Quantitative Determination of Four Lignans in Phyllanthus niruri L. by HPLC
A simple analytical method using high performance liquid chromatography (HPLC) with ultraviolet (UV) detection was developed for the simultaneous determination of four main lignans (niranthin, nirtetralin B, hypophyllanthin, and phyltetralin) in Phyllanthus niruri L. plant samples from Guangxi province, China. The aim was to validate the accumulation rule in the growth periodicity of the active lignans for anti-hepatitis B virus (HBV). The results indicated that the active lignans niranthin and nirtetralin B were mainly distributed in the leaves of Phyllantthus niruri L., and the highest total content was found in the end of July to August (full fruiting period). Therefore, the Phyllanthus niruri L. of Guangxi province (China) should be collected for antihepatitis B treatment on the full fruiting period (the end of July to August), and its leaves might be the main part for collection. The results of the study provided a strong support for the use of Phyllanthus niruri L. from Guangxi province, China.
Most traditional Chinese medicines come from plants, and their chemical components are the secondary metabolites of these plants, such as artemisinin, a medium anti-malaria drug, and paclitaxel, an anti-cancer drug (1–3). The study of the change process of the secondary metabolites in plant growth periodicity could help to know the accumulation rules of medicinal components. Furthermore, it also benefits for the full utilization of the medicinal plant resources (4,5).
Phyllanthus niruri L. (Euphorbiaceae), widely distributed in the south of China, is known as a traditional Chinese herb used in fever, colic, diarrhea, liver protection, and antihepatitis B (6,7). Previously, on the basis of its reputation in liver protection and antihepatitis B, we initiated a series of studies to investigate the bioactive constituents of the genus Phyllanthus niruri L. from Guangxi province, China. A series of lignans were discovered, in which some were shown to possess significant anti-hepatitis B virus (HBV) activity (8–10). Lignans niranthin, nirtetralin, nirtetralin A, and nirtetralin B showed significant anti-HBV activity, while lignans hypophyllanthin and phyllanthin with the similar structure showed little activity. These lignans were secondary metabolites of Phyllanthus niruri L., and the similar skeleton structure suggested a similar biosynthesis pathway in the plant. It was hypothesized that, in some period of the Phyllanthus niruri L growth cycle, the active lignans are produced in greater quantity than the inactive lignans, so the research on the bioavailability of the active lignans in the growth cycle of the plant is of great significance.
Similar studies were reported (11,12), which focus on developing and improving the analytical method of the lignans in Phyllanthus niruri L. from Malaysia. There were no related reports about the new lignans we isolated from the herb from Guangxi province, China. The regional disparity of the herb may result in different structure of active constituents, as well as the accumulation rule of the active lignans in the growth cycle of the plant. Thus, a study on accumulation rule of the active lignans of this herb from Guangxi province was also necessary for providing a support for making full use of the herb from this region.
The focus of this study was to validate the accumulation rule in the growth periodicity of the main lignans of Phyllanthus niruri L. The lignans have ultraviolet (UV) absorption in the UV terminal, so in this study, the HPLC-UV method was developed for the analysis of the lignans, which were representatives of root, stem and leaves.
Materials and Methods
Standards, Samples and Chemicals
Four lignans, as shown in Figure 1a, niranthin [1], nirtetralin B [2], hypophyllanthin [3], and phyltetralin [4], isolated from Phyllanthus niruri L. according to the protocol established described previously (8–10), were used as standards for quantification of the plant samples. Briefly, the powder of air-dried plant of Phyllantthus niruri L. (100 kg) was extracted with 80% ethanol (3 × 300 L) for 2 d. The pooled extractions yielded 10.2 kg crude residue after evaporated under vacuum. This residue was dispersed in water and extracted with petroleum ether to yield 3.1 kg petroleum ether extract residue. This residue, dissolved in ethanol and water, was introduced to the ethanol solution in a ratio of 3:2 (ethanol:water, V/V). After precipitant was removed, the aqueous ethanol solution was evaporated to provide a residue (450 g). This residue (450 g) was subjected chromatography on a silica gel column (200–300 mesh, 10 × 100 cm, 10000 g) with a gradient eluated increase of ethyl acetate in petroleum ether (0, 4, 10, 20, 30, 40, and 50 %, each 50–100 L and offered fraction 1 (105 g), fraction 2 (95 g), fraction 3 (85 g), fraction 4 (50 g), fraction 5 (45 g), and fraction 6 (30 g). The six fractions were merged according to TLC, and yielded fraction A1, A2, and A3. Fraction A1 was subjected chromatography on a silica gel column with ethyl acetate-petroleum ether 2:1(v:v) to afford 1 (13.6 g). Fraction A2 was subjected chromatography on a silica gel column with ethyl acetate-petroleum ether 2:1(v:v) to afford 2 (6.8 g). Compound 3 (25.6 g) and 4 (25.6 g) were yielded from fraction A3. The purity of the compounds was confirmed to be more than 95% by a Thermo Fisher UItiMate 3000 HPLC system. Their structures were confirmed by comparison of the NMR and MS spectra reported previously (8–10).
