Semivolatile Analysis Using an Inertness-Performance-Tested Agilent J&W DB-5ms Ultra Inert Column
The Application Notebook
Semivolatile analyses using methods similar to US EPA method 8270 (1) are important in environmental laboratories worldwide. A number of acidic compounds such as benzoic acid or 2,4-dinitrophenol and strong bases such as pyridine or benzidine are active species found in the semivolatile sample set. These highly polar species are particularly susceptible to adsorption into active surfaces in the sample flow path, including the column itself. System and column inertness are critical for effective analysis of these active chemical species.
Kenneth Lynam, Agilent Technologies, Inc.
Semivolatile analyses using methods similar to US EPA method 8270 (1) are important in environmental laboratories worldwide. A number of acidic compounds such as benzoic acid or 2,4-dinitrophenol and strong bases such as pyridine or benzidine are active species found in the semivolatile sample set. These highly polar species are particularly susceptible to adsorption into active surfaces in the sample flow path, including the column itself. System and column inertness are critical for effective analysis of these active chemical species.
Semivolatile Challenging Analytes
The evaluation of column performance began with the initial testing of the column, using the Über One probe mix (see "Agilent J&W Ultra Inert GC Columns: A New Tool to Battle Challenging Active Analytes" in this issue.) Next, we looked at an abbreviated list of compounds specific to the US EPA Method 8270 sample set, as noted in Figure 1. These species were selected to range in polarity from basic to acidic species and from very early eluting nitrosamine to late eluting polynuclear aromatic hydrocarbons (PAHs). Figure 1 is a total ion chromatogram of the challenging analyte mix with a 5 ng on-column loading of each component.
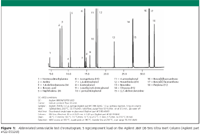
Figure 1
One key assessment criterion for US EPA 8270 system suitability is the response factor for 2,4-dinitrophenol and its most closely eluting internal standard acenaphthene-D10. The minimum acceptable average response factor (over the entire concentration range) is 0.050 and the typical range is between 0.1 and 0.2. This response tends to decrease at lower concentrations and as the chromatographic system or the standard starts to deteriorate. In Figure 1, response factors for 2,4-dinitrophenol are greater than 0.1 and for 4-nitrophenol, they are greater than 0.2, each at a concentration of 5 μg/mL. These values are indicative of excellent column performance even at low standard concentration.
The recovery of benzidine is another key indicator of inertness performance for semivolatile analysis. This particular base is subject to thermal breakdown in the inlet and to oxidation from standing in solutions. Injection temperatures above 260°C cause the recovery of benzidine to drop dramatically. It is necessary to balance the recovery of benzidine with the elution of heavier PAHs when setting injection port temperatures. An injection port temperature setting of 260°C gives good recoveries for benzidine and is still hot enough for higher molecular weight PAHs to volatilize.
Semivolatile Large Mix
Figure 2 shows a 5-ng on-column loading of a broader range of semivolatile analytes. This large mixture was prepared by combing AccuStandard semivolatile mixes 1, 2, 3, 4(a), 4(b), 5 and 6 all at nominal concentration of 5 μg/mL. In total, 93 semivolatile compounds are included in this mix, ranging from the very low-boiling N-nitrosodimethylamine to the high-boiling benzo[g,h,i]perylene. In addition, a wide diversity of analyte polarities is represented. The highlighted area in Figure 2 shows the elution and peak shape of highly basic benzidine and its response relative to the nearest eluting peak, flouranthene. Even in this large mix, benzidine gives good relative response and peak shape.
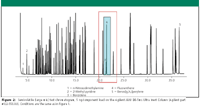
Figure 2
Conclusions
Rigorous column inertness testing with aggressive probes ensures consistent and reliable column inertness performance for active analytes.
Columns with well-defined inertness baselines provide a reliable platform for the analyst to being analysis of semivolatiles. The DB-5ms Ultra Inert column used in this series of experiments demonstrates excellent inertness performance for some of the most difficult analytes in the semivolatile sample set. The good recoveries and peak shapes observed for these difficult species, even with 5 ng on-column loading, are indicative of successful semivolatile analyses on these new DB-5ms Ultra Inert columns.
References
1. US EPA Method 8270D, Revision 4, February (2007), "Semivolatile Organic Compounds by Gas Chromatography/Mass Spectrometery (GC/MS)"
2. Mitch Hastings, Allen K. Vickers and Cameron George, "Inertness Comparison of Sample of 5% Phenyldimethylpolysiloxane Columns," Poster Presentation, 54th Annual Pittsburg Conference, Orlando, Florida, USA, March (2003).
3. Jim Luong, Ronda Gras, and Walter Jennings, J. Sep. Sci. , 30, 2480–2492 (2007).
4. Mike Szelewski and Bill Wilson, "Improvements in the Agilent 680-5973 GC/MSD System for Use with USEPA Method 8270," Agilent Technologies publication 5988-3072EN

Agilent Technologies Inc.
2850 Centerville Road, Wilmington, DE 19808
tel. +1 800-227-9770 fax +1 302-633-8901
Email: chem@agilent.com

Separation of Ultra-Short and Long Chain PFAS Compounds Using a Positive Charge Surface Column
December 11th 2024A separation of ultra-short and long chain PFAS (C1-C18) is performed on a HALO®PCS Phenyl-Hexyl column along with a HALO®PFAS Delay column which demonstrates excellent retention for both hydrophilic and hydrophobic analytes.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)














