Novel Sorptive Sample Preparation Techniques for Separation Science
The primary analytical challenge is to selectively extract the target analytes using a suitable sample preparation technique and introduce them into the downstream analytical instrument. The critical step in the chemical analysis is sample preparation. Sorptive sample preparation techniques are among the new generation of microextraction approaches, and are compliant with green analytical chemistry principles. A recent intercontinental collaboration between two academic research laboratories—the Aristotle University of Thessaloniki, Greece, and the Florida International University, USA—has yielded a significant number of analytical/bioanalytical methods using fabric phase sorptive extraction (FPSE), magnet integrated fabric phase sorptive extraction (MI-FPSE), and capsule phase microextraction (CPME) for the isolation of various analytes from different complex sample matrices. A brief description of these techniques with regards to principle, synthesis, applications, and advantages and disadvantages along with paradigms is presented.
Analytical chemists are routinely tasked with a wide range of challenges, ranging from statistically meaningful sampling all the way to the final step of reporting the analytical data. To execute a successful analytical workflow, all relevant factors must be thoughtfully considered so that reliable analytical data are produced as the final outcome of the process. As an integral component of the workflow, sensitive, precise, accurate, and robust analytical/bioanalytical methods are required to deal with a wide variety of complex sample matrices where target analytes may possess broadly dispersed physicochemical properties and be present at trace levels. In addition, many types of interfering compounds may also be present in the sample matrices, making the isolation of analytes of interest a challenging task.
Apparently, the most critical step in the analytical/bioanalytical workflow is sample preparation. No analytical/bioanalytical chemist can argue that this is also the most tedious, time-consuming, and error-prone step in the entire workflow, and may lead to erroneous results and wrong conclusions. At the same time, this is also the most challenging and uncertain step for separation scientists (1,2).
Fortunately, a plethora of classical and modern sample preparation techniques have been developed during the last couple of decades to achieve the following objectives:
1. To reduce the sample size, simplify the sample matrix and quantitative release of the target analytes from the sample matrix;
2. To make the sample compatible with the analytical instrument used;
3. To remove interferences and cleaning up the sample;
4. To preconcentrate the target analytes;
5. To obtain the highest amount of analytical information from the sample,
thus maximizing the advantages of sensitive and sophisticated instrumentation;
6. To meet the demands of modern high-throughput analytical/bioanalytical laboratories by optimizing relevant parameters including the analysis time, cost, sensitivity, and compliance with strict regulatory guidelines;
7. To comply with the principles of sustainable development and green analytical chemistry demands with regards to samples, reagents, method, waste, operator, and instrument (3,4).
As such, the primary challenge in sample preparation is to selectively and efficiently extract the target analytes from the sample matrix and elute in a suitable solvent compatible with the chromatographic system. As of now, different solvents, sorbent materials, extraction mechanisms, and design configurations are available.
Sorbent-based sorptive extraction techniques have enjoyed tremendous growth during the past decade and continue to be more popular compared to solvent-based microextraction techniques. It should be mentioned that sorbent-based extraction techniques are considered to be able to cover most of the analytical/bioanalytical sample preparation needs, if not all (4).
To this end, a recent bilateral academic research collaboration between two laboratories, one at the Aristotle University of Thessaloniki in Greece, and the other at Florida International University in the USA, have teamed up to advance the benefit of novel sol-gel-based material systems and unique delivery devices, which have yielded a significant number of novel analytical methods. The research collaboration was primarily focused on novel sorbent-based sample preparation techniques including fabric phase sorptive extraction (FPSE), magnet integrated fabric phase sorptive extraction (MI-FPSE), and capsule phase microextraction (CPME) for the isolation of various analytes from complex sample matrices such as environmental water, food, and biological fluids. Under a memorandum of understanding, Florida International University was responsible for developing new, environmentally friendly sample preparation technologies, while Aristotle University of Thessaloniki assumed the responsibility of developing new applications for these innovative sample preparation technologies. A brief description of these techniques with regards to principle, synthesis of sorbents, fabrication of the devices, and advantages and disadvantages along with paradigms are illustrated in this article.
Sorbent-Based Sorptive Extraction Techniques
The seminal invention of solid-phase microextraction (SPME) by Janusz Pawliszyn and Catherine L. Arthur in 1990 (5) and subsequent invention of stir-bar sorptive extraction (SBSE) by Erik Baltussen, Pat Sandra, Frank David, and Carel Cramers in 1999 (6) have initiated a new era of sorbent-based sample preparation technologies. These techniques gained attention as alternatives to solid-phase extraction (SPE) and liquid–liquid extraction (LLE) due to their solventless/solvent minimized operation and many other positive attributes. Although SPME, SBSE, and their different modifications including thin-film microextraction (TFME) (7) undoubtedly deserve credit for beginning a new direction in analytical/bioanalytical sample preparation known as “sorbent-based microextraction”, and for highlighting the importance of sample preparation in the overall analytical/bioanalytical workflow, they also suffer from several shortcomings. These include a limited number of commercially available sorbents, a narrow range of pH stability (pH 2–10), instability and swelling of the coating in organic solvents, poor extraction performance for highly polar analytes, and fragility of the coated fibres; in the case of SPME, limited commercially available sorbent chemistries and recommended only for nonpolar analytes (Log Kow >4), and for SBSE, the necessity of special instruments for thermal desorption.
Most of these shortcomings originate from the viscous nature of the pristine polymers (polydimethylsiloxane, polyethylene glycol) and the polymer immobilization technology on the substrate surface that is primarily based on a free-radical cross-linking process. The high viscosity of the polymers used as the sorbents in SPME and SBSE slows down the mass transfer of the analytes from the sample matrix to the extracting sorbent, resulting in prolonged sample preparation time. In addition, SPME and SBSE do not provide sample preparation solutions for acidic, basic, or zwitterionic compounds. Both techniques primarily serve for gas chromatographic (GC) separation, although there are a few SPME fibres for liquid chromatographic (LC) analysis via solvent desorption, with a potential risk of sorbent coating loss. It is fair to say that SPME and SBSE do not provide the complete sample preparation solution that separation scientists need to serve their analytical challenges (5–8).
As such, many academic research groups have continued to address the shortcomings of SPME and SBSE by developing new sorbent chemistries that possess higher extraction efficiency, faster extraction kinetics, selectivity, pH stability, porosity, and thermal stability. Unfortunately, these new sorbents are not commercially available.