FIGURE 1: (a) Chemical structures of lignans isolated from Phyllanthusniruri L., and (b) their HPLC spectrum.

Samples for analysis comprising the root, stem, and leaves of Phyllanthus niruri L. were collected from Guangxi province, China, and identified by Dr Shifeng Ni, Northwest University, Xi'an, China. A voucher specimen was deposited in the Department of Chemistry at Guangxi University. The HPLC grade acetonitrile was purchased from Merck. Deionized water for HPLC was redistilled water.
Instrumentation and Optimized Analytic Procedure
The HPLC-UV system was Thermo Fisher UItiMate 3000 equipped with a UV detector. A Thermo Hypersil Gold C18 column (2.1 × 250 mm, 5 µm) was used with the following analytical conditions: a mobile phase of acetonitrile-water (55:45, v:v) with a flow rate of 1 mL/min at a column temperature of 30 oC. UV detection was at 230 nm, and the sample injection volume was 10 µL.
Preparation of Standard Solutions
The four reference standards were weighed accurately and dissolved in methanol in a volumetric flask to form a stock solution of standards; the concentration of each compound was: [1] niranthin, 11.810 mg/mL; [2] nirtetralin B, 7.850 mg/mL; [3] hypophyllanthin, 5.820 mg/mL; and [4] phyllanthin, 21.36 mg/mL. Working standard solutions were prepared from the stock solution by further dilution with the appropriate volume of methanol. Working solutions of 1 was prepared at the following concentrations: 16.2, 48.6, 145.8, 437.4, 1312.2, 3936.7, and 11810.0 µg/mL, of 2 at 10.8, 32.3, 96.9, 290.7, 872.2, 2616.7 and 7850.0 µg/mL, of 3 at 8.0, 23.9, 71.9, 215.6, 646.7, 1940.0 and 5820.0 µg/mL, and of 4 at 1.4, 6.8, 34.2, 170.9, 854.4, 4272.0, and 21360.0 µg/mL to cover the range of sample concentrations. All the solutions were stored in a refrigerator at 4 °C prior to analysis.
Preparation of Phyllanthusniruri L. Samples for Analysis
The quantification of lignans in Phyllantthus niruri L. plant was carried out in two parts. The first part involved the quantification of the lignans in the petroleum ether extracts from the whole plant. The second part involved the quantification of the lignans in the petroleum ether extracts from the different parts (root, stem, and leaves) of the plant.
After collection, the samples were dried at room temperature and then pulverized by mechanical comminution to fine particles. Samples of 2.0 g each were accurately weighed and put into a 50-mL conical flask, and then extracted with 3 × 20 mL petroleum ether in an ultrasonic bath (ultrasonication frequency 40 kHz) for 30 min. The combined extracts were evaporated to dryness under a nitrogen flow. The residue was reconstituted in 5 mL of methanol. The sample solution was filtered through a 0.45 μm filter prior to analysis.
Method Validation
The analytical parameters of selectivity, linearity, limits of detection (LODs) and quantification (LOQs), accuracy, precision, expressed as repeatability, and stability were evaluated. The established procedures were validated following SANTE/11813/2017 validation criteria for quantitative analytical methods.
