Selectivity: Are You Making the Most of It?
Do you understand the benefits of selectivity as a method development opportunity?
Photo Credit: Login/Shuttestock.com

Do you understand the benefits of selectivity as a method development opportunity?
I’m sure you are all aware of the relationship shown in Figure 1, which explains the relationship between chromatographic resolution and the other terms within the Purnell equation.
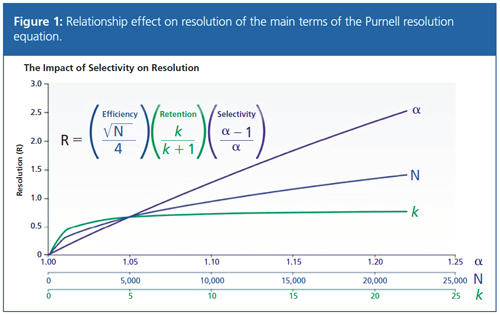
Essentially, Figure 1 shows the return on investment for efforts to optimize resolution in a separation using efficiency (N), retention factor (k), and selectivity (α), and the usual teaching point is that selectivity is a highly effective way to achieve the desired resolution.
Most of us will know that the selectivity of a separation is dictated by the nature of the chemical and physico-chemical interactions between analytes, mobile phase (in certain cases), and the stationary phase and may be controlled by altering the chemistry of the eluent or stationary phase as well as, in condensed phase separations, the degree of ionization of the eluent.
Contrast this then with the modern zeitgeist where, for more than a decade, we have hurtled headlong into the concept of high-efficiency chromatographic separations. Fast gas chromatography (GC) and ultrahigh-pressure liquid chromatography (UHPLC) have become bywords for innovation, time saving, and for separations where high peak capacity is held up as the metric of choice to judge the worthiness of a new product.
I’m not saying that high efficiency isn’t a very worthwhile goal in any chromatographic separation, but as can be inferred from the Purnell equation, it is somewhat limited in terms of diminishing returns, which anyone who has ever worked with impedance or Poppe plots will realize.
My point here is that perhaps we have somewhat defocused on selectivity as a primary variable in chromatography and for sample preparation. Whilst high efficiency may make our lives easier, it cannot and will not ever improve a separation where the selectivity of the system is unable to chemically distinguish between improperly separated analytes.
Now I can picture those with mass spectrometric detectors smiling smugly as they think about how they can resolve these analytes “spectrometrically” and that therefore the concept of good sample preparation or chromatographic selectivity is outmoded. The space afforded me by the publishers for this column is not long enough for me to explain the absolute folly of this attitude and the numbers of ways in which one can come unstuck by relying on resolution afforded in either time or space within the mass spectrometer. For those of you who remain to be convinced of this folly, for those thinking “Well it works perfectly well for my analysis”, time alone will deliver you an analysis that does not perform when using this approach. One day an unseen, unresolved impurity, a matrix effect, or a nonlinear calibration assumed to be linear will catch up with you. Be warned!
I’ll assume that I have piqued your interest and that you are willing to look with fresh eyes on selectivity as a method development or optimization opportunity.
Perhaps the trouble with selectivity is that it dictates that one understands something of the chemistry of analytes and matrix. It’s such an effort to change column or make up a fresh batch of eluent. It’s not so convenient or easy to understand and therefore perhaps less appealing, but in my humble experience, the ability to truly exploit this variable is the difference between a chromatographer and a user of chromatography.
Perhaps the issue is that we have lost sight of the usefulness of this highly important chromatographic variable. I’ve therefore selected a few examples to remind us of the ways in which selectivity can be exploited to very good effect in our work.
On my last count, there are over 300 “C18” phase variants available to purchase from a variety of different manufacturers. Whilst we consider “C18” as a single category of bonded phase, the various types of silica used, bonding chemistry, deactivation chemistry, and other surface treatments means that the variation in selectivity of this single-phase designation can be huge. So, if you’ve tried a C18 column and it hasn’t produced the separation you need, rather than turning to a different bonded phase ligand (cyano, C8, PFP) why not try a different C18 variant instead? I know of several companies that include three or even four C18 columns-all with different selectivity characteristics-within their column screening toolkit. Sticking with a favourite manufacturer can also limit the selectivity landscape that one might explore and I have personal experience of situations in which moving from a C18 to a different bonded phase chemistry from the same manufacturer has resulted in less change in selectivity than moving between C18 columns from different manufacturers because of the changes in base silica and bonding chemistry. Be open to exploring the whole selectivity landscape available from different manufacturers.
