A Fast, Robust, and Reliable Method for Sensitively Screening Drugs of Abuse in Human Urine for Forensic Toxicology
The Column
Analyzing drugs of abuse (DoA) in human bodily fluids is crucial for clinical research and forensic toxicology. In these routine analyses, a large number of samples must be investigated, with a potentially high laboratory cost for each sample. As such, a reliable and affordable method is required for analysis. In this article, a fast, robust, and reliable method is presented for routine, high-throughput drug screening of urine samples.
Photo Credit: lokkis/Shutterstock.com

Analyzing drugs of abuse (DoA) in human bodily fluids is crucial for clinical research and forensic toxicology. In these routine analyses, a large number of samples must be investigated, with a potentially high laboratory cost for each sample. As such, a reliable and affordable method is required for analysis. In this article, a fast, robust, and reliable method is presented for routine, high-throughput drug screening of urine samples.
One of the most important requirements for analyzing drugs of abuse in human bodily fluids is that the method must be sensitive enough to selectively detect major drug groups-amphetamines, opiates, synthetic cannabinoids, and others-in one single method, and at very low levels. The challenge that modern laboratories face is that in addition to being sensitive, the process must also have a simple, costâeffective sample preparation, and a robust analytical method that is easy to implement.
For drugs of abuse, the matrix screened is usually urine, within which drugs of abuse (DoA) can be detected for approximately up to one week after use. Urine, like all human bodily fluids, is biologically complex and can vary with the metabolism and the lifestyle habits of the subjects. Consequently, drug substances and their metabolites will often be present at low levels within the sample, which can make accurate selective detection challenging. Therefore, additional sample preparation steps, such as solidâphase extraction (SPE), can be worthwhile to reduce the chemical background and to concentrate the analytes of interest. Gas chromatography–mass spectrometry (GC–MS), operated in electron ionization (EI) mode, is often used for this application because the spectra are easily searchable against existing commercial and private libraries (1).
This article presents a robust method that uses both SPE and EI in combination to produce a technique that can sensitively screen DoA in urine for use in highâthroughput and routine laboratories.
Experimental
Sample Preparation: Over 700 urine samples containing various low-level drug traces underwent an SPE procedure. A 30âμL measure of β-glucuronidase was added to 3 mL of urine and incubated for 60 min at 56 °C. A HyperSep Verify AX cartridge (Thermo Fisher Scientific), 6 mL/200 mg, was conditioned with 3 mL methanol followed by 3 mL 0.1% formic acid. Urine was mixed with 3 mL of 2 M acetate buffer at pH 4.8. Urine was checked for accurate pH. The sample was added to the cartridge and sucked through very slowly. Interference elution was performed with a mixture of 1 mL water + 0.1% formic acid, total volume 3 mL, followed by a mixture of 1 mL 50:50 methanol–water + 0.1% formic acid, total volume 3 mL. The cartridge was dried after interference elution with a strong vacuum. Elution was performed by adding a mixture of methanol and 5% ammonia solution at pH 9 with a volume of 0.5 mL. For complete elution, this step was repeated. The sample was evaporated under nitrogen until dry. It was then placed in 100-µL mass spectrometry (MS) certified vials and 50 µL of methanol added. It was subsequently centrifuged to precipitate the particles before placement in the autosampler.
The DoA excreted in urine are in the form of glucoronidate conjugates. Therefore, beta-glucuronidase was used for enzymatic hydrolysis, to cleave off glucuronides and sulphate esters prior to GC–MS analysis.
Analytical Conditions: Compound separation and detection was achieved using a Trace 1310 GC system (Thermo Fisher Scientific) coupled with an ISQ 7000 single quadrupole GC–MS system (Thermo Fisher Scientific), which features an advanced electron ionization (AEI) source. Sample introduction was performed using a TriPlus 100 LS autosampler (Thermo Fisher Scientific), injecting 1 μL on the instant connect split/splitless (SSL) injector module (Thermo Fisher Scientific).
DoA Identification: Data were acquired in full-scan mode and processed using Chromeleon Chromatography Data System (CDS) software (Thermo Fisher Scientific). The data subsequently underwent automated spectral deconvolution and peak detection, followed by library searching for putative compound identification with AnalyzerPro software (SpectralWorks).