Results and Discussion
Development of the Chromatographic Method
The HPLC-UV conditions were investigated in this study, and different mobile phase compositions (water-methanol and water-acetonitrile), column temperatures (25, 30, 35 oC), injection volume (5, 10, 20 µL), and water:acetonitrile ratios (50:50, 55:45, 45:55, 40:60, v:v) were tested and compared. Compared to water-methanol as mobile phase, water-acetonitrile achieved a better shape of the peaks and separation of the four lignans. Retention time was too large in the condition of water-acetonitrile (50:50, v:v) whereas the four lignans could not be separate completely when the water-acetonitrile ratio was 45:55 or 40:60 (v:v). Water-acetonitrile (55:45, v:v) was finally used as mobile phase, because it effectively improved the shape of the peaks and a better separation of the four lignans was achieved. Thus, the optimal chromatographic conditions were as follows: column temperature, 30 oC; flow rate, 1 mL/min; mobile phase, acetonitrile-water (55: 45, v: v); injection volume, 10 µL; and the UV detection wavelength was 230 nm. Thus, well-resolved peaks of the four major lignans from Phyllantthus niruri L .(niranthin [1], nirtetralin B [2], hypophyllanthin [3], and phyllanthin [4]) were achieved with a total run time of 60 min (Figure 1b). The corresponding retention time of standard niranthin [1], nirtetralin B [2], hypophyllanthin [3, and phyllanthin [4] were 50.308 min, 45.332 min, 35.805 min, and 33.553 min, respectively, indicating long retention times of >30).
Validation of the HPLC method
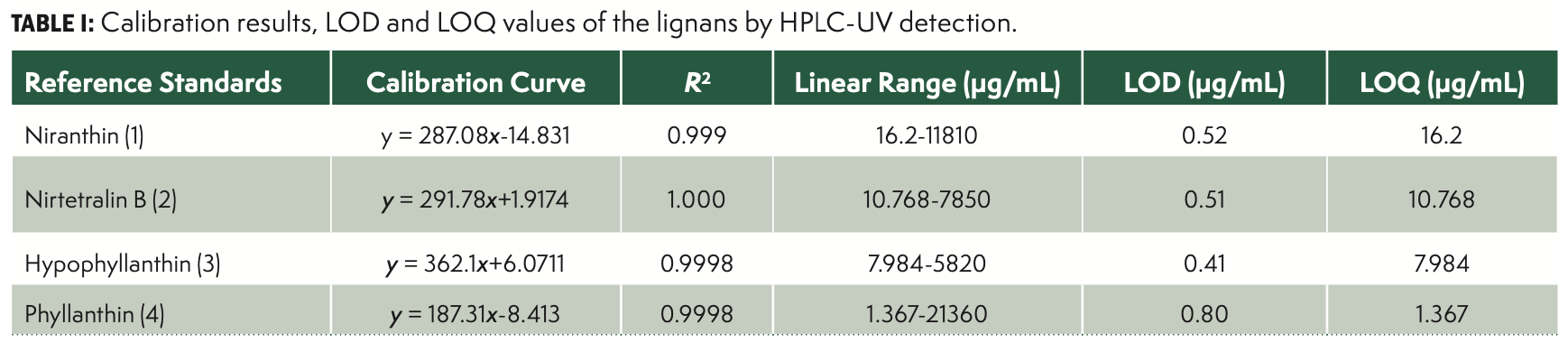
Calibration Curves, Limits of Detection, and Quantification
The reference compounds were dissolved in methanol and stored in a refrigerator at 4 °C. Seven levels of standard solutions of the four lignans were used, in the high and low working ranges. For the HPLC-UV detection method, the data provided a linear function for all reference lignans following the equation:
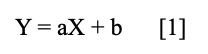
with Y being the value of the peak area, and X being the value of the amount of the reference lignans injected into HPLC. The signal/noise ratios of 3:1 and 10:1 were used to establish LOD and LOQ of the four lignans under the present chromatographic conditions. The calibration results, LOD, and LOQ values of the lignans were shown in Table I. The equations were obtained using the least squares method and showed excellent coefficients of determination (R2 ≥ 0.99), indicating good adherence to the linear model for all the lignans studied.
