Gas Chromatography Detection of Nitrosamines in Food and Beverages
This article discusses the detection of N-nitroso compounds (NOC) in a range of different food and beverages.
Markus Bormann /stock.adobe.com

This article discusses the detection of N-nitroso compounds (NOC) in a range of different food and beverages.
There has been a surge in media attention in recent years surrounding the potential cancerâcausing properties of a number of foods, namely processed meats, such as bacon and sausages (1). In 2015, news stories broke linking the preservatives in these foods to an increased risk in developing cancer, and the International Agency for Research on Cancer (IARC) listed processed foods as a “probable” cause of cancer. The decision was based on the findings of 22 scientists from a cohort study on the carcinogenicity of the consumption of red and processed meat (2), which suggested that certain chemicals used to preserve meats, called nitrites, can lead to the generation of N-nitroso compounds (NOC).
NOCs can be placed into two broad categories (Figure 1). The first are nitrosamines, formed from the reaction of secondary amines, which are both natural and manâmade compounds, with a nitrosating agent, such as nitrites. The second group are N-nitrosomides, which have a carbonyl group attached to the nitrogen bearing the NO group, and include N-nitrosocarbanates and N-nitrosoureas. Conditions such as pH and temperature can influence the formation of NOCs in food, as well as some drinks like beer. Some of the most frequently observed NOCs in food and drink include N-nitrosodimethylamine (NDMA) and cyclic nitrosamines, as well as nonvolatile nitrosamines (NVNA).
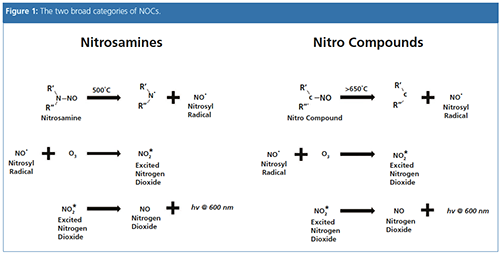
The primary sources of human exposure to NDMA are tobacco, diet (cured meats, beer, fish, and cheese), detergents, and pesticides. It can enter the body by inhaling contaminated air or ingesting contaminated food and drink, and is broken down in the liver into other substances.
Detrimental Health Effects
Many NOCs are carcinogenic and genotoxic, some are not and their potency varies depending on their molecular structure (3). NDMA is a highly potent carcinogen, commonly detected in foods and often used as an indicator compound for nitrosamines. The carcinogenic properties of nitrosamines can vary dramatically: N-nitrosodiethylamine (NDEA) is the most potent, while N-nitrosodiphenylamine (NDP(h)A) is 15,000 times less potent.
The metabolic activation of NOC produces intermediates that can react with DNA, proteins, and RNA. The carcinogenic hazard of NOC has been classified by a number of international organizations and regulatory authorities. The US Environmental Protection Agency (EPA) classifies NDMA as a “probable human carcinogen” (category 2B) and IARC classifies it as “probably carcinogenic to humans” (group 2A).
There is a large pool of research on the carcinogenic impact of nitrosamines on animals. For example, NDMA has been shown to produce liver tumours in rats when administered in drinking water (4) and in the diet (5). NDMA also acts as a transplacental carcinogen when administered to pregnant rats, and increases the incidence of tumours in the lung, liver, and kidney of rats and mice exposed via inhalation (6). Information published by the World Health Organization (WHO) identifies the cancer-causing mechanism of NDMA as the biotransformation of the compound into reactive metabolites by liver microsomal enzymes. These metabolites then form DNA adducts, such as O6âmethylguanine, as carcinogenic agents.
In August 2018, the US Food and Drug Administration (FDA) issued the voluntary recall of several drug products containing the active ingredient valsartan, after it was found that there was a contamination risk with NDMA (7). It was then later discovered that, in addition to the pharmaceutical manufacturing process, these carcinogens can be detected across other markets, such as food and beverage.
Furthermore, in March 2019, the FDA revealed Hetero Labs Ltd. in India had recalled 87 lots of losartan potassium tablets because the impurity N-Nitroso-N-methyl-4-aminobutyric acid (NMBA) had been detected. Recent testing of these recalled lots of losartan potassium tablets showed NMBA levels higher than the FDA’s interim acceptable intake limits. This is the first angiotensin II receptor blocker (ARB) recall resulting from the presence of NMBA, which is the third type of nitrosamine impurity detected in ARB medicines (8).