A critical review on the desired attributes of a robust sample preparation technique reveals that such a technique should deliver the following qualities:
(a) Capable of handling all sorts of original samples (biological, environmental, pharmaceutical, food, forensic) without any matrix clean-up to minimize irreversible loss of analytes during the sample preparation;
(b) Capable of simultaneously preconcentrating neutral, acidic, and basic, as well as trace metals;
(c) Able to prepare the sample in a way that can be analyzed by GC, LC, or capillary electrophoresis (CE) to maximize the analytical information;
(d) Easily field-deployable to reduce the cost of transportation, minimize the change in the sample matrix during transportation and storage, and minimize the potential loss of analytes due to the adsorption by the collection container;
(e) Able to eliminate or minimize the usage of organic solvent;
(f) Should be able to support the extensive demands of high-throughput laboratories;
(g) Compatible with green analytical chemistry principles, and meeting as many of these as possible with regards to solvent consumption, energy consumption, waste produced, human health, and environmental impact;
(h) Able to retain the extracted analytes for a prolonged period so that instrument breakdown/natural calamity/power failure or other unintended incidents do not incur any economic/scientific loss to the concerned laboratory;
(i) Economical and able to favour multiple use to reduce the overall cost;
(j) Able to select the best sorbent material for a specific application using a mathematical model-based approach without requiring a subjective decision;
(k) Able to simplify the overall sample preparation workflow;
(l) Able to offer a holistic solution so that an analyst doesn’t need to use different techniques for different type of samples or analytes;
(m) Able to maximize the number of available sorbents so that the analyst has enough freedom in sorbent selection (9).
The players in the “game” of sample preparation include: analytical performance, time and safety issues, sample matrix, sample type, interferences and undesired components, analytes of interest with regards to their physicochemical characteristics and concentration levels, and the analytical instrumentation with regards to the operating costs and availability in the marketplace.
Conventional sample preparation techniques such as SPE and LLE may exhibit better extraction efficiency or clean-up performance, but modern microextraction techniques comply better with the demands for green analytical chemistry principles. A new principle, known as white analytical chemistry, has also been proposed recently that aims to reconcile the principles of green analytical chemistry and functionality (10).
Sorptive extraction techniques are widely used prior to chromatographic analyses in a range of applications. FPSE, MI-FPSE, and CPME are notable among the novel approaches, with minimum steps needed to achieve satisfactory clean-up and analyte preconcentration, without any prior (filtration/centrifugation/protein precipitation) or post (solvent evaporation
and sample reconstitution) sample modification needed. These novel sample preparation techniques were introduced recently by the research group in Florida International University, as described in the patents cited in the references herein (11–14).
Fabric Phase Sorptive Extraction (FPSE)
Fabric phase sorptive extraction is a new generation sample preparation technique that was invented to address most of the sample preparation challenges that other sorptive microextraction techniques do not. Unlike other microextraction techniques, FPSE combines the extraction principles of SPME (equilibrium extraction) and SPE (exhaustive extraction) by its unique design. The simultaneous exploitation of the two major extraction principles into a single device results in faster mass transfer, short sample preparation time, and exhaustive or near exhaustive extraction of the target analytes even when the extraction is performed in direct immersion extraction mode (as practised in SPME). FPSE also exploits the surface chemistry of the fabric substrate (hydrophilic/hydrophobic), linker chemistry (polarity enhancer or reducer), and organic/organic-inorganic polymer chemistry that collectively determine the overall selectivity and extraction efficiency of the FPSE membrane. By changing any of the three building blocks of the FPSE membrane, one may fine-tune the selectivity and extraction efficiency of the membrane. Unlike classical microextraction techniques, FPSE has adopted sol-gel coating technology for the sorbent coating. A sol-gel coating chemically binds the organic/organic-inorganic polymer to the fabric substrate via a sol-gel linker that results in thermal, chemical, and solvent stability to the sorbent coating, with pH stability ~1–13. Sol-gel-derived sorbents are inherently porous and possess sponge-like porous morphology. As a result, the sample matrix can easily permeate through the sorbent as well as the pores of the membrane and the analytes are extracted onto the sol-gel sorbent coated on the FPSE membrane via numerous analyte-sorbent intermolecular/ionic interactions. Due to the strong intermolecular/ionic interactions between the analytes and the sorbent, analytes remain retained on the FPSE membrane for a prolonged time (more than a year), even when the FPSE membranes are stored in a regular laboratory environment. The sponge-like porous morphology of the sol-gel sorbent requires a very small volume of organic solvent for quantitative analyte back-extraction. As a result, no solvent evaporation and sample preconcentration is needed in a FPSE workflow, minimizing the risk of analyte loss and improving the quality of the analytical data (15).