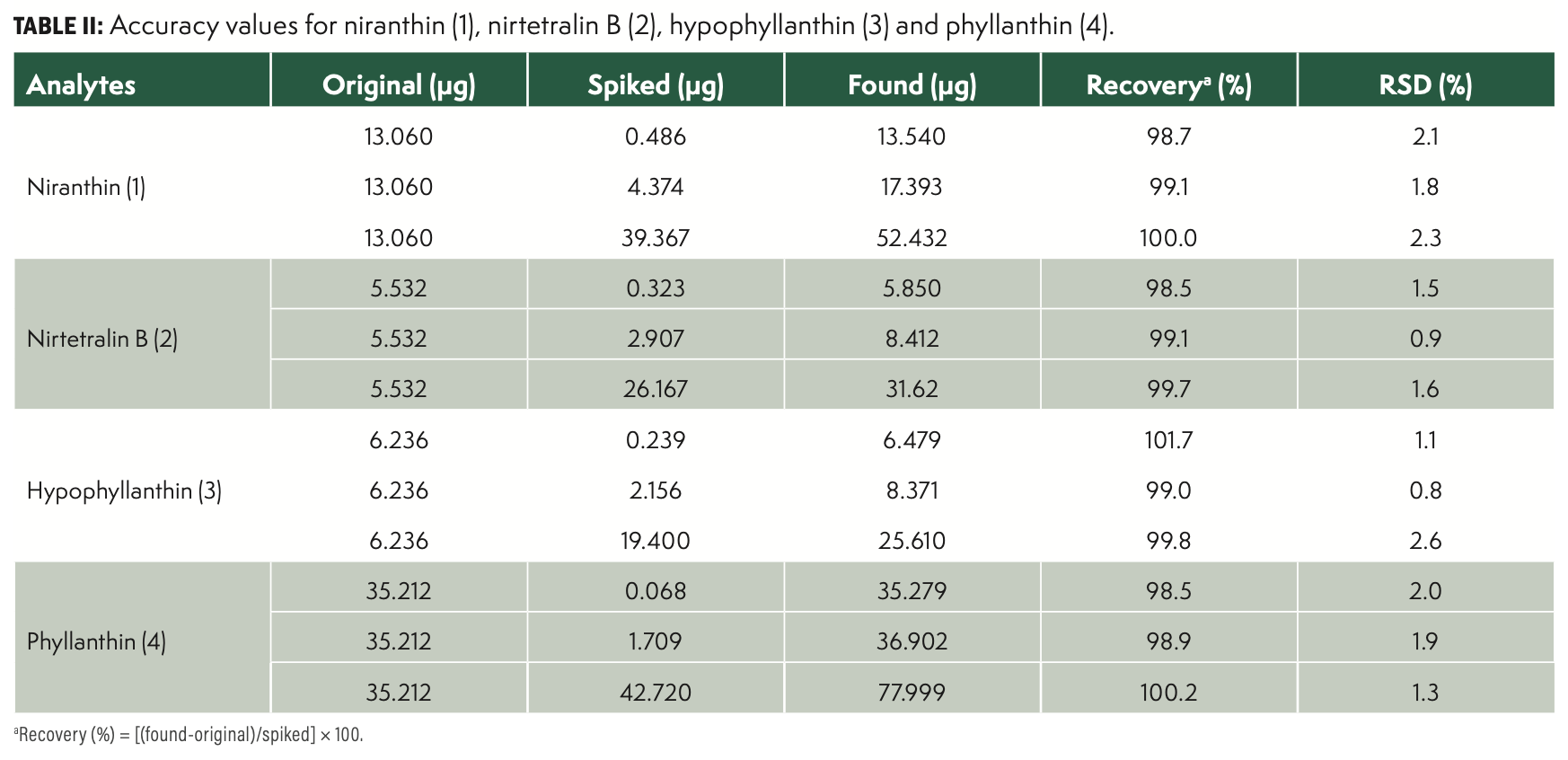
Accuracy
Accuracy was assessed through recovery experiments. An appropriate amount of reference standards was weighed and spiked with a known amount of each reference compound, and then prepared and analyzed in accordance with the methods mentioned earlier. The experiments were performed at three concentrations to cover two values belonging to each of the linear ranges applied in the determination of the lignans. The mean recovery values were calculated for five replicates and are listed in Table II.
