The Synthetic Cannabinoid Chemical Arms Race and Its Effect on Pain Medication Monitoring
Special Issues
An investigation of C18 and phenyl-hexyl column chemistries for definitive identification of 13 synthetic cannabinoid metabolites in patient samples.
Sheng Feng, Brandi Bridgewater, Gregory L. McIntire, and Jeffrey R. Enders, Ameritox Ltd., Greensboro, North Carolina, USA.
In recent years, synthetic cannabinoids (“K2” or “spice”) have experienced a boom in popularity. The negative health effects of these drugs coupled with their increasing popularity led to placement onto Schedule I by the Drug Enforcement Administration (DEA). In response, the chemists behind these illicit compounds frequently invent new compounds to circumvent the law. Thus, new classes and new examples within classes of “spice” continue to become available for illicit use. In this paper, we examine the use of two column chemistries (C18 and phenyl-hexyl) in an effort to definitively identify synthetic cannabinoid compounds in patient samples. Distinct synthetic cannabinoid compounds interact differently with specific stationary phases and the hope is that this extra dimension of data will help to rule out similar interferent compounds that would otherwise cause false-positive results.
Synthetic cannabinoids, commonly known as “K2”, “spice”, or “synthetic marijuana”, are often sprayed onto or mixed with dried plant materials and sold in convenience stores, gas stations, smoke shops, and on the internet. This ready availability causes confusion about their safety and legality (1). In recent years, synthetic cannabinoids have become increasingly popular among adolescents and young adults as one of several frequently abused substances. These synthetic drugs mimic delta-9-tetrahydrocannabinol (THC), but can be much more potent, which results in psychoactive doses less than 1 mg (2). In fact, synthetic cannabinoids, which have a similar psychoactive effect as cannabis, have strong addictive properties often coupled with unknown physiological impacts on users. A recent study indicates that the use of synthetic cannabinoids can be a cause of death (3).
Because of the high abuse potential and lack of medical knowledge or usage, these synthetic cannabinoids have been added to the Schedule I list by the United States Drug Enforcement Administration (DEA), as “necessary to avoid imminent hazard to the public safety” (4). In response, the chemists instigating this illegal proliferation have synthesized many new K2 analogues by slightly altering chemical structures (5). Therefore, compared with the relatively stagnant pool of other compounds, such as opiates, that most pain medication monitoring laboratories deal with, the number of agents on the list of synthetic cannabinoids has been and continues to be increasing (6). Testing for synthetic cannabinoids has become a routine demand among pain treatment clinics.
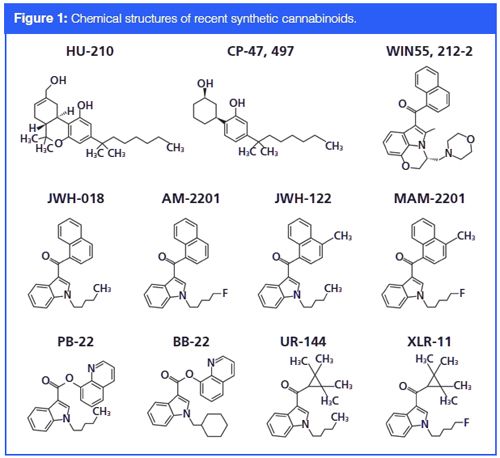
There are various types of synthetic cannabinoids with different modifications on the core structure. The first THC analogues, including HU-210 (7) and CP-47, 497 (8), were synthesized in the 1980s. Their inventions allowed the discovery of G protein-coupled receptors, CB1 and CB2 (9). Later on, a structurally different analogue, WIN55, 212â2, was reported. Surprisingly, WIN55, 212-2 has higher affinity towards CB1 and CB2 than THC does (10). Subsequently, John W. Huffman developed a series of “JWH compounds” by simply replacing the aminoalkyl group in WIN55, 212-2 with simple alkyl chains (11). JWH-018 has become the prototypical JWH compound. Synthetic cannabinoids have also been developed by generating fluoroâderivatives of JWH compounds. For example, AM-2201 and MAMâ2201 are fluoro-derivatives of JWH 018 and JWH 122, respectively (12). By replacing the ketone in the 3-indole position of JWH-018 with an ester linkage, PB-22 and BB-22 compounds have been synthesized (13). Furthermore, another class of synthetic cannabinoids contains the tetramethylcyclopropyl ketone indoles, such as UR-144 and its fluoro-derivative, XLR-11 (14). Both UR-144 and XLR-11 have cyclopropyl rings, and are therefore likely to exhibit similar retention times in liquid chromatography (LC).