Results and Discussion
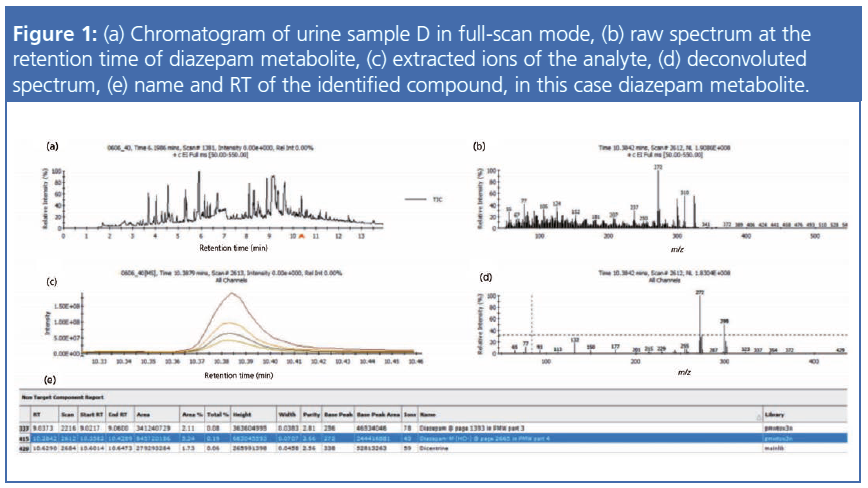
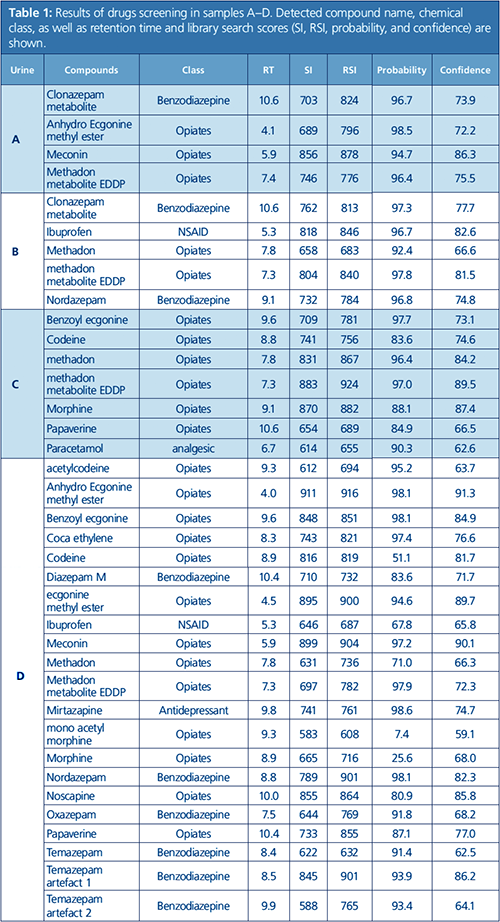
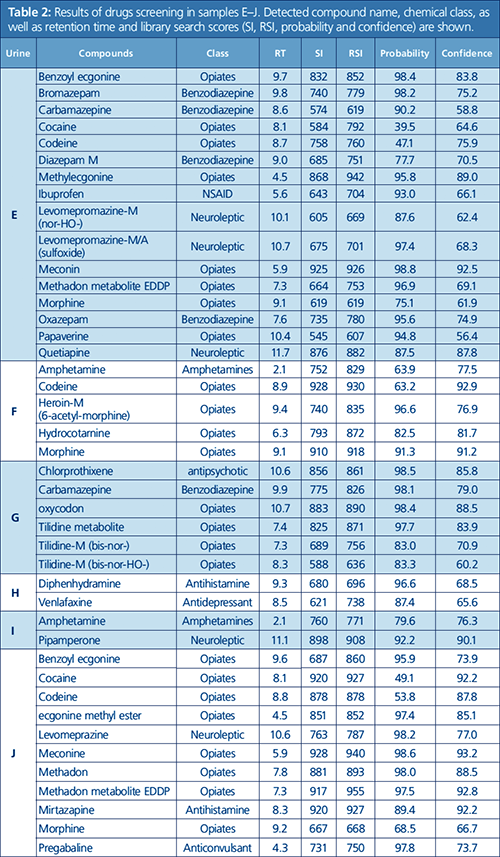
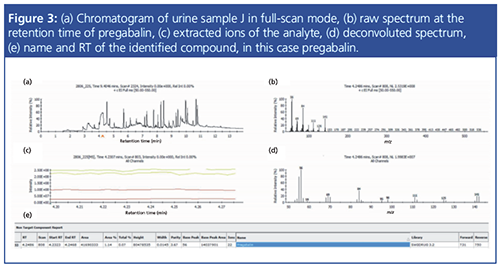
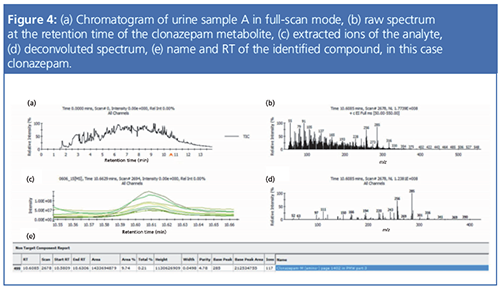
In excess of 700 urine samples were analyzed as part of this study. Representative results were selected to show the capacity of this method to detect and quantify challenging drug metabolites in urine, as well as some typical urine sample profiles obtained from drug addicts. Ten urine samples (A to J) were found to be the most interesting, and data for these samples are summarized in Tables 1 and 2. Figures 1 to 4 provide detailed views of some of the more challenging metabolites. One of the examples shown is pregabalin, a drug that can be prescribed for the treatment of conditions such as neuropathic pain and fibromyalgia. However, when consumed in larger quantities this drug has similar effects to classic drugs of abuse. There is a recent trend in Germany of pregabalin prescriptions being sought out by drug abusers. The other examples provided are benzodiazepines, a class of drugs that undergoes significant metabolism and is difficult to analyze at low levels. Even so, the metabolites are identified relatively easy.