Hydrophilic interaction liquid chromatography (HILIC) is an obvious choice to explore a very different selectivity in high performance liquid chromatography (HPLC), but I wonder how many of us truly exploit the huge selectivity differences available by using an ionizable HILIC phase in a meaningful way. Bare silica and amide ligands can be ionized using variations in mobile phase pH, which presents us with a unique combination of electrostatic and partitioning behaviour that can be exploited to really fine-tune difficult separations. Of course, this dictates that we know something of the chemistry of the stationary phase and the analyte and takes more effort to truly understand the separation, and as I highlighted above, often requires deeper thinking about a separation.
In both reversed phase and HILIC modes in HPLC we have mixed mode and zwitterionic phases, which intentionally have ionizable groups built into the bonded phase ligands and again offer the opportunity to tune the selectivity of a separation using the eluent pH or counter ion strength and concentration. I often meet resistance to the use of these types of phase as they are “too complex” or “non-reproducible”, but I must say that these statements typically come from those folks who are less experienced or unable to get to grips with the mechanisms of interaction and who lose control of the important variables that control the selectivity such as eluent pH, buffer choice, and strength.
Sticking with the theme of condensed phase separations, many laboratories I visit use acetonitrile as a first intent solvent. This is fine and there are many compelling reasons why this should be the case, but I see few laboratories changing to methanol to explore the different selectivity that this solvent can deliver before switching to an alternative stationary phase. Could this be related to the amount of effort required to prepare a different mobile phase?
Modern capillary gas chromatography is a technique truly driven by efficiency, yet I can cite many examples of folks struggling to optimize a temperature programme when the only real option to exploit the chemistry differences between analytes and obtain a suitable separation is to choose one of the many different stationary phases available. Having some basic knowledge of the interaction of the five main categories of stationary phase chemistries and your analytes can save a huge amount of time in GC method development. Whilst I fully appreciate that a very large number of separations can be performed using a 5% phenyl polydimethylsiloxane phase, there are times when changing the nature of the chemical interaction between analytes
and stationary phase (that is, changing the selectivity) can produce excellent results and will often lead to faster separations.
In sample preparation, selectivity is perhaps even more important. In a simple sample preparation protocol such as liquid–liquid extraction (LLE), which of us would consider using an extraction solvent of different polarity, altering the pH of the aqueous phase, or using a high salt concentration to alter the selectivity of the extraction? All of these variables can be used to improve the extraction efficiency by exploiting the selectivity of the method.
In solid-phase extraction, selectivity is the key to achieving a cleaner extract, which ultimately may affect the sensitivity of the method and the time required to develop and optimize the post-extraction chromatographic separation. Sorbents such as C18, which use hydrophobic interactions, are the least selective of all SPE sorbents. Those sorbents containing ionizable ligands where protocol solvent pH and counter ion strength and concentration can all be used to dictate the ionic interaction between the analyte and the sorbent are often the most selective. Again one needs to understand the chemistry of the analyte–sorbent interaction and be aware of the kinetics of the point to point nature of the electrostatic interaction, taking care with sample flow rates, using soak steps and titrating pH or ionic strength to ensure optimum recovery and cleanliness.
I could cite many more examples, but I’m sure you have got the point by now. The one common factor that links all the selectivity optimization examples above? One needs an understanding of chromatography and the basic chemistry of stationary phases, mobile phases, and analytes to truly finesse a separation or obtain a really clean sample from a preparative method. The question you might ask yourself is: “Do I have a deep enough understanding to exploit the most important chromatographic variable of them all?” Only you can decide.
Contact author: Incognito
E-mail:admin@chromatographyonline.com

A Final Word from Incognito—The Past, Present, and Future of Chromatography
February 10th 2022After 14 years in print, Incognito’s last article takes a look at what has changed over a career in chromatography, but it predominantly focuses on what the future might hold in terms of theory, technology, and working practices.
Sweating the Small Stuff—Are You Sure You Are Using Your Pipette Properly?
October 7th 2021Most analytical chemists believe their pipetting technique is infallible, but few of us are actually following all of the recommendations within the relevant guidance. Incognito investigates good pipetting practice and busts some of the urban myths behind what is probably the most widely used analytical tool.