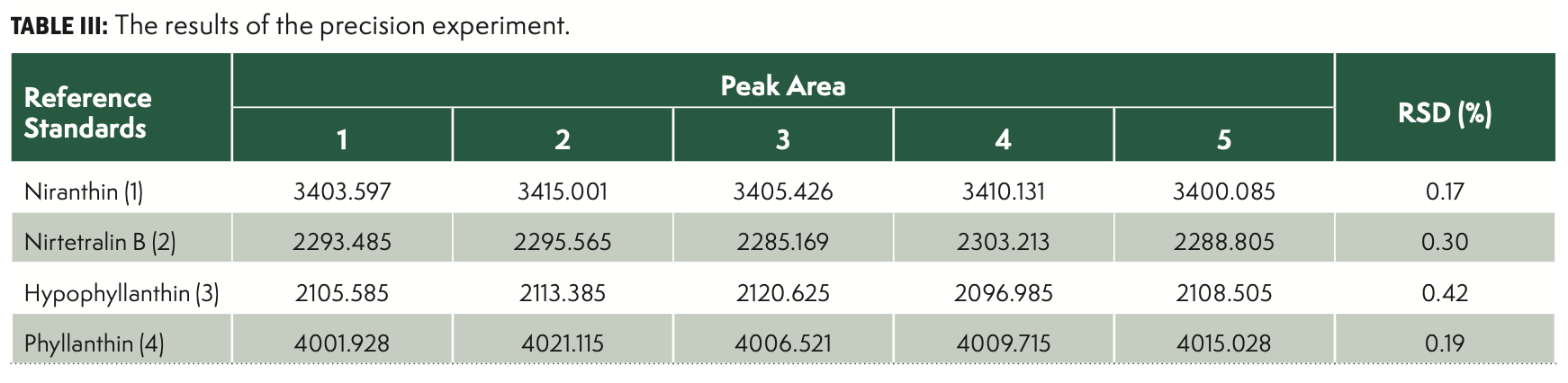
Precision
The precision of the method was evaluated with standard solution by within-laboratory repeatability under the selected optimal conditions five times. The repeatability experiments were executed on the same day by the same analyst. The relative standard deviation (RSD [%]) was taken as a measure of precision. As shown in Table III, the RSD values were less than 4.5% in all cases, which comply with the established by the European Commission (EC, 2002) and the criteria of the AOAC International (2016).
Stability
The stability of the method was evaluated with a sample solution of Phyllantthus niruri L. on a specified period (No. 1) under the selected optimal conditions. Then, 10 µL of the sample was injected and analyzed at 0, 2, 4, 6, 8, and 10 h. The value of the peak area of niranthin [1], nirtetralin B [2], hypophyllanthin [3], and phyllanthin [4] were recorded with the RSDs were all less than 4%, indicating that the sample solution was stable within 10 h. The results were shown in Table IV.

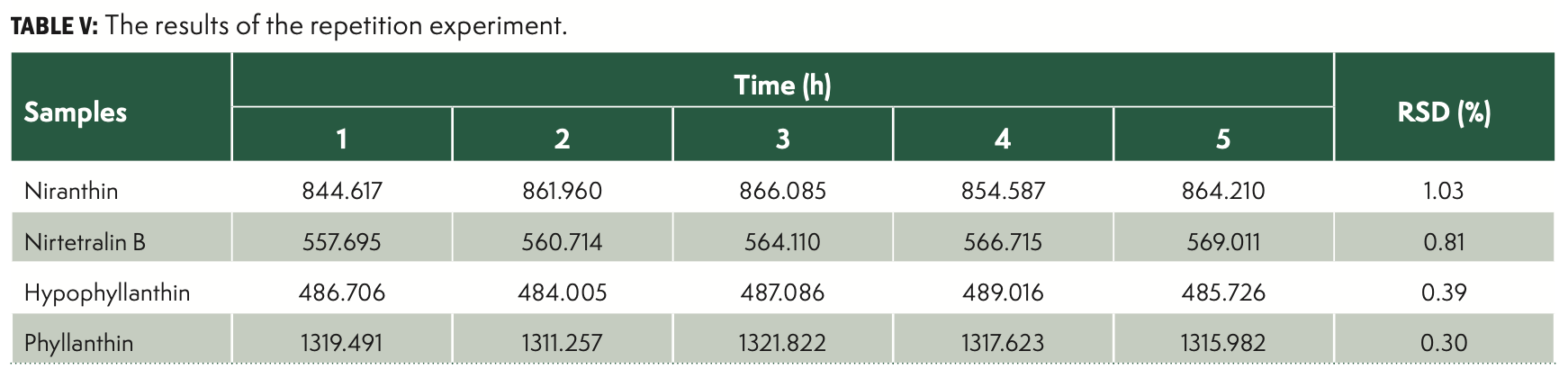
Repeatability
To further evaluate the repeatability of the assay, the leaves of Phyllantthus niruri L. were analyzed in five replicates as described earlier. The RSDs were all less than 2%, indicating that the method has a good repeatability. The results were shown in Table V.
