Enhancing the Sensitivity of Atmospheric Pressure Ionization Mass Spectrometry Using Flow Modulated Gas Chromatography
The Column
Peak intensity enhancement is one highly desirable outcome of comprehensive two-dimensional gas chromatography (GC×GC). When coupled to mass spectrometry (MS), such enhancement is usually achieved with a thermal modulator using a technique called cryogenic zone compression (CZC). Differential flow modulation is a simple and cost-effective alternative to thermal modulation, but the requisite high flow rates are generally perceived as being incompatible with most (electron ionization [EI] and chemical ionization [CI]) mass spectrometers. The past decade has witnessed resurgent interest in coupling GC to atmospheric pressure chemical ionization (APCI), which requires high gas flows to assist ionization. This article reports on the modification of a GC–APCI system with a flow modulator and evaluates its potential to enhance the sensitivity towards selected trace organics.
Photo Credit: Dee Browning/Shutterstock.com

Peak intensity enhancement is one highly desirable outcome of comprehensive two-dimensional gas chromatography (GC×GC). When coupled to mass spectrometry (MS), such enhancement is usually achieved with a thermal modulator using a technique called cryogenic zone compression (CZC). Differential flow modulation is a simple and cost-effective alternative to thermal modulation, but the requisite high flow rates are generally perceived as being incompatible with most (electron ionization [EI] and chemical ionization [CI]) mass spectrometers. The past decade has witnessed resurgent interest in coupling GC to atmospheric pressure chemical ionization (APCI), which requires high gas flows to assist ionization. This article reports on the modification of a GC–APCI system with a flow modulator and evaluates its potential to enhance the sensitivity towards selected trace organics.
The exposome represents all environmental exposures over the course of a lifetime (1). The identities of most environmental contaminants and their roles in causing chronic diseases are unknown. This is partly a result of the analytical challenges associated with biomonitoring trace levels of environmental toxicants in small sample quantities of serum or tissue. It is estimated that standard gas chromatography–mass spectrometry (GC–MS) and liquid chromatography (LC)–MS platforms are not sufficiently sensitive to identify approximately 90% of unknown contaminants (2).
Inspired by advances in comprehensive twoâdimensional gas chromatography (GC×GC) (3), Patterson and co-workers elegantly showed that (targeted) subâfemtogram detection of environmental pollutants is achievable using a technique termed cryogenic zone compression (CZC) (4). With CZC, the 3–8 s width of a GC peak is compressed to approximately 200 ms, resulting in a significant enhancement in signal intensity (4–6). This has opened the door to the possibility of monitoring environmental toxicants using less invasive and inexpensive sampling techniques, such as collection of dried blood spots (7). The cost of requisite hardware however, viz. a thermal modulator, and associated cryogens and refrigeration is a drawback.
Signal enhancement is also possible using a valve-based flow modulator, a technology pioneered by J.V. Seeley (Oakland University, Michigan, USA) (8,9). In contrast to thermal modulation, a flow modulator requires a high carrier gas flow (>10 mL/min) to transfer the column effluent from the modulator into the mass spectrometer. The potential of flow modulation has not been fully realized because of the perception that high column flows are not compatible with most (electron ionization [EI] and chemical ionization [CI]) mass spectrometers. This is usually addressed by effluent splitting at the cost of sensitivity. Improved pumping capacity of modern turbomolecular pumps can mitigate the problem, but still this hyphenation is generally considered difficult (10,11).
GC coupled to atmospheric pressure chemical ionization (APCI) (12–16) is by contrast ideally suited to high flows. In GC–APCI, the GC effluent is swept into the ion source, which is held at atmospheric pressure, by a high flow of nitrogen gas (>100 mL/min). There is also the additional benefit that the elevated pressures promote collisional cooling of the incipient ions. Consequently, very little fragmentation occurs, resulting in further signal enhancement of the molecular ion. With widespread adoption of atmospheric pressure ionization techniques, such as electrospray ionization (ESI) and APCI, there is growing interest in coupling GC–MS using atmospheric pressure ionization. This article reports on proofâofâconcept experiments using a simple, cost-effective flow modulator (17) coupled to a quadrupole time-of-flight mass spectrometer (QTOFâMS). The results indicate that environmental contaminants can be detected at low femtogram levels using a full-scanning instrument capable of nontargeted analysis.
