Hydrophobic Interaction Chromatography for Biopharmaceutical Analysis
LCGC North America
HIC analysis is carried out using a reversed salt gradient, starting with a high salt concentration and moving to low salt concentration to facilitate protein elution.
Hydrophobic interaction chromatography (HIC) is a milder form of reversed-phase liquid chromatography (LC). Separation of analytes is based on hydrophobic interactions with the stationary phase; therefore, the elution order in HIC enables proteins to be ranked on the basis of their relative hydrophobicity. HIC employs nondenaturing conditions, does not require the use of organic solvents or high temperatures, and separations are carried out at physiological pH, which allows the preservation of protein structure. Historically HIC has been used for the determination of the relative hydrophobicity of proteins and was applied on a preparative scale for protein purification; it is now applied at all stages of the purification process, including high-yield capture, polishing monoclonal antibodies (mAbs), characterization of mAbs and antibody–drug conjugates (ADCs), removal of truncated species from full-length forms, separation of active and inactive forms, and clearing viruses.
HIC analysis is carried out using a reversed salt gradient, starting with a high salt concentration and moving to a low salt concentration to facilitate protein elution. When starting at a high salt concentration, proteins are retained on the stationary phase because the hydrophobic interaction between the solute and stationary phase is increased by the salt in the buffer, which reduces the solvation of the proteins; as solvation decreases hydrophobic regions become exposed and are adsorbed to the stationary phase. The more hydrophobic the molecule the less salt is needed to promote binding.
The mobile phase is typically an aqueous solution of a nondenaturing salt such as ammonium sulfate (1–2 M) or sodium chloride (3 M) and a buffer to control pH (usually phosphate, 6 ≤ pH ≤ 7). High concentrations of salt, especially ammonium sulfate, may precipitate proteins; therefore, solubility should be checked under the initial gradient (binding) conditions. The strength of the interaction between the protein and stationary phase decreases with increasing pH as a result of increased charge on the protein caused by the ionization of acidic groups. This effect can vary depending on the protein; thus, pH can impact the level of protein binding and selectivity. Changes in pH do not have a significant effect over moderate ranges.
Stationary phases generally consist of silica bonded with ligands of relatively limited hydrophobicity (for example, butyl, phenyl, ether, amide, or propyl). Straight-chain alkyl ligands exhibit hydrophobic character, whereas aryl ligands provide mixed-mode behavior where both aromatic and hydrophobic interactions are possible. The choice of ligand type will be determined empirically. Normally stationary phases are nonporous; however, there are also some porous polymethacrylate-based particles available. HIC columns can be operated at pressures in the 100–400 bar range, and are typically packed with 5-, 3-, or 2.5-µm particles.
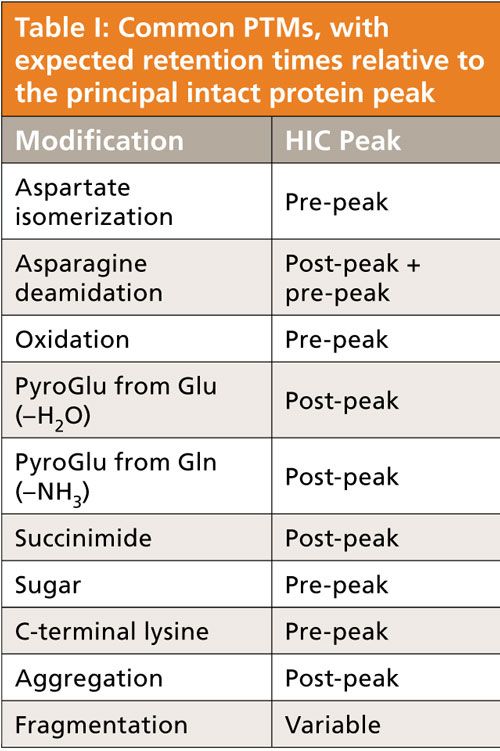
HIC can be applied to the analysis of mAb heterogeneity resulting from post-translational modifications (Table I). HIC has found particular applicability to the analysis of ADCs. ADCs contain a lipophilic cytotoxic drug attached to a monoclonal antibody; the mAb affords the selectivity while the drug provides the efficiency of treatment. Separation occurs since most cytotoxins are hydrophobic. The hydrophobicity of the ADC depends on the number of conjugated drugs; therefore, ADCs with different drug-to-antibody ratios (DAR) can be separated in HIC. In terms of elution order, mAbs that contain no drug will be eluted first and those with the highest number of drugs attached will be eluted last. The relative distribution of the ADC species can be determined on the basis of peak area percentages. The weighted DAR can then be determined based in the percentage peak area information and the drug load numbers. Because of the high salt concentrations used, HIC is generally incompatible with mass spectrometry (MS) detection (although successful studies have implemented HIC–MS analysis of intact proteins using ammonium tartrate as an MS-compatible salt in place of the more traditional ammonium sulfate). Multidimensional chromatography using a reversed-phase LC desalting step before MS analysis allows online characterization of ADCs as the ADCs dissociate into their respective subunits under the denaturing reversed-phase LC conditions.
Get the full tutorial at www.CHROMacademy.com/Essentials (free until May 20).

Separating Impurities from Oligonucleotides Using Supercritical Fluid Chromatography
February 21st 2025Supercritical fluid chromatography (SFC) has been optimized for the analysis of 5-, 10-, 15-, and 18-mer oligonucleotides (ONs) and evaluated for its effectiveness in separating impurities from ONs.