The increasing number of sophisticated reversed-phase LC separations has led to the need for optimized stationary phases to offer improved selectivity and efficiency (15). In the present work, we investigate C18 and phenyl-hexyl column chemistries for definitively identifying 13 synthetic cannabinoid metabolites in standards and patient samples.
Materials and Methods
Chemicals: Reference standards of AKB48 5-hydroxypentyl metabolite, AKB48 pentanoic acid metabolite, AM2201 4-hydroxypentyl metabolite, BB-22 3-carboxyindole metabolite, JWH-018 pentanoic acid metabolite, JWH-073 butanoic acid metabolite, JWH-122 5-hydroxypentyl metabolite, MAM-2201 4-hydroxypentyl metabolite, PB-22 3-carboxyindole metabolite, PB-22 pentanoic acid metabolite, UR-144 5-hydroxypentyl metabolite, UR-144 pentanoic acid metabolite, and XLR11 4-hydroxypentyl metabolite were purchased from Cayman Chemical Company. Reference standards of 11-nor-9-Carboxy-Δ9-THC (THCA), THCA glucuronide, and THCA-D9 were purchased from Cerilliant Corporation. Solvents including methanol (optima grade), acetonitrile (optima grade), and formic acid (88%) were purchased from VWR. Dimethylsulphoxide (DMSO) (HPLC grade), ethyl acetate (optima grade), and ammonium hydroxide (A.C.S. Plus) were purchased from Fisher Scientific. Recombinant β-glucuronidase enzyme was purchased from IMCS. Drugâfree normal human urine (NHU) was purchased from UTAK Laboratories, Inc. Deionized (DI) water was obtained in-house from a Thermo Scientific Barnstead Nanopure water purification system.
Sample Preparation: Reference standards not already in solution were dissolved in DMSO. Solutions of reference standards were aliquoted, dried, and reconstituted with NHU to make a low calibrator concentration at 1 ng/mL for all analytes except BB-22 3-carboxyindole metabolite and THCA with low calibrator levels at 5 ng/mL and 10 ng/mL, respectively. A high calibrator concentration of 100 ng/mL in NHU was used for all analytes. An 18.5-ng/mL THCA glucuronide hydrolysis–negative control (HNEG) and a 20-ng/mL positive control (20CON) were similarly prepared in NHU. This protocol uses THCA glucuronide as a hydrolysis control. Accordingly, every curve and patient batch has a hydrolysis control that contains 18.5 ng/mL of THCA glucuronide. For this control to be considered passing, it must return the expected THCA (parent) concentration within 30%.
Into 13 mm × 10 mm borosilicate glass tubes, 800 µL of calibrators, controls, and samples were each aliquoted and combined with 200 µL of THCA-D9 (2.5 µg/mL)/recombinant β-glucuronidase (1000 enzyme units/mL) solution in 25:25:50 methanol–DI water–pH 7.5 phosphate buffer. All samples were vortexed, transferred to SPEware CEREX PSAX 3 mL/35 mg extraction columns in sample racks by SPEware, and heated in a VWR Symphony oven for 15 min at 60 °C. Samples were cooled for 5 min and placed on an automated liquid dispensing-II (ALD-II) system for extraction. A light positive pressure was applied to push the samples onto the solid-phase extraction (SPE) packing. The ALD-II system then washed columns with 85:14:1 DI water–acetonitrile–ammonium hydroxide, washed with 30:70 DI water–methanol, and finally eluted samples into 1800-μL amber autosampler vials using 98:2 ethyl acetate–formic acid. Samples were dried under nitrogen for ~35 min at 25 °C in a SPEware Cerex sample concentrator, then each reconstituted with 400 μL of 50:50 DI water–methanol. Samples were capped, vortexed for 20 s, and spun for 5 min at 4000 rpm on a Sorvall ST 40 centrifuge.