The results collected during this study highlight the potential this method holds in the detection of DoA in urine for criminal and forensic investigations and clinical research. The developed methods are not only applicable to classic drug compounds, such as heroine and amphetamines, but also suited for the detection of new psychoactive substances such as fentanyl, the NBOMe family of drugs, and various Spice-type drugs.
Observable trends can be revealed from the analyses of the subjects’ urine profiles. For instance, urine sample D shows a rapid ingestion of all available drugs before the start of the forensic investigation; whereas urine sample J could present a subject going to multiple doctors, attempting to get a prescription for pregabalin, which in sufficient quantities can give a feeling of euphoria. Sample H shows a person taking medication to cope with withdrawal symptoms. Interestingly, in almost all the urine samples, markers for smoking, such as cotinine and nicotine, were found, which could be a reflection of smoking as a coping mechanism for withdrawal (2).

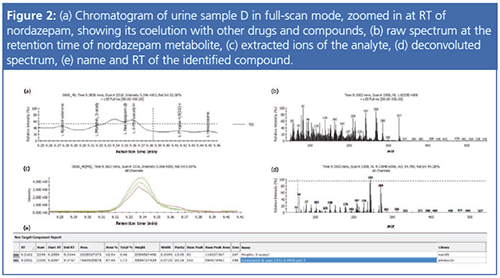
The data demonstrate that the method presented provided a rapid and robust method for the general unknown screening of drugs of abuse. This method can be used for routine analysis of DoA in runs with a sample throughput of more than 12,000 samples a year.
The concentration factor of 60 gained through sample preparation, and the improved sensitivity of the ion source, made it possible to reach low detection limits. These low detection limits are much more sensitive than the results obtained in immunological testing, which could be especially beneficial for analysis of new psychoactive substances. This would also enhance the integrity of the data by reducing the variability that is commonly seen in immunoassays.
The data provide reliable, libraryâsearchable spectra and can indicate which group of drugs the new compound belongs. It is possible by fragmentation rules to see to which group the analyte belongs, without a complex collection of drugs. When hundreds of substances show up in the chromatogram, chromatographic deconvolution is an incredibly useful tool for this kind of analysis. In contrast, a manual data investigation would require a highly experienced scientist and would be a lengthy and laborious process.
These complete methodologies provided a rapid, reliable, and robust method of screening for drugs of abuse in urine samples in routine and high-throughput laboratories.
References
- C.A. Hammett-Stabler, A.J. Pesce, and D.J. Cannon, Clin. Chim. Acta315, 125–135 (2002).
- J.J. Prochaska, K.H. Delucchi, and M. Sharon, J. Consult. Clin. Psychol.72, 1144–1156 (2004).
- For Forensic Use Only.
Luzia Schaaf is a research pharmacist at the LVR Klinik Viersen, Germany, and is interested in the analysis of new narcotic drugs.
Petra Gerhards is EMEA regional marketing manager for CCS at Thermo Fisher Scientific, Dreieich, Germany.
Inge de Dobbeleer is EMEA regional marketing manager for GC and GC–MS at Thermo Fisher Scientific, Breda, The Netherlands.
E-mail:inge.dedobbeleer@thermofisher.comWebsite: www.thermofisher.com

University of Rouen-Normandy Scientists Explore Eco-Friendly Sampling Approach for GC-HRMS
April 17th 2025Root exudates—substances secreted by living plant roots—are challenging to sample, as they are typically extracted using artificial devices and can vary widely in both quantity and composition across plant species.
Miniaturized GC–MS Method for BVOC Analysis of Spanish Trees
April 16th 2025University of Valladolid scientists used a miniaturized method for analyzing biogenic volatile organic compounds (BVOCs) emitted by tree species, using headspace solid-phase microextraction coupled with gas chromatography and quadrupole time-of-flight mass spectrometry (HS-SPME-GC–QTOF-MS) has been developed.
Fundamentals of Benchtop GC–MS Data Analysis and Terminology
April 5th 2025In this installment, we will review the fundamental terminology and data analysis principles in benchtop GC–MS. We will compare the three modes of analysis—full scan, extracted ion chromatograms, and selected ion monitoring—and see how each is used for quantitative and quantitative analysis.
Quantifying Microplastics in Meconium Samples Using Pyrolysis–GC-MS
March 26th 2025Using pyrolysis-gas chromatography and mass spectrometry, scientists from Fudan University and the Putuo District Center for Disease Control and Prevention detected and quantified microplastics in newborn stool samples.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)