Experimental
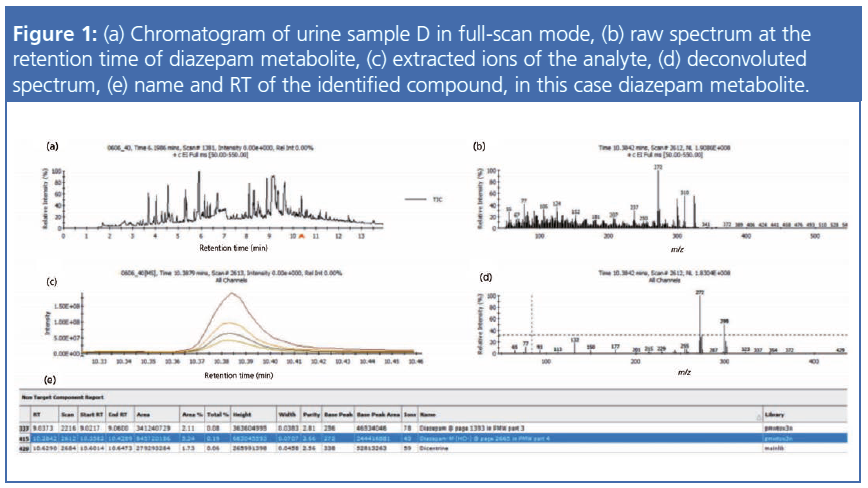
The experiments were performed using a Xevo G2-XS quadrupole time-of-flight mass spectrometer (Waters) coupled to a modified 7890B gas chromatograph (18,19) (Agilent). In GC–APCI, the GC effluent is swept into the ion source by a 350 mL/min make-up flow (N2). A schematic of GC–APCI is shown in Figure 1(a).
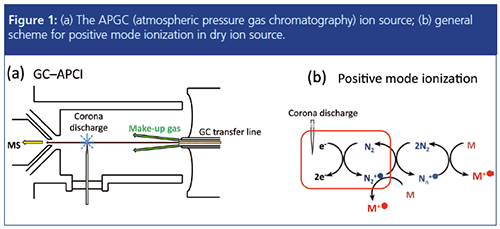
Ions formed by a corona discharge needle set to 3 µA were transferred into the mass spectrometer, aided by 20 V potential and cone and auxiliary gas flows of 175 L/h and 100 L/h, respectively. In the charge transfer mode (Figure 1[b]), ionization was typically initiated by the formation of N2•+ and Nn•+ cluster ions, which may undergo charge exchange with analyte molecules (M) provided the ionization energy (IE) of M is lower than the recombination energy (RE) of N2•+. Non-reactive collisions between the incipient ions and the surrounding N2 results in collisional cooling that significantly reduces fragmentation and therefore also the detection limits for the molecular ions (M•+). Other ionization schemes are also possible by introducing dopants to the ion source. For example, H2O can be used to promote protonation. O2•- is the primary charge carrier in negative ion mode APCI, which can result in selective ionization and structure diagnostic fragmentation of halogenated compounds (18,20). The technique of ionizing a sample of molecules by gas-phase ion-molecule reactions is well studied (20), and since its inception an enormous number of studies have appeared on its applications in analytical MS.
A ZX2 thermal modulator (Zoex) was used for CZC experiments. A multimode modulator (17) designed and constructed by J.V. Seeley was useds to explore the potential of coupling flow modulation to GC–APCI. The construction, operation, and performance of this device has been described recently (17). Briefly, the multimode modulator (17) of this study was constructed around a 1/8” outside diameter (o.d.) liner (standard PTV injector liner) (Figure 2).