Patient Sample Collection: Patient urine specimens were collected at clinics and shipped to Ameritox Ltd. These de-identified patient samples were treated similarly to standards, that is, they were diluted, extracted, and subjected to liquid chromatography–tandem mass spectrometry (LC–MS–MS). Patient samples were selected for this study that were deemed positive by the current method’s criteria, but were then deemed negative upon closer manual inspection.
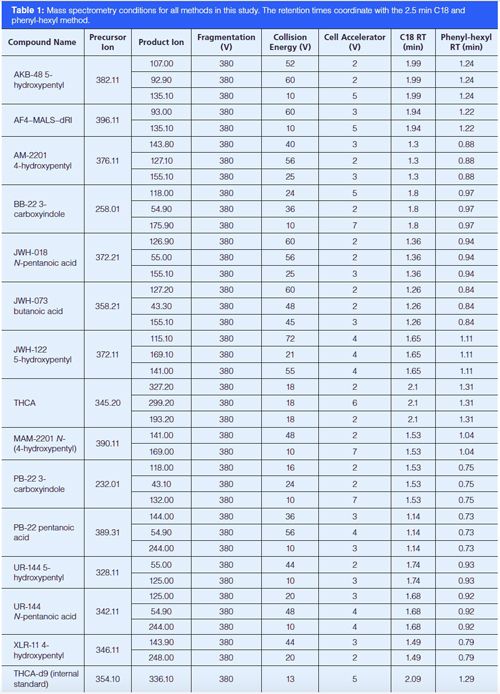
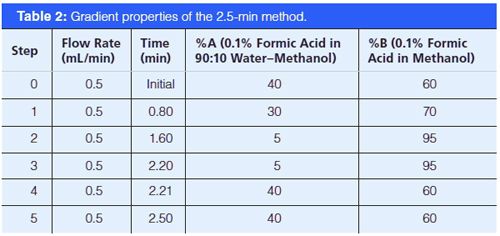
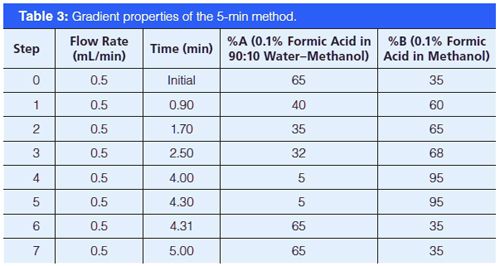
Instrumentation: All analyses were conducted by LC–MS–MS on an Agilent 6490 triple-quadrupole system run in electrospray ionization (ESI) positive mode using an Agilent 1290 chromatographic system (1290 Inifinity binary pump, 1290 TCC, 1290 autosampler, and 1290 thermostat) with a 100 mm × 2.1 mm, 2.7-μm dp Agilent Poroshell 120 EC-C18 or 50 mm × 2.1 mm, 2.6 µm Phenomenex Kinetex PhenylâHexyl column. Source conditions were optimized with a 250 °C gas temperature, gas flow at 19 L/min, nebulizer set to 45 psi, sheath gas heater at 300 °C, sheath gas flow at 11 L/min, capillary voltage at 3.5 kV, and charging voltage at 2 kV. The run time for this method is 2.21 min with a cycle time of approximately 2.5 min. A longer chromatographic method (roughly 5 min) was also used in this study to help resolve questionable interferences. All of these assays monitor two or three transitions for each of the following 14 analytes: AKB48 5-hydroxypentyl metabolite, AKB48 pentanoic acid metabolite, AM2201 4-hydroxypentyl metabolite, BB-22 3-carboxyindole metabolite, JWH 018 pentanoic acid metabolite, JWH 073 butanoic acid metabolite, JWH 122 5-hydroxypentyl metabolite, MAM2201 4-hydroxypentyl metabolite, PB-22 3-carboxyindole metabolite, PB-22 pentanoic acid metabolite, UR-144 5-hydroxypentyl metabolite, UR-144 pentanoic acid metabolite, XLR11 4-hydroxypentyl, and THCA; and one transition for one internal standard, THCA-D9. THCA is analyzed by the mass spectrometer, but it is not actively monitored in patient samples. MS method parameters are shown in Table 1. The chromatographic starting conditions are 40% mobile-phase A (0.1% formic acid in 90:10 water–methanol) and 60% mobile-phase B (0.1% formic acid in methanol) with a 0.5-mL/min flow throughout (Tables 2 and 3). The 2.5-min phenyl-hexyl method was validated according to a previously published procedure (16).