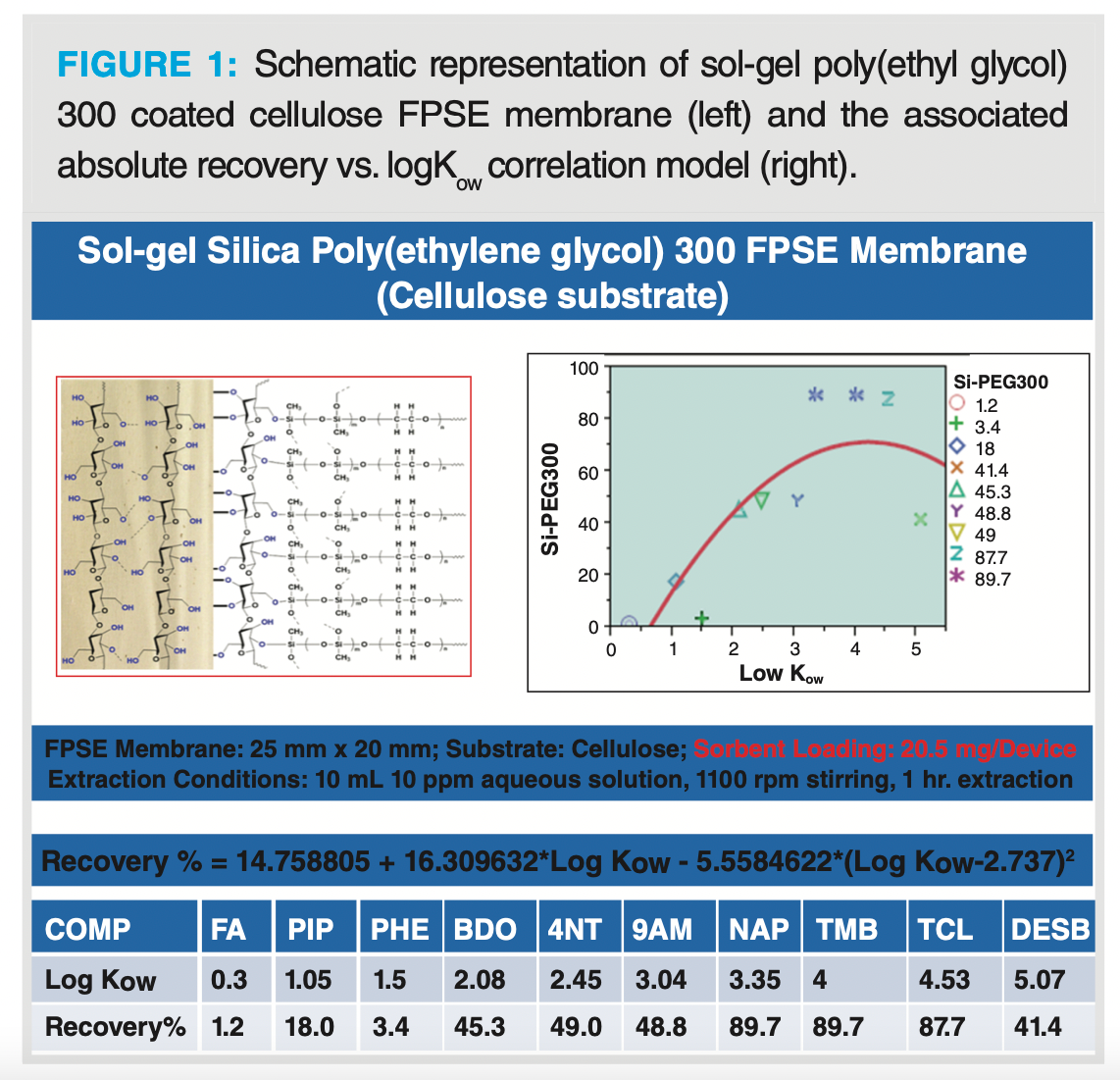
Fabric phase sorptive extraction has also expanded the available sorbent chemistries by developing phases that are either exclusively used as SPME and SBSE coating or as an SPE sorbent. As of now, FPSE offers ~30 unique sorbent phases including polydimethyl siloxane (PDMS), polyethylene glycol (PEG), polyacrylate (PA), C18, C8, C4, cation exchanger, anion exchanger, mixed-mode, zwitterionic, and multimode zwitterionic sorbents. The selection of an SPME fibre and SBSE phase primarily depends on the experience of the analyst. To eliminate the error involved in the subjective judgement of the analyst during sorbent selection for a given application, FPSE has developed a mathematical model-based approach using the logKow (octanol-water coefficient) values of the analytes. Every FPSE sorbent chemistry has its own absolute recovery calculation equation. The analyst can easily calculate the predicted percentage absolute recovery value of a specific analyte on a particular FPSE sorbent using this model, and can decide which sorbent to try for the analysis (9,16). For example, absolute recovery in sol-gel poly(ethyl glycol) 300 coated cellulose FPSE membrane can be calculated using the equation: Recovery % = 14.758805 +16.309632*Log Kow - 5.5584622*(Log Kow-2.737)2. Figure 1 demonstrates the quadratic mathematical model developed for a sol‑gel poly(ethylene glycol)-coated cellulose FPSE membrane. The model was created using 10 compounds at a concentration of 10 µg/mL. Extraction conditions are stated in Figure 1. The 10 test compounds with varied physicochemical properties and functional compositions included: furfural alcohol (FA), piperonal (PIP), phenol (PHE), 4-nitrotoluene (4NT), 9-anthracene methanol (9AM), naphthalene (NAP), tetramethyl benzene (TMB), tricosan (TCL), and diethylstilbestrol (DESB). As can be seen, the absolute recovery of a test compound depends on the logKow as well as the functional composition of the analyte. As such, absolute recovery of phenol was lower than piperonal in sol-gel poly(ethyl glycol) 300 sorbent, despite the fact that piperonal is more polar than phenol.
Unlike other sorbent-based extraction and microextraction techniques, FPSE simultaneously exploits the surface chemistry of at least three building blocks: (a) the fabric substrate; (b) the sol-gel linker; and (c) the organic/inorganic-organic polymer. The overall polarity/selectivity/extraction efficiency of an FPSE membrane depends on all three building blocks, although the individual impact may not be equal. The FPSE membranes behave differently when the substrate is changed from 100% cotton cellulose to 100% polyester, keeping the other two building blocks unchanged. A schematic representation of an FPSE membrane and its different building blocks is presented in Figure 2.