A piece of tubing or capillary can also be used for primary effluent collection, but the 1/8” o.d. liner provided the volume required (~200 µL) to accommodate a 4 s modulation period and 3 mL/min carrier gas flow. One end of the joining liner was attached to a cross union, or as shown in Figure 2, a modified tee. The exit of the primary column (15 m × 0.25 mm, 0.1-µm db-5 [Agilent Technologies]) was inserted through the cross and then into the joining liner so that the column exit sat approximately 1 cm from the gooseneck of the liner. The opposite end of the liner was attached to a tee union. A 0.5 m × 0.53 mm Rxi Guard column (Restek) that served as the transfer line to the MS was inserted into the tee and butted against the gooseneck. Auxiliary flow (125 mL/min) was delivered to the tee and cross unions via a two-way, three-port solenoid valve (not shown). A flow restrictor was connected to the cross connection. The operation of the multimode modulator for the purpose of signal enhancement is described below.
Results and Discussion
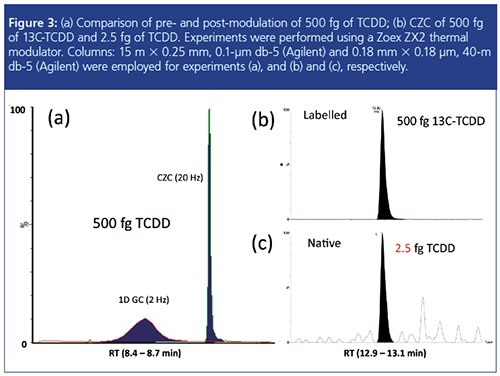
CZC relies on the same principles of operation as GC×GC, whereby a pulse of cooled nitrogen gas is used to condense and focus GC effluent (primary peak), followed by a pulse of heated gas for re-injection (secondary peak). The timing of the two pulses (modulation period) is configured so that the width of a GC peak is compressed. This is illustrated in Figure 3(a), which displays a ~3 s wide peak prior to modulation and a narrow (250 ms wide) peak post modulation. The 10âfold enhancement in signal intensity (Figure 3[a]) enables low femtogram detection of 2378âTCDD (tetrachlorodibenzo-p-dioxin) using QTOF, see Figure 3(c). Note that the identity of the native compound (Figure 3[c]) is confirmed by comparison with the corresponding 13C-labelled compound (Figure 3[b]).
A flow modulator does not condense or focus the GC effluent. Instead, the effluent is trapped in a sample loop of sufficient volume to accommodate the primary flow. The width of the secondary peak can still be controlled by the secondary flow. Figures 4(a) and 4(b) show the flow modulator of this study in the collection and injection states, respectively. In the collection state, effluent from the primary column (3 mL/min) backfills into the liner as primary carrier gas escapes via the flow restrictor. In the injection state (Figure 2[c]), the secondary flow (125 mL/min) is diverted through the sample loop, flushing the accumulated contents into the mass spectrometer.

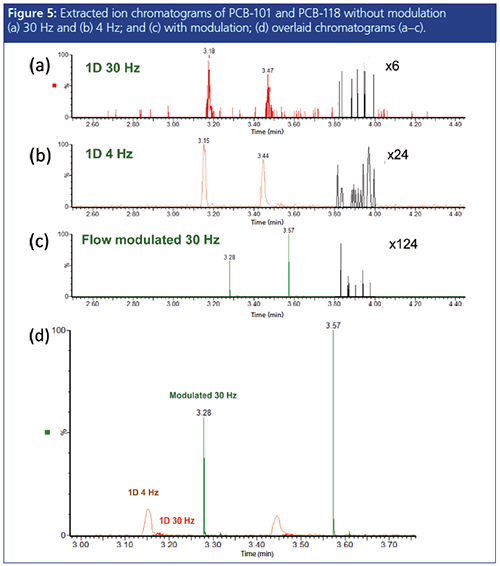
From the ratio of the primary and secondary flows, one anticipates a signal enhancement of approximately 40×. As shown in Figure 5(a), the signal-to-noise ratios (S/N) of two polychlorinated biphenyls (101 and 118) are approximately 6:1. When the modulator is activated, this S/N increases by a factor of 20 to 124:1 (see Figure 5[c]). Note that the acquisition time of the mass spectrometer is normally set to acquire ~10 data points across the peak. As shown in Figure 5(b), the S/N of the primary peaks increases to 24:1 when the acquisition rate is adjusted from 30 Hz to 4 Hz. Still, signal enhancement of approximately 10:1 is evident from the overlaid chromatograms in Figure 5(d).