Results and Discussion
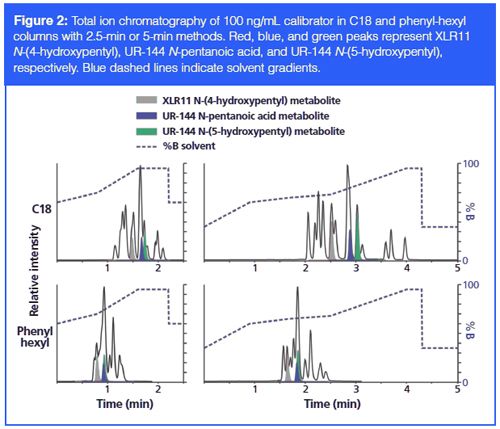
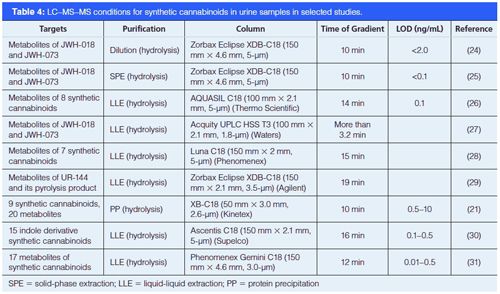
Various methods including colorimetric detections (17), immunochemical assays (18), nuclear magnetic resonance (NMR) (19), gas chromatography–mass spectrometry (GC–MS) (20), and LC–MS–MS (21), have been developed for the analysis of synthetic cannabinoids. With those methods, many synthetic cannabinoids have been successfully analyzed in different samples such as plant materials, human hair, saliva, serum, and urine. Several analytical reviews have summarized the identification and quantification techniques for synthetic cannabinoids that are currently popular (22,23). Among those methods, LC–MS–MS has clear advantages of ease and speed of sample preparation and the capability of automation. However, most of the current methods only focus on a few synthetic cannabinoids, or need a very long chromatographic gradient to affect resolution of spice compounds of interest (usually longer than 10 min, see Table 4). To improve the analysis of synthetic cannabinoids, we developed new LC–MS–MS methods with two different column chemistries (C18 and phenyl-hexyl), which take either 2.5 min or 5 min for each sample to achieve optimal resolution. These methods were applied to the analysis of 13 synthetic cannabinoids.
We have analyzed a 100-ng/mL synthetic cannabinoid calibrator that includes all the K2 and spice compounds of interest to this work with both the 2.5-min or 5-min methods in two different columns. Most of the compounds were eluted in similar order in the different columns, though the elution time changed. Overall, the compounds in the phenyl-hexyl column are eluted earlier compared with ones in the C18 column under both the 2.5âmin and 5-min methods, which may be solely due to the shorter length of the column or a combination of length and selectivity. In addition, the three compounds that share the tetramethylcyclopropyl ketone indole structural moiety (that is, XLR11 N-[4-hydroxypentyl], UR-144 N-pentanoic acid, and UR-144 N-[5-hydroxypentyl]) exhibit changed elution order in the two different columns. In both the 2.5-min and 5-min methods, those three compounds were eluted much earlier in order with the phenylâhexyl column compared to the C18 column. This change in elution order is not because of the change in the column length. However, it might be due to their tetramethylcyclopropyl structure having a higher affinity towards the C18 column than for the phenyl-hexyl column. Although this observation may seem trivial, it helps illustrate the breadth of chemical components inherent in a synthetic cannabinoid method. This challenge of chemical breadth can be used as an advantage, however, if one considers that synthetic cannabinoids with different chemical structures will have different elution behaviours in two distinct column chemistries. In most cases, newly invented spice compounds only slightly change the side chains of the banned chemicals. It is possible that evaluating potential patient positives for this class of compounds using two different column chemistries might help better separate compounds with similar chemical structures, thereby improving the detection of novel compounds from existing agents.