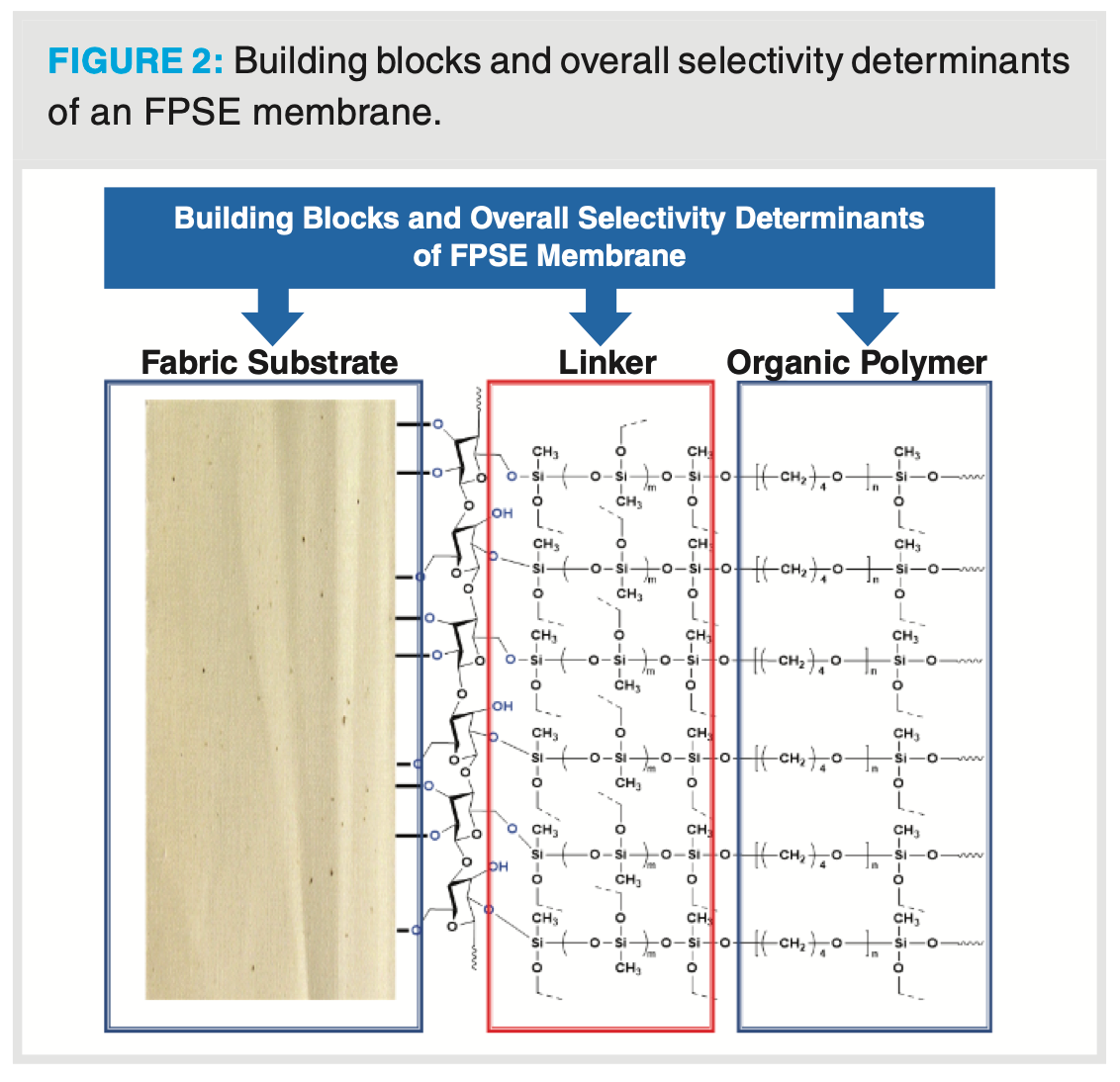
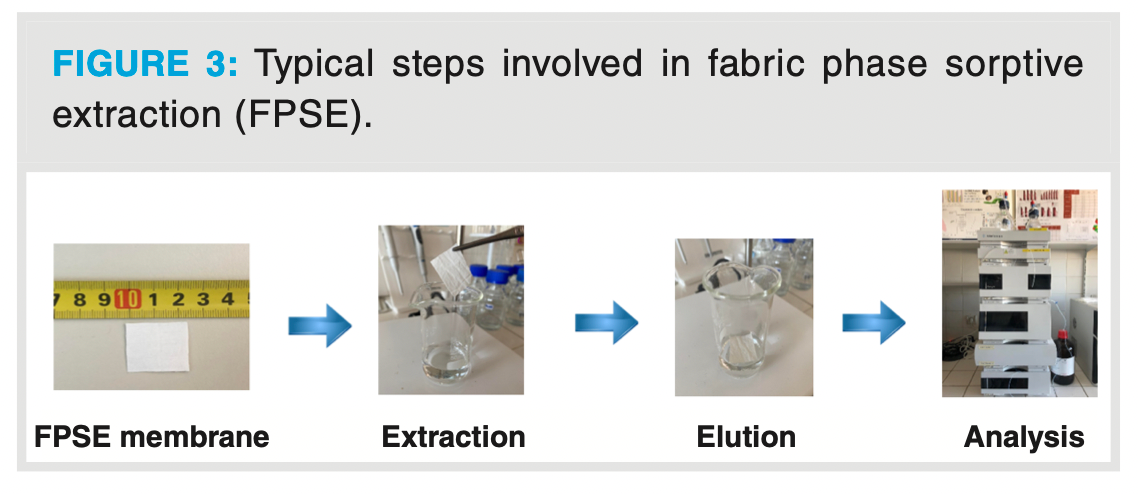
Figure 3 displays the typical steps involved in an FPSE process.
Magnet Integrated Fabric Phase Sorptive Extraction (MI-FPSE)
Magnet integrated fabric phase sorptive extraction is a new type of FPSE that was created (a) to improve the extraction reproducibility; (b) to expedite the analyte mass transfer; (c) to eliminate the necessity of an external magnet; and (d) to use multiple sorbent chemistry in-tandem to maximize the extractive power when a wide range of analytes (neutral, acidic, basic, polar, nonpolar) are simultaneously present in the sample matrix and are all analytes of interest. MI-FPSE offers all the advantages of classical FPSE in addition to several others. Due to the precise control of the spinning rate (rpm), MI-FPSE demonstrates higher run-to-run extraction reproducibility. In addition, the sample matrix permeates through the FPSE membrane in MI-FPSE at a faster rate compared to classical FPSE, with the extraction equilibrium a significantly shorter time. Figure 4 presents MI-FPSE devices and the extraction process (17).

MI-FPSE improves extraction reproducibility by providing controllable spinning on a magnetic stirrer. During the spinning, it collides with the aqueous sample matrix and forces the sample to permeate through the membrane, resulting in faster mass transfer and shorter extraction equilibrium times. Although FPSE allows sandwiching of several FPSE membranes together, MI‑FPSE by design requires a minimum of two membranes in the device. As a result, MI-FPSE allows easy adaptation of different sorbent chemistries to maximize the extraction efficiency.
Capsule Phase Microextraction (CPME)
Although FPSE and MI-FPSE have provided a holistic solution to the analytical challenges often encountered in modern analytical and bioanalytical laboratories, when the sample contains high percentages of interfering components such as plant debris, soil particles, proteins, fat molecules, and other cellular debris, the sample generally requires sample pretreatment processes such as filtration or centrifugation. Sample pretreatment is not only laborious and time-consuming but it may also inflict loss of analytes during the process. Direct contact of the interfering elements with the sorbent may shorten its active service life. The interferents may irreversibly adsorb onto the surface of the extraction sorbent coating and severely impede analyte-sorbent interaction, resulting in poor extraction recovery. To eliminate the need for sample pretreatment from the sample preparation workflow, the CPME device was developed as a stand-alone sample preparation device. The apparatus works as a filtration and extraction device, with built-in stirring capability. Figure 5 presents the CPME device, the scanning electron microscope (SEM) image of the membrane, and the image of a magnet used in the device fabrication (18).