Summary
A flow modulator was constructed and evaluated using QTOF-MS. Like CZC, flow modulation produces sharp peaks (<200 ms wide) with a significant improvement in signal intensity compared to unmodulated peaks. Aside from the obvious implications to quantitative analysis, the enhanced sensitivity also helps address the critical need for nontargeted identification of unknown environmental toxicants. The analytical platform described in this study is capable of acquiring high quality, full scan mass spectra of femtogram level toxicants and as such may be a promising tool in environmental monitoring, biomonitoring, and exposomic research (20).
References
- C.P. Wild, International Journal of Epidemiology41, 24 (2012).
- S.M. Rappaport, D.K. Barupal, D. Wishart, P. Vineis, and A. Scalbert, Environmental Health Perspectives122, 769 (2014).
- L. Ramos, Ed., Comprehensive Two Dimensional Gas Chromatography (Elsevier, Amsterdam, Netherlands, 2009).
- D.G. Patterson Jr., S.M. Welch, W.E. Turner, A. Sjodin, and J-F. Focant, J. Chrom. A1218, 3274 (2011).
- B. L’Homme and J.-F. Focant, Analytical Methods 8, 249 (2016).
- C. Brasseur, B. L’Homme, and J.-F. Focant, Organohalogen Compounds74, 55 (2012).
- R. Guthrie and A. Susi, Pediatrics32, 338 (1963).
- J.V. Seeley, N.J. Micyus, J.D. McCurry, and S.K. Seeley, American Laboratory 38, 24 (2006).
- J.V. Seeley, J. Chrom. A1255, 24 (2012).
- R. Shellie, LCGC Europe21(11), (2008).
- M. Poliak, M. Kochman, and A. Amirav, J. Chrom. A1186, 189 (2008).
- E.C. Horning, M.G. Horning, D.I. Carroll, I. Dzidic, and R.N. Stillwell, Anal. Chem45, 936 (1973).
- D-X. Li, L. Gan, A. Bronja, and O.J. Schmitz, Anal. Chim. Acta891, 43 (2015).
- A. Ballesteros-Gomez, J. de Boer, and P.E.G. Leonards, Anal. Chem. 85, 9572 (2013).
- C.N. McEwen and R.G. McKay, J. Am. Soc. Mass Spectrom.16, 1730 (2005).
- C.N. McEwen, R.G. McKay, and B.S. Larsen, Anal. Chem. 77, 7826 (2005).
- J.V. Seeley, N.E. Schimmel, and S.K. Seeley, J. Chrom. A1536, 6–15 (2018).
- S. Fernando, M.K. Green, K. Organtini, F. Dorman, R. Jones, E.J. Reiner, and K.J. Jobst, Anal. Chem. 88, 5205 (2016).
- D. Megson, M. Robson, K.J. Jobst, P.A. Helm, and E.J. Reiner, Anal. Chem.88, 11406–11411 (2016).
- A.G. Harris, Chemical Ionization Mass Spectrometry (CRC Press, Boca Raton, USA, 1983).
- J.R. Sobus, J.F. Wambaugh, K.K. Isaacs, A.J. Williams, A.D. McEachran, A.M. Richard, C.M. Grulke, E.M. Ulrich, J.E. Rager, M.J. Strynar, and S.R. Newton, J. Exposure Sci. Environ. Epidemiol. (2017) DOI: 10.1038/s41370-017-0012-y.
Karl Jobst is a development scientist with the Ontario Ministry of the Environment and Climate Change (MOECC). His research interests focus on the development and application of mass spectrometric techniques for the analysis of complex environmental samples.
John Seeley is a professor of chemistry at Oakland University in Rochester, Michigan, USA. For the past 20 years, his research has largely focused on the development of flow modulation GC×GC and its application to separations of complex mixtures of volatile organic compounds.
Eric Reiner is an adjunct professor at the University of Toronto and emeritus mass spectrometry research scientist at MOECC. He has spent over 30 years at MOECC working on the development of analytical methods for the analysis of halogenated persistent organic pollutants (POPs).
Lauren Mullin is a