Sample Analysis
The established HPLC-UV method was applied to the simultaneous determination of the four main lignans niranthin [1], nirtetralin B [2], hypophyllanthin [3], and phyllanthin [4] in the whole plant and the different parts (root, stem, and leaves) of Phyllantthus niruri L. of Guangxi province, China. The sample solution of different growth period of Phyllanthus niruri L. and its root, stem, and leaves were analyzed in three replicates under the selected optimal conditions described earlier.
Samples for analysis were collected for the first time on June 20, with an interval of 10 d for further collection. The concentration changes of lignans niranthin [1], nirtetralin B [2], hypophyllanthin [3], and phyllanthin [4] of Phyllantthus niruri L. during its growth period were shown in Figure 2. The lignans profile from the different parts of the plant can also be differentiated. The major lignans were found in all parts of the plant, while the highest total content for lignans 1–4 was found in the leaves throughout the whole growth cycle of the plant. And the lowest total content for lignans 1–4 was found in the root. For the concentration changes of niranthin [1] in Phyllantthus niruri L. during the whole growth period, the concentration of niranthin [1] increased from June to the end of July, namely, the seedling stage, flowering stage, and early fruiting period. The highest total content for niranthin [1] was found in the full fruiting period, followed with the decreasing trend in August until withered. The variation trend of niranthin [1] in the leaves was in keeping with that in the whole plant, whereas the variation trends of niranthin [1] in the root and stem were observed to show some difference. In the root and stem, the concentration of niranthin [1] decreased from seedling period to flowering period, then increased from early fruiting period to full fruiting period. The highest total content was also found in the end of July, then decreased after August. The variation trends of nirtetralin B [2] and phyllanthin [4] were like niranthin [1]. However, the concentration of hypophyllanthin [3] was increased from seedling period to the end of November (maturation period), and the highest total content was also found in the end of November. The variation trend of hypophyllanthin [3] in the leaves was in keeping with that in the whole plant, whereas the highest total content in the root and stem were found in the end of August (full fruiting period).
FIGURE 2: Concentrations of [1] Niranthin, [2] Nirtetralin B, [3] Hypophyllanthin, and [4] Phyllanthin in P. niruri L. samples collected from different times.

Conclusion
The lignans from Phyllantthus niruri L. was proved to be the active constituent for its anti-HBV activity. Among the four main lignans niranthin, nirtetralin B, hypophyllanthin, and phyllanthin of Phyllanthus niruri L. from Guangxi province, China, lignans niranthin and nirtetralin B were proved to show significant anti-HBV activity, while lignans hypophyllanthin and phyllanthin showed inactivity for HBV. Our study was focus on the accumulation rule of the four main lignans in the whole growth cycle of the Phyllantthus niruri L. The results indicated that the active lignans niranthin and nirtetralin B were mainly distributed in the leaves of Phyllantthus niruri L., and the highest total content was found in the end of July to August (full fruiting period). Therefore, the Phyllanthus niruri L. of Guangxi province (China) should be collected for antihepatitis B treatment on the full fruiting period (the end of July to August), and its leaves might be the main part for collection. The results of the study provided a strong support for the use of the traditional Chinese herb Phyllanthus niruri L. from Guangxi province, China.