NOC Detection
As a result of limitations in analytical methodology, very little information is available on the levels of NVNAs and other NOCs in food and drink. However, their discovery has been greatly improved by the emergence of highly specific and sensitive gas chromatography (GC) detection, such as thermal energy analysis (TEA). TEA has long been an industry standard for nitrosamine analysis since its emergence in the late 1960s because it is able to quickly identify and analyze NOCs.
GC is used to initially separate components, and the effluent is then introduced into a pyrolyzer. Here, under vacuum, nitrosoâcontaining compounds cleave at the N-NO bonds, releasing the nitrosyl radical, which then reacts with the ozone to produce electronically excited NO2. This rapidly decays, emitting near infrared light, which is then detected by a sensitive photomultiplier.
TEA relies on selective thermal cleavage of the N–NO bond and detection of the liberated NO radical, using the chemiluminescence signal generated by its reaction with the ozone.
Food Testing
In the US, there are (nonfederal) limits for NDMA or total nitrosamines in bacon, barley malt, ham, and malt beverages, and in Switzerland there is a limit for total N-nitrosamines in beer (0.5 µg/kg). The current lack of other regulatory limits for NOC in foods in the EU spurred the initiation of a study sponsored by the Food Standards Agency, to identify and determine constituent amounts of NOC in foods formed as a direct result of the manufacturing process (9).
Premier Analytical Services (PAS) developed a method to measure and validate apparent total N-nitroso compounds (ATNC) to provide a measure of the total NOC in target foods (10). Since thermal processes could have a significant impact on NOCs, it was proposed that foodstuffs should be tested as consumed by cooking or preparing prior to analysis.
Most of the methods for determining NOC relied on the conversion of NOC to NO detected using TEA and chemical stripping (de-nitrosation). PAS wanted to validate the ATNC approach to determine how robust it was and how useful it could be in highlighting the potential risk of NOC contamination in all candidate foods and beverages. A rapid and selective (by extraction) ATNC method for screening retail foods was developed and validated with respect to the known dietary NOC of toxicological concern. During the course of the development it was noted that these methods, which rely on semi-selective chemical denitrosation reactions, can, in some instances, give false positive results and should therefore be considered as potential indicators for NOC contamination. Application of the ATNC method to target foods showed that 36/63 samples gave a positive result for ATNC.
Further analysis of these samples by ATNC showed detectable (>0.7 µg/kg) VNA contamination in only two of the 25 samples tested. These included NDMA in the dried shrimp (9.4 µg/kg, confirmed by GC–TEA) and N-nitrosopiperidine (NPIP) in the pepperoni (1.3 µg/kg), present in amounts that were consistent with historical data for these products (11).
Beer Analysis
In addition to numerous food products, the presence of NOC has been detected in beer. In the course of a malting operation, during the kilning of germinated barley, nitrosamines, namely NDMA, can form from the reaction of nitrogen oxide and the amines in the barley. Despite recent efforts to minimize the formation of nitrosamines, low levels of these carcinogens can still ultimately end up in the end product. The colour and flavour of beer is derived from malted barley, and it is also the source of sugars that are fermented into the beer. The malting process allows the barley to partially germinate, freeing the grain’s natural resources for the brewer. The production of NDMA during the malting process is a key risk for brewing laboratories. NDMA now features as part of the malt specification for many laboratories.
ATNC levels can also be used as an indicator of cleaning efficacy of vessels used for brewing, for example, in the Safe and Local Supplier Approval (SALSA) guidance notes, providing micro-breweries and bottlers with the key information required to manage their food safety management systems. Under the hazard analysis and critical control point (HACCP) and management systems section (2.1.2), the notes require the business to “identify any physical, chemical and biological hazards (including allergens) that must be prevented, eliminated or reduced to acceptable levels.” It identifies thermophilic, non-spoilage bacteria, such as Obesumbacterium proteus, as a potential cause of ATNC production (12).
GC has long been used by large breweries for quality control and quality assurance, but it has not been a financially viable technology for smaller craft breweries until the recent development of easy-to-use, affordable equipment. The surge in popularity of GC–TEA for NDMA detection, in combination with the craft brewing sector growth, has seen a rapid uptake of this technology in brewing laboratories (13).
Melamine in Milk Powder
Melamine is an organic base chemical most commonly found in the form of white crystals. Rich in nitrogen, melamine is widely used in plastics, adhesives, countertops, dishware, and whiteboards. The addition of melamine into food is not approved by the Food and Agriculture Organization of the United Nations, the World Health Organization, Codex Alimentarius (food standard commission), or by any national authorities.