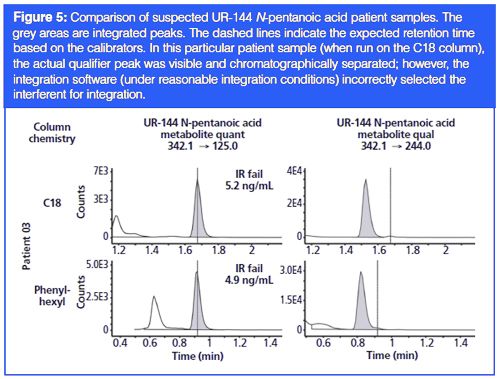
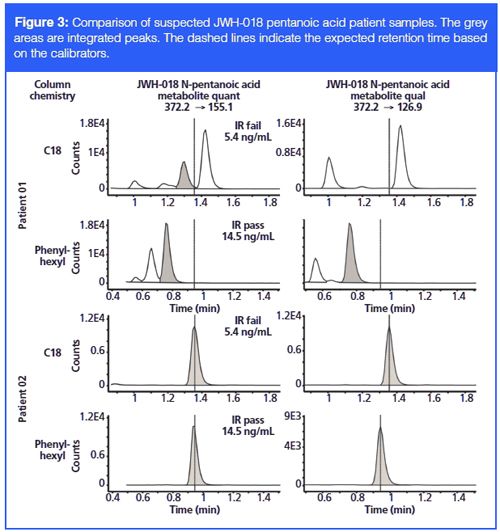
These new methods for analyzing synthetic cannabinoids were applied to suspected patient positive samples identified from a production method. When the urine sample of patient 01, positive for JWH-018 pentanoic acid metabolite, was analyzed using both C18 and phenyl-hexyl columns, both quantifier (quant) and qualifier (qual) peaks for JWH-018 pentanoic acid metabolite came out earlier than expected based on calibrators (Figure 3). However, the ion ratio failed in the analysis on the C18 column because of a missing qual peak, whereas the ion ratio passed in the analysis with the phenyl-hexyl column. Regardless of column chemistry, a human reviewer would likely review this sample as negative or “unable to confirm” since retention times do not perfectly line up. However, with the phenyl-hexyl column data the peaks that passed the ion ratio criteria were not all that far off with regards to retention time. On a production floor it is not unreasonable for peaks to drift 0.3 min (18 s) over a given day or week, especially if this instrument is used to run two different methods that may or may not use different columns and solvents.
Meanwhile, in the test of patient 02, also potentially positive for JWH-018 pentanoic acid, all peaks showed up at the expected retention times. The ion ratios passed on the phenyl-hexyl column, but failed on the C18 column, which is consistent with the result of patient 01. The data suggests the phenyl-hexyl column significantly improved the detection of JWH-018 pentanoic acid metabolite in our methods compared to the C18 column. The fact that this patient sample fails ion ratio (IR) on the C18 column and passes on the phenylâhexyl possibly indicates that an interferent coeluted with one or both of the C18 peaks, thereby throwing off the ion ratio. Cannabinoids (synthetic or otherwise), due to their chemical makeup, are generally fat soluble and by extension they also tend to be chromatographically coeluted with any lipid content that may be in a sample. It is possible that this interferent, which is throwing off the ion ratio in the C18 sample, is a lipid component that was able to survive the hydrolysis and extraction protocol to be coeluted on the C18 column, but on the phenyl-hexyl column it is sufficiently separated. It is also possible that the compound from the patient sample is isobaric with JWHâ018 pentanoic acid and possesses the same multiple reaction monitoring (MRM) transitions as JWH-018, but at different ratios than the true calibrator compound. This is possible if a small change in side chain configuration is envisioned (for example, straight chain versus branched chain). The technical and ethical issues associated with making a positive call on such samples are not trivial.
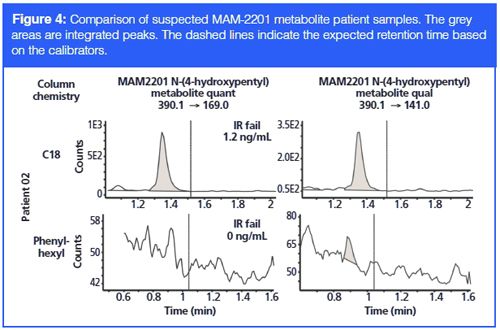
Next, for a suspected MAM-2201 N-(4-hydroxypentyl) metabolite, we found that patient sample 02 showed an interfering peak, with slightly incorrect retention time, on the C18 column. The chemistry of this interferent seems to be drastically different compared to the MAM-2201 N-(4-hydroxypentyl) metabolite, since it was not observed in the window for the phenyl-hexyl column. These types of interferences are rampant among positive and questionably positive synthetic cannabinoid patient samples.
