysis
The SGE BPX5 (5% Phenyl Polysilphenylene-siloxane) can be effectively used to analyse the USEPA 16 component priority pollutant PAH mixture. Figure 1 shows the critical pairs that must be sufficiently resolved to ensure proper identification in GC–MS analysis due the pairs sharing the same quantification ions. The temperature programme was optimized to achieve the best separation of the critical isomer pairs. No internal standards or surrogates were used in these evaluations.
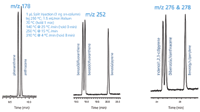
Figure 1: BPX5 30 m à 0.25 mm à 0.25 μm. Routine 16 component PAH analysis - Critical pairs.
Expanded PAH Analysis
Three individual components were added to the routine 16 component standard to mimic a typical expanded scan: 1-methylnapthalene, 2-methylnapthalene and benzo(j) fluoranthene. As commonly seen with 5% phenyl stationary phases, the benzo(j)fluoranthene isomer is not completely resolved on the SGE BPX5. The increase in phenyl functional groups in the SGE BPX50 (50% Phenyl Polysilphenylenesiloxane) stationary phase increases retention of this phenyl containing compound to meet the quantitative and qualitative requirements. The temperature programme optimized for SGE BPX5 was used for the analysis.
Results
The SGE BPX50 results showed significant improvement for the fluoranthene isomers over the SGE BPX5 phase as shown in Figure 2. The benzofluoranthene isomers are nearly baseline resolved. There was, however, a slight loss in resolution of phenanthrene and anthracene but it is still sufficient to qualify these compounds effectively.

Figure 2: BPX50 30 m à 0.25 mm à 0.25 μm. Expanded PAH analysis, 19 component - Critical pairs.
Conclusion
Simple changes in stationary phases can have a profound effect on selectivity for PAHs. The SGE BPX50 is an excellent phase for the analysis of expanded PAH scans, specifically the benzo-fluoranthene isomers. These isomers are nearly baseline resolved and can easily be quantified as distinct peaks. One limitation is the slight loss in resolution of the phenanthrene/anthracene isomers, analysts should consider this affect particularly when performing the analysis via GC–MS SIM mode.

SGE Analytical Science
1 Potters Lane, Kiln Farm, Milton Keynes, MK11 3LA, UK
tel: +44 800 2790 8999 fax: +44 800 2626 2609
E-mail: Europe@sge.com Website: www.sge.com

Regulatory Deadlines and Supply Chain Challenges Take Center Stage in Nitrosamine Discussion
April 10th 2025During an LCGC International peer exchange, Aloka Srinivasan, Mayank Bhanti, and Amber Burch discussed the regulatory deadlines and supply chain challenges that come with nitrosamine analysis.



