Acknowledgments
This work was financially supported by the Research Start-up Fund for Doctor in Yantai University (YX20B03), the Science and Technology Support Program for Youth Innovation in Universities of Shandong (2020KJM003).
Declaration of Competing Interest
The authors declare no competing financial interests.
References
(1) Mami, Y.; Jun-ichiro, N.; Mutsuki, Y.; et al. Metabolomics and Differential Gene Expression in Anthocyanin Chemo-Varietal Forms of Perilla frutescens. Phytochemistry 2003, 62, 987–995.
(2) Oliver, F. Metabolic Networks of Cucurbita maxima Phloem. Phytochemistry 2003, 62, 875–886.
(3) Erika, B.; Marco, B.; Camilla, B.; et al. Characterization and Quantification of Thiol-Peptides in Arabidopsis thaliana Using Combined Dilution and High Sensitivity HPLC-ESI-MS-MS. Phytochemistry 2019, 164, 215–222.
(4) Sun, Y.C.; Li, B.M.; Lin, X.T.; et al. Simultaneous Determination of Four Triterpenoid Saponins in Aralia elata Leaves by HPLC-ELSD Combined with Hierarchical Clustering Analysis. Phytochem. Anal. 2017, 28, 202–209.
(5) Willbrord, M.K.; Kenn, F.; Luc, P.; Sheila, M. HPLC-SPE-NMR Analysis of Furanosesquiterpenoids from Bark Exudates of Commiphora swynnertonii Burrt. Phytochem. Lett. 2020, 38, 128–132.
(6) Perry, L.M; Metzger, J. Medicinal Plants of the East Southeast Asia: Attributed Properties and Uses; MIT Press, Cambridge, 1980, pp. 150.
(7) Kaur, N.; Kaur, B.; Sirhindi, G. Phytochemistry and Pharmacology of Phyllanthus niruri L.: A Review. Phytother. Res. 2017, 31 (7), 980–1004.
(8) Wei, W.X.; Li, X.R.; Wang, K.W.; Zheng, Z.W.; Zhou, M. Lignans with Anti-Hepatitis B Virus Activities from Phyllanthus niruri L. Phytother. Res. 2017, 26, 964–968.
(9) Liu, S.; Wei, W.X.; Shi, K.C.; et al. In vitro and in vivo Anti-Hepatitis B Virus Activities of the Lignan Niranthin Isolated from Phyllanthus niruri L. J. Ethnopharmacol. 2014, 155, 1061–1067.
(10) Liu, S.; Wei, W.X.; Li, Y.B; et al. In vitro and in vivo Anti-Hepatitis B Virus Activities of the Lignan Nirtetralin B Isolated from Phyllanthus niruri L. J. Ethnopharmacol. 2014, 157, 62–68.
(11) Vikneswaran, M.; Chan, K.L. Determination of Four Lignans in Phyllanthus niruri L. by a Simple High performance Liquid Chromatography Method with Fluorescence Detection. J. Chromatogr. A 2007, 1154, 198–204.
(12) Vikneswaran, M.; Chan, K.L. Analysis of Lignans from Phyllanthus niruri L. in Plasma Using a Simple HPLC Method with Fluorescence Detection and Its Application in a Pharmacokinetic Study. J. Chromatogr. B. 2007, 852, 138–144.
Jianzeng Xin and Sheng Liu are with the Key Laboratory of Molecular Pharmacology & Drug Evaluation, the Collaborative Innovation Center of Advances Drug Delivery Systems, and the School of Pharmacy, School of Life Sciences, at Yantai University, in Shandong, China. Direct correspondence to Sheng Liu at: liusheng87@126.com

New Study Reviews Chromatography Methods for Flavonoid Analysis
April 21st 2025Flavonoids are widely used metabolites that carry out various functions in different industries, such as food and cosmetics. Detecting, separating, and quantifying them in fruit species can be a complicated process.
Extracting Estrogenic Hormones Using Rotating Disk and Modified Clays
April 14th 2025University of Caldas and University of Chile researchers extracted estrogenic hormones from wastewater samples using rotating disk sorption extraction. After extraction, the concentrated analytes were measured using liquid chromatography coupled with photodiode array detection (HPLC-PDA).

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)