CPME devices utilize two porous polypropylene tubes with internal diameter (i.d.) 1.8 mm, outside diameter (o.d.) 2.7 mm, and wall thickness 0.45 mm conjoined together with a magnet encapsulated in one tube and the sorbent microparticles in the other. The sol-gel-derived sorbents are distributed within the pores of the tube as well as in the lumen of the tube. The sorbent is created by an in situ sol-gel synthesis while keeping the porous polypropylene tubes submerged into the sol solution. The sol solution enters the porous tubular membrane and fills up the lumen. The sol solution subsequently turns into a solid gel, forming scattered particles inside the porous walls and a monolithic bed inside the lumen of the empty tube. To expand the contact surface area of the sol-gel sorbent, the CPME device is subjected to ultra-sonication, during which the monolithic bed inside the lumen disintegrates into microparticles and expands the available contact surface area greatly. As such, CPME devices possess equivalent sorbent loading and comparable contact surface area when compared to FPSE membranes. When a CPME device is exposed to the sample for analyte extraction, the aqueous sample easily permeates through the porous walls (0.2 μm pore size) and the analytes interact with the sorbent particles trapped inside the porous walls, as well as with the particles inside the lumen, and are extracted rapidly. The porous walls of the device act like a filter. As such, the device can be inserted directly in a sample containing high percentages of matrix interferents. The magnet encapsulated within the device spins the device on a magnetic stirrer. After the extraction, the CPME device is transferred into a back-extraction tube and back-extraction is performed using 200–500 μL organic solvent. Similar to FPSE and MI-FPSE, an CPME device can be used many times without sacrificing its extracting power (18).
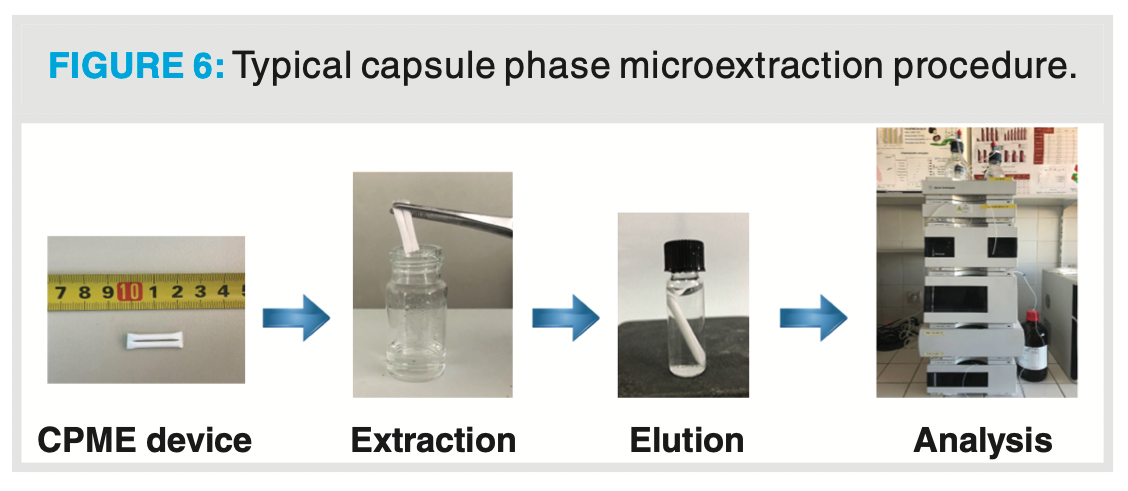
The workflow for CPME is presented in Figure 6.
One can compare a CPME device with hollow fibre membrane-protected SPME; however, they are built on very different concepts. Hollow fibre membrane-protected SPME utilizes a porous polypropylene capillary to protect the SPME coating from fouling. It does not have a built-in stirring mechanism. A CPME device is a stand-alone sample preparation device with built-in filtration and mechanical stirring capability. The sol-gel monolith sorbent technology allows the sol-gel sorbent to be created as a monolithic bed within the lumen, as well as inside the porous walls of the polypropylene capillary tube. The monolithic bed of the sol-gel sorbent is disintegrated into microparticles. In this regard, a CPME device mimics a protected micro-SPE device.
FPSE, MI-FPSE, and CPME Method Development
Similar to classical sorbent-based microextraction techniques, FPSE, MI‑FPSE, and CPME need to optimize several process parameters to maximize the extraction efficiency of the target analytes from a given sample matrix. These process parameters include: (a) selection of the sorbent chemistry; (b) optimization of the sample volume; (c) optimization of the extraction time; (d) optimization of the desorption solvent; (e) optimization of the desorption time; and (f) optimization of the stirring speed. The volume of the desorption solvent can be optimized, but instead of optimizing the solvent volume, the solvent volume can be set at the level that is needed to submerge the membrane or the device. If the analytes are polar, evaluating the impact of adding salt to the sample is also recommended. The addition of salt decreases the solubility of polar compounds in water and forces the analytes to be extracted into the microextraction membrane or device. Instead of this one factor at a time (OFAT) approach, statistical design of experiments (DOE) can also be used to screen the relevant process parameters and optimize the most applicable parameters using response surface methodology (RSM) (9).
Applications of FPSE, MI‑FPSE, and CPME
The successful and highly productive research collaboration was begun in 2014, and 22 analytical/bioanalytical methods have now been published, covering a wide range of applications. A list of these methods are presented in Table 1 (19–39).