Patient 03 had a very strong well separated quant peak for UR-144 N-pentanoic acid, but the qual peak showed an interferent just a few seconds away from the targeted retention time. This interferent made detection of the qual peak of interest very difficult. The qual peak is still visible in the C18 separation; however, the software (under reasonable integration conditions) incorrectly integrated the interferent. With the phenyl-hexyl column chemistry, the qualifier peak has coalesced into the interferent peak entirely and is not able to be resolved, even with manual integration intervention. The fact that this interferent moved proportionally with reference to the expected UR-144 N-pentanoic acid retention time indicates that this interferent might share some chemical functionality as discussed above.
Conclusions
A rapid, selective, and sensitive LC–MS–MS method identifying 13 synthetic cannabinoids in patient urine samples has been described. Two different column chemistries (that is, C18 and phenyl-hexyl) have been applied using this method. Three compounds, including XLR-11 N-(4-hydroxylpentyl), UR-144 N-pentanoic acid, and UR-144 N-(5-hydroxylpentyl) metabolites, demonstrate the different order of elution on a phenyl-hexyl column compared to the C18 column, while most of the compounds maintain their elution order. The fact that newly invented synthetic cannabinoids often only slightly change the side chains of the banned drugs makes the detection of those compounds more difficult. At our laboratory, synthetic cannabinoids are requested in roughly 20% of our total samples and therefore should not be written off as a fringe interest in the pain medication monitoring arena in spite of the very low positivity rate. Using a second LC–MS–MS method to confirm patient positives (as illustrated here) is potentially useful for large scale laboratories on a daily basis because of the low positivity rates observed.
References
- S.M. Gurney, K.S. Scott, S.L. Kacinko, B.C. Presley, and B.K. Logan, Forensic Sci. Rev.26, 53–78 (2014).
- C.R. Harris and A. Brown, J. Emerg. Med.44, 360–366 (2013).
- L.M. Labay, J.L. Caruso, T.P. Gilson, R.J. Phipps, L.D. Knight, N.P. Lemos, I.M. McIntyre, R. Stoppacher, L.M. Tormos, A.L. Wiens, E. Williams, and B.K. Logan, Forensic Sci. Int.260, 31–39 (2016).
- K.B. Shelimov, D.E. Clemmer, R.R. Hudgins, and M.F. Jarrold, J. Am. Chem. Soc.119, 2240–2248 (1997).
- J.L. Wiley, J.A. Marusich, and J.W. Huffman, Life Sci.97, 55–63 (2014).
- O. Cottencin, B. Rolland, and L. Karila, Curr. Pharm. Des.20, 4106–4111 (2014).
- R. Mechoulam, J.J. Feigenbaum, N. Lander, M. Segal, T.U. Jarbe, A.J. Hiltunen, and P. Consroe, Experientia44, 762–764 (1988).
- A. Weissman, G.M. Milne, and L.S. Melvin, Jr., J. Pharmacol. Exp. Ther.223, 516–523 (1982).
- E.S. Graham, J.C. Ashton, and M. Glass, Front Biosci. (Landmark Ed.) 14, 944–957 (2009).
- D.R. Compton, L.H. Gold, S.J. Ward, R.L. Balster, and B.R. Martin, J. Pharmacol. Exp. Ther.263, 1118–1126 (1992).
- J.W. Huffman, G. Zengin, M.J. Wu, J. Lu, G. Hynd, K. Bushell, A.L. Thompson, S. Bushell, C. Tartal, D.P. Hurst, P.H. Reggio, D.E. Selley, M.P. Cassidy, J.L. Wiley, and B.R. Martin, Bioorg. Med. Chem.13, 89–112 (2005).