WHO and the European Food Safety Authority (EFSA) have set daily exposure limits to melamine at 0.2 mg/kg body weight each day. An estimation for relevant required detection limits can be calculated-an average 6âmonthâold baby weighs 7.5 kg and consumes 0.75 litre of milk per day. Therefore, a 0.2 mg/kg exposure would equate to solution concentrations of 2 mg/L (2 ppm). TEA can be used to test milk products for the presence of melamine to protect consumers against a harmful addition to milk powder.
Other Applications
Any item containing nitrites could potentially contain nitrosamines. In addition, any process that uses secondary amines, where the manufacturing process results in an introduction of atmospheric nitrous oxide, could produce nitrosamines. Therefore, N-nitrosamines and nitrosatable substances from elastomer or rubber teats, soothers, and toys must be tested for nitrosamines. Current EU and U.S. legislation for nitrosamines recommend that baby products should contain less than 10 µg/kg of any individual and 20 µg/kg of total nitrosamines per item sold (14).
Whilst manufacturers continue to reduce the exposure risk, it is paramount to reduce babies’ and children’s exposure to these carcinogenic compounds through strict monitoring and control.
Conclusions
Recent advances in GC–TEA technology, as well as simpler usability and lower costs, have improved nitrosamine detection capabilities for both small and large laboratories. Sensitive detection for organic nitrogen makes TEA a viable option for NOC detection in food and beverages.
References
- https://www.nhs.uk/news/cancer/processed-meats-bacon-may-increase-breast-cancer-risk/
- V. Bouvard, D. Loomis, K.Z. Guyton, et al., The Lancet – Oncology 16(16), 1599–1600 (2015).
- N.E. Selin, Regulations for Nitrosamines: A Policy Summary: http://www.tcmda.com/Global/Aminrapporter/MIT%20nitrosamines%20report%20final.pdf
- H. Druckrey, R. Preussmann, S. Ivankovic, and D. Schmaehl, Z. Kerbsforsch.69(2), 103–201 (1967).
- B. Terracini, P.N. Magee, and J.M. Barnes, Br. J. Cancer.21, 559–565 (1967).
- U.S. Environmental Protection Agency (EPA) Integrated Risk Information System (IRIS) N-Nitrosodimethylamine; CASRN 62-75-9 (2002).
- Jen Christensen, 2018. CNN - FDA expands recall of blood pressure drug valsartan due to cancer concern: https://edition.cnn.com/2018/08/08/health/valsartan-recall-fda-expanded/index.html.
- Nikki Withers, 2019. FDA finds another carcinogenic impurity in ARB blood pressure drug: https://www.europeanpharmaceuticalreview.com/news/84516/carcinogenic-impurity/
- C.G. Hamlet and L. Liangi, An investigation to establish the types and levels of N-nitroso compounds (NOC) in UK consumed foods (Food Standards Agency, 2017).
- An Investigation to establish the types and levels of N-nitroso compounds (NOC) in UK consumed foods A report prepared for the Food Standards Agency: https://www.food.gov.uk/sites/default/files/media/document/an-investigation-to-establish-the-types-and-levels-of-n-nitroso-compounds-noc-in-uk-consumed-foods.pdf
- A.R. Tricker and R. Preussmann, Mutation Research/Genetic Toxicology259(3–4), 277–289 (1991).
- SALSA Guidance Notes with Specific Guidance for the Beer Module, Issue 1, June 2015, https://www.salsafood.co.uk/downloads/SALSA%20Plus%20Beer,%20Issue%202.%20Guidance%20Notes.%20January%202017.pdf
- A. James, The Column 14(11), 23–27 (2018).
- Scientific Committee on Consumer Products, SCCP - OPINION ON the Presence and Release of Nitrosamines and Nitrosatable Compounds from Rubber Balloons: https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_121.pdf
Andrew James is Marketing Director at Ellutia. Andrew has worked at Ellutia for over 20 years and has been involved with many aspects of the business from product development to strategic planning. This wide range of involvement has developed an extensive wealth of knowledge and experience in the chromatography industry. Andrew has been in charge of the company’s marketing for the last eight years, working to continually grow both the Ellutia brand and company as a whole.
E-mail:Andrew.James@ellutia.comWebsite:www.ellutia.com

Regulatory Deadlines and Supply Chain Challenges Take Center Stage in Nitrosamine Discussion
April 10th 2025During an LCGC International peer exchange, Aloka Srinivasan, Mayank Bhanti, and Amber Burch discussed the regulatory deadlines and supply chain challenges that come with nitrosamine analysis.