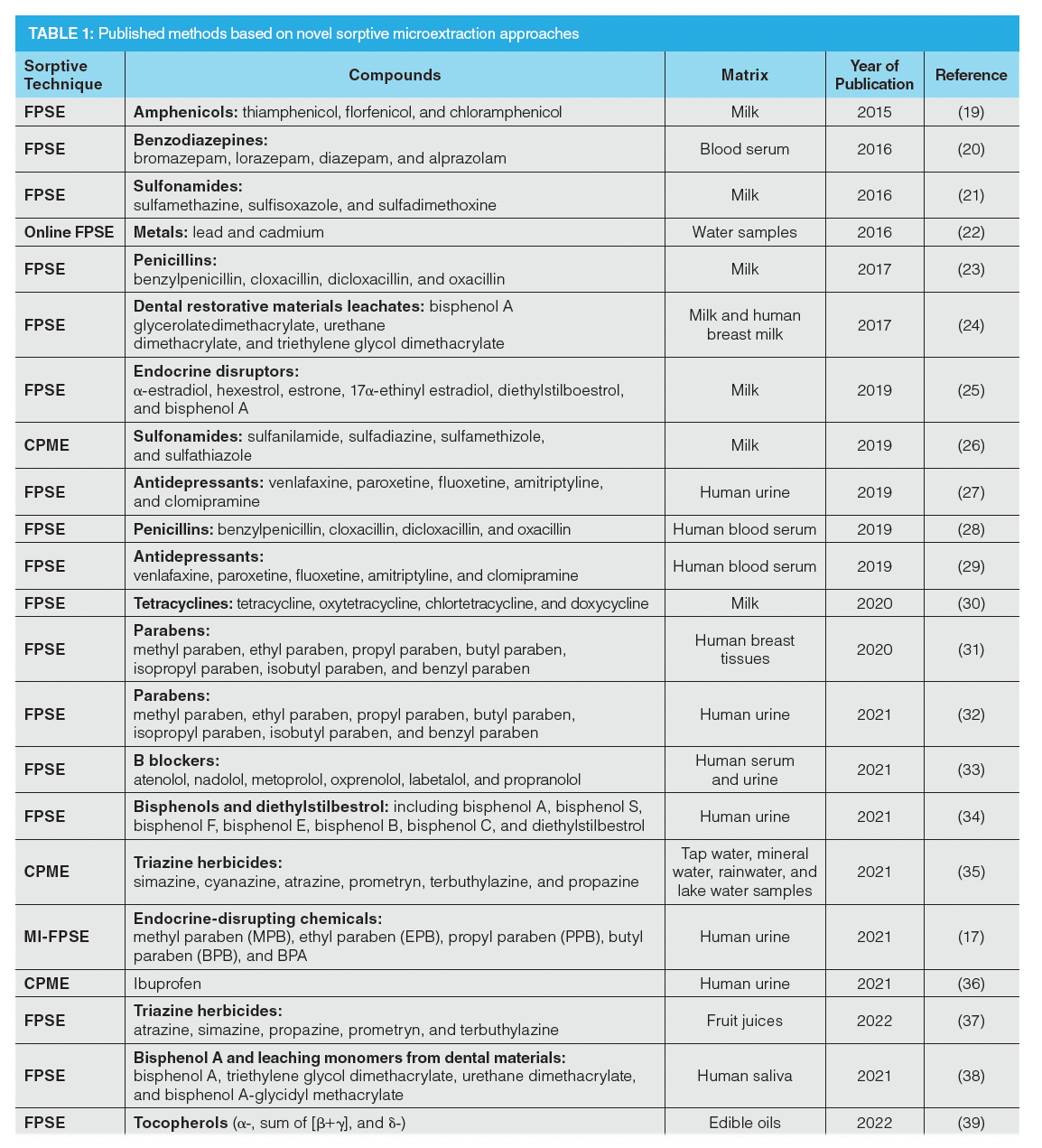
All sample preparation materials were prepared at the International Forensic Research Institute, Department of Chemistry and Biochemistry of Florida International University, and the protocols in each application were developed and evaluated in the Laboratory of Analytical Chemistry at the School of Chemistry of Aristotle University of Thessaloniki.
It should be mentioned that FPSE, MI-FPSE, and CPME offer many biocompatible sorbents. As a result, no protein precipitation step was needed when dealing with any biological samples presented in the list.
Conclusions
The new generation sorbent-based sorptive extraction techniques based on FPSE, MI-FPSE, and CPME seem to be very promising for the extraction and preconcentration of various target analytes from a wide range of sample matrices. The unique geometrical shape and characteristics of the FPSE membrane result in the rapid extraction of analytes due to the fast extraction equilibrium. Usage of a small volume of solvent, as well as elimination of sample pretreatment, solvent evaporation, and sample reconstitution steps from the sample preparation workflow have made these sample preparation techniques environmentally friendly and cost-effective. With regards to bioanalysis, the FPSE membrane can be directly introduced into the whole blood, intact urine, or unfiltered saliva samples without deproteinization or other sample pretreatment processes. A minimum number of sample preparation steps reduces potential sources of errors. The direct introduction to the high performance liquid chromatography (HPLC) instrument after extraction makes this method suitable for routine analysis either for clinical studies or forensic purposes. The FPSE membrane can be reused several times depending on the application.
MI-FPSE efficiently integrates the stirring and extraction mechanism into a single sample preparation device. This configuration permits the development of simple, direct, reproducible, and accurate processes. Fast extraction equilibrium can be achieved, resulting in shorter extraction times. The extraction device can be reused several times with no carryover effect and no discernible loss in the extraction efficiency.
CPME offers simplicity in sample handling and rapid extraction kinetics, and it can be considered as an environmentally friendly extraction technique compared to conventional sample preparation approaches. Moreover, the extraction step can be parallelized (for example, on a multi‑position stirring plate) for the simultaneous preparation of many samples. The microextraction capsules are reusable at least 10 times. High enrichment factors result in satisfactory limits of detection (LOD) and limits of quantification (LOQ) values that may also be improved using more sensitive instrumentation (LC–tandem mass spectrometry [MS/MS] or GC–MS/MS instruments).
Finally, an important benefit of CPME is that the method does not require sample filtration prior to the sample preparation. As such, CPME can be employed for the direct sample preparation of environmental samples or biological samples that contain solid particles.
References
- Malcangi, S.; Romagnoli, M.; Beccaria, M.; et al. Modern Sample Preparation Approaches for Small Metabolite Elucidation to Support Biomedical Research. Adv. Sample Prep. 2022, 2, e100017. DOI: 10.1016/j.sampre.2022.100017
- Clark, K. D.; Zhang, C.; Anderson, J. L. Sample Preparation for Bioanalytical and Pharmaceutical Analysis. Anal. Chem. 2016, 88 (23), 11262–11270. DOI: 10.1021/acs.analchem.6b02935
- Gałuszka, A.; Migaszewski, Z.; Namieśnik, J. The 12 Principles of Green Analytical Chemistry and the SIGNIFICANCE Mnemonic of Green Analytical Practices. Trends Anal. Chem. 2013, 50, 78–84. DOI: 10.1016/j.trac.2013.04.010
- Kabir, A.; Locatelli, M.; Ulusoy, H. I. Recent Trends in Microextraction Techniques Employed in Analytical and Bioanalytical Sample Preparation. Separations 2017, 4, 36. DOI: 10.3390/separations4040036
- Arthur, C. L.; Pawliszyn, J. Solid Phase Microextraction with Thermal Desorption Using Fused Silica Optical Fibers. Anal. Chem. 1990, 62 (19), 2145–8. DOI: 10.1021/ac00218a019
- Baltussen, E.; Sandra, P.; David, F.; Cramers, C. Stir Bar Sorptive Extraction (SBSE), A Novel Extraction Technique for Aqueous Samples: Theory and Principles. J. Microcolumn Separations 1999, 11 (10), 737–747. DOI: 10.1002/(SICI)1520-667X(1999)11:10<737::AID-MCS7>3.0.CO;2-4
- Bruheim, I.; Liu, X. C.; Pawliszyn, J. Thin-Film Microextraction. Anal. Chem. 2003, 75, 1002–1010. DOI: 10.1021/ac026162q
- Lord, H.; Pawliszyn, J. LCGC Supplement 1999, 17 (9S), S25–S30.