- K. Zaitsu, H. Nakayama, M. Yamanaka, K. Hisatsune, K. Taki, T. Asano, T. Kamata, M. Katagai, Y. Hayashi, M. Kusano, H. Tsuchihashi, and A. Ishii, Int. J. Legal Med. 129, 1233–1245 (2015).
- N. Uchiyama, S. Matsuda, M. Kawamura, R. Kikura-Hanajiri, and Y. Goda, Forensic Toxicol.31, 223–240 (2013).
- S.D. Banister, J. Stuart, R.C. Kevin, A. Edington, M. Longworth, S.M. Wilkinson, C. Beinat, A.S. Buchanan, D.E. Hibbs, M. Glass, M. Connor, I.S. McGregor, and M. Kassiou, ACS Chem. Neurosci.6, 1445–1458 (2015).
- H.A. Claessens, M.A. Van Straten, C.A. Cramers, M. Jezierska, and B. Buszewski, J. Chromatogr. A826, 135–156 (1998).
- J.R. Enders and G.L. McIntire, J. Anal. Toxicol.39, 662–667 (2015).
- R.C. Isaacs, Forensic Sci. Int.242, 135–141 (2014).
- A. Arntson, B. Ofsa, D. Lancaster, J.R. Simon, M. McMullin, and B. Logan, J. Anal. Toxicol.37, 284–290 (2013).
- C. Rollins, S. Spuhler, K. Clemens, D. Predecki, and J. Richardson, “#1398 Qualitative and quantitative analysis of fluorine containing synthetic cannabinoids using NMR,” presented at the 245th ACS National Meeting & Exposition, New Orleans, Louisiana, USA, 2013.
- H. Choi, S. Heo, S. Choe, W. Yang, Y. Park, E. Kim, H. Chung, and J. Lee, Anal. Bioanal. Chem.405, 3937–3944 (2013).
- A. Wohlfarth, K.B. Scheidweiler, X. Chen, H.F. Liu, and M.A. Huestis, Anal. Chem.85, 3730–3738 (2013).
- M.A. Elsohly, W. Gul, A.S. Wanas, and M.M. Radwan, Life Sci.97, 78–90 (2014).
- A. Namera, M. Kawamura, A. Nakamoto, T. Saito, and M. Nagao, Forensic Toxicol.33, 175–194 (2015).
- C.L. Moran, V.H. Le, K.C. Chimalakonda, A.L. Smedley, F.D. Lackey, S.N. Owen, P.D. Kennedy, G.W. Endres, F.L. Ciske, J.B. Kramer, A.M. Kornilov, L.D. Bratton, P.J. Dobrowolski, W.D. Wessinger, W.E. Fantegrossi, P.L. Prather, L.P. James, A. RadominskaâPandya, and J.H. Moran, Anal. Chem.83, 4228–4236 (2011).
- K.C. Chimalakonda, C.L. Moran, P.D. Kennedy, G.W. Endres, A. Uzieblo, P.J. Dobrowolski, E.K. Fifer, J. Lapoint, L.S. Nelson, R.S. Hoffman, L.P. James, A. Radominska-Pandya, and J.H. Moran, Anal. Chem.83, 6381–6388 (2011).
- A.D. de Jager, J.V. Warner, M. Henman, W. Ferguson, and A. Hall, J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.897, 22–31 (2012).
- E.G. Yanes and D.P. Lovett, J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.909, 42–50 (2012).
- M. Hutter, S. Broecker, S. Kneisel, and V. Auwarter, J. Mass Spectrom.47, 54–65 (2012).
- A. Grigoryev, P. Kavanagh, A. Melnik, S. Savchuk, and A. Simonov, J. Anal. Toxicol.37, 265–276 (2013).
- M. Mazzarino, X. de la Torre, and F. Botre, Anal. Chim. Acta837, 70–82 (2014).
- J.L. Knittel, J.M. Holler, J.D. Chmiel, S.P. Vorce, J. Magluilo, Jr., B. Levine, G. Ramos, and T.Z. Bosy, J. Anal. Toxicol.40(3), 173–86 (2016).
Sheng Feng, Brandi Bridgewater, Gregory L. McIntire, and Jeffrey R. Enders are with Ameritox Ltd., in Greensboro, North Carolina, USA. Direct correspondence to: Jeffrey.Enders@ameritox.com


.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)