- Kabir, A.; Samanidou, V. Fabric Phase Sorptive Extraction: A Paradigm Shift Approach in Analytical and Bioanalytical Sample Preparation. Molecules 2021, 26, 865. DOI: 10.3390/molecules26040865
- Nowak, P. M.; Wietecha-Posłuszny, R.; Pawliszyn, J. White Analytical Chemistry: An Approach to Reconcile the Principles of Green Analytical Chemistry and Functionality.Trends Anal Chem. 2021, 138, 116223. DOI: 10.1016/j.trac.2021.116223
- Kabir, A.; Furton, K. G. Fabric Phase Sorptive Extractors. US 9283544, 2014.
- Kabir, A.; Furton, K. G. Fabric Phase Sorptive Extractors. US 9557252, 2016.
- Kabir, A.; Furton, K. G. Field Sampling Kit for Chemical Recovery, Storage, and Profiling, Method of Making and Using the Kit, and Dynamic Fabric Phase Sorptive Extraction (DFPSE) Medium. US 9527059, 2016.
- Kabir, A.; Furton, K. G. Microextraction Capsules and Method of Making. US 10352833, 2019.
- Kabir, A.; Furton, K. G.; Malik, A. Innovations in Sol-Gel Microextraction Phases for Solvent-Free Sample Preparation in Analytical Chemistry. Trends Anal. Chem. 2013, 45, 197–218. DOI: 10.1016/j.trac.2012.11.014
- Zilfidou, E.; Kabir, A.; Furton, K. G.; Samanidou, V. Fabric Phase Sorptive Extraction: Current State of the Art and Future Perspectives. Separations 2018, 5 (3), 40. DOI: 10.3390/separations5030040
- Alampanos, V.; Kabir, A.; Furton, K.; Samanidou, V. Magnet Integrated Fabric Phase Sorptive Extraction of Selected Endocrine Disrupting Chemicals from Human Urine Followed by High-Performance Liquid Chromatography - Photodiode Array Analysis. J. Chromatogr. A 2021, 1654, 462459. DOI: 10.1016/j.chroma.2021.462459
- Samanidou, V.; Georgiadis, D. -E.; Kabir, A.; Furton, K. G. Capsule Phase Microextraction: The Total and Ultimate Sample Preparation Approach. J. Chromatogr Sep. Tech 2018, 9, 395. DOI: 10.4172/2157-7064.1000395
- Samanidou, V.; Kabir, A.; Galanopoulos, L.; Furton, K. G. Fast Extraction of Amphenicols Residues from Raw Milk Using Novel Fabric Phase Sorptive Extraction Followed by High-Performance Liquid Chromatography-Diode Array Detection. Anal. Chim. Acta 2015, 855, 41–50. DOI: 10.1016/j.aca.2014.11.036
- Samanidou, V.; Kaltzi, I.; Kabir, A.; Furton, K. G. Simplifying Sample Preparation Using Fabric Phase Sorptive Extraction Technique for the Determination of Benzodiazepines in Blood Serum by High-Performance Liquid Chromatography. Biomed. Chromatogr. 2016, 30 (6), 829–836. DOI: 10.1002/bmc.3615
- Karageorgou, E.; Manousi, N.; Samanidou, V.; Kabir, A.; Furton, K. G. Fabric Phase Sorptive Extraction for the Fast Isolation of Sulfonamides Residues from Raw Milk Followed by High Performance Liquid Chromatography with Ultraviolet Detection. Food Chem. 2016, 196, 428–436. DOI: 10.1016/j.foodchem.2015.09.060
- Anthemidis, A.; Kazantzi, V.; Samanidou, V.; Kabir, A.; Furton, K. G. An Automated Flow Injection System for Metal Determination by Flame Atomic Absorption Spectrometry Involving On-Line Fabric Disk Sorptive Extraction Technique. Talanta 2016, 156–157, 64–70. DOI: 0.1016/j.talanta.2016.05.012
- Samanidou, V.; Michaelidou, K.; Kabir, A.; Furton, K. G. Fabric Phase Sorptive Extraction of Selected Penicillin Antibiotic Residues from Intact Milk Followed by High Performance Liquid Chromatography with Diode Array Detection. Food Chem. 2017, 224, 131–138. DOI: 10.1016/j.foodchem.2016.12.024
- Philippou, O.; Marinou, E.; Kabir, A.; Furton K.; Samanidou, V. Sol-Gel-Graphene-Based Fabric-Phase Sorptive Extraction for Cow and Human Breast Milk Sample Cleanup for Screening Bisphenol A and Residual Dental Restorative Material Before Analysis by HPLC with Diode Array Detection. J. Sep. Sci. 2017, 40 (12), 2612–2619. DOI: 10.1002/jssc.201700256
- Mesa, R.; Kabir, A.; Samanidou, V.; Furton, K. G. Simultaneous Determination of Selected Estrogenic Endocrine Disrupting Chemicals and Bisphenol A Residues in Whole Milk Using Fabric Phase Sorptive Extraction Coupled to HPLC-UV Detection and LC-MS/MS. J. Sep. Sci. 2019, 42, 598–608. DOI: 10.1002/jssc.201800901
- Georgiadis, D. -E.; Tsalbouris, A.; Kabir, A.; Furton, K.; Samanidou, V. Novel Capsule Phase Microextraction in Combination with High Performance Liquid Chromatography with Diode Array Detection for Rapid Monitoring of Sulfonamide Drugs in Milk. J. Sep Sci. 2019, 42, 1440–1450. DOI: 10.1002/jssc.201801283
- Lioupi, A.; Kabir, A.; Furton, K.; Samanidou, V. Fabric Phase Sorptive Extraction for the Isolation of Five Common Antidepressants from Human Urine Prior to HPLC-DAD Analysis. J. Chromatogr. B 2019, 1118–1119, 171–179. DOI: 10.1016/j.jchromb.2019.04.045
- Alampanos, V.; Kabir, A.; Furton, K. G.; Samanidou, V.; Papadoyannis, I. Fabric Phase Sorptive Extraction for Simultaneous Observation of Four Penicillin Antibiotics from Human Blood Serum Prior to High Performance Liquid Chromatography and Photo-Diode Array Detection. Microchem. J. 2019, 149, 1039642. DOI: 10.1016/j.microc.2019.103964
- Zilfidou, E.; Kabir, A.; Furton, K.; Samanidou, V. An Improved Fabric Phase Sorptive Extraction Method for the Determination of Five Selected Antidepressant Drug Residues in Human Blood Serum Prior to High Performance Liquid Chromatography with Diode Array Detection. J. Chromatogr B 2019, 1125, 121720. DOI: 10.1016/j.jchromb.2019.121720
- Agadellis, E.; Tartaglia, A.; Locatelli, M.; et al. Mixed-Mode Fabric Phase Sorptive Extraction of Multiple Tetracycline Residues from Milk Samples Prior to High Performance Liquid Chromatography-Ultraviolet Analysis. Microchemical J. 2020, 159, 105437. DOI: 10.1016/j.microc.2020.105437
- Alampanos, V.; Kabir, A.; Furton, K.; et al. Fabric Phase Sorptive Extraction Combined with High-Performance-Liquid Chromatography-Photodiode Array Analysis for the Determination of Seven Parabens in Human Breast Tissues: Application to Cancerous and Non-Cancerous Samples. J. Chromatogr A 2020, 1630, 461530. DOI:10.1016/j.chroma.2020.461530
- Rigkos, G.; Alampanos, V.; Kabir, A.; et al. An Improved Fabric-Phase Sorptive Extraction Protocol for the Determination of Seven Parabens in Human Urine by HPLC–DAD. Biomed Chromatogr. 2021, 35 (2), e4974. DOI: 10.1002/bmc.4974
- Mazaraki, K.; Kabir, A.; Furton, K. G.; et al. Fast Fabric Phase Sorptive Extraction of Selected β-blockers from Human Serum and Urine Followed by UHPLC-ESI-MS/MS Analysis. J. Pharm. Biomed. Anal. 2021, 199, 114053. DOI: 10.1016/j.jpba.2021.114053
- Alampanos, V.; Kabir, A.; Furton, K. G.; Samanidou, V. Rapid Exposure Monitoring of Six Bisphenols and Diethylstilbestrol in Human Urine Using Fabric Phase Sorptive Extraction Followed by High Performance Liquid Chromatography - Photodiode Array Analysis. J. Chromatogr. B 2021, 1177, 122760. DOI: 10.1016/j.jchromb.2021.122760
- Manousi, N.; Alampanos, V.; Priovolos, I.; et al. Designing a Moderately Hydrophobic Sol-Gel Monolithic Carbowax 20 M Sorbent for the Capsule Phase Microextraction of Triazine Herbicides from Water Samples Prior to HPLC Analysis. Talanta 2021, 234, 122710. DOI: 10.1016/j.talanta.2021.122710
- Manousi, N.; Kabir, A.; Furton, K.; Samanidou, V.; Zacharis, C. Exploiting the Capsule Phase Microextraction Features in Bioanalysis: Extraction of Ibuprofen from Urine Samples. Microchemical J. 2022, 172 (4), 106934. DOI:10.1016/j.microc.2021.106934
- Manousi, N.; Alampanos, V.; Priovolos, I.; et al. Exploring Sol-Gel Zwitterionic Fabric Phase Sorptive Extraction Sorbent as a New Multi-Mode Platform for the Extraction and Preconcentration of Triazine Herbicides from Juice Samples. Food Chemistry 2022, 373 Part B, 31517. DOI: 10.1016/j.foodchem.2021.131517
- Andreasidou, E.; Mourouzis, P.; Daktylidi, L.; et al. A Fabric Phase Sorptive Extraction Method for the LC-UV Determination of Bisphenol A and Leaching Monomers from Dental Materials in Human Saliva. J. Chromatography B 2022, 1188, 1230731. DOI: 10.1016/j.jchromb.2021.123073
- Kalogiouri, N.; Kabir, A.; Olayanju, B.; Furton, K. G.; Samanidou, V. Development of Highly Hydrophobic Fabric Phase Sorptive Extraction Membranes and Exploring their Applications for the Rapid Determination of Tocopherols in Edible Oils Analyzed by High Pressure Liquid Chromatography-Diode Array Detection. J. Chromatogr. A 2022, 1664, 462785. DOI: 10.1016/j.chroma.2021.462785
Victoria Samanidou is full professor, director of the Laboratory of Analytical Chemistry, and vice president of the School of Chemistry of Aristotle University of Thessaloniki, Greece.
Abuzar Kabir is a research associate professor at the Department of Chemistry and Biochemistry, Florida International University, Miami, Florida, USA.

Thermodynamic Insights into Organic Solvent Extraction for Chemical Analysis of Medical Devices
April 16th 2025A new study, published by a researcher from Chemical Characterization Solutions in Minnesota, explored a new approach for sample preparation for the chemical characterization of medical devices.
Study Explores Thin-Film Extraction of Biogenic Amines via HPLC-MS/MS
March 27th 2025Scientists from Tabriz University and the University of Tabriz explored cellulose acetate-UiO-66-COOH as an affordable coating sorbent for thin film extraction of biogenic amines from cheese and alcohol-free beverages using HPLC-MS/MS.
Multi-Step Preparative LC–MS Workflow for Peptide Purification
March 21st 2025This article introduces a multi-step preparative purification workflow for synthetic peptides using liquid chromatography–mass spectrometry (LC–MS). The process involves optimizing separation conditions, scaling-up, fractionating, and confirming purity and recovery, using a single LC–MS system. High purity and recovery rates for synthetic peptides such as parathormone (PTH) are achieved. The method allows efficient purification and accurate confirmation of peptide synthesis and is suitable for handling complex preparative purification tasks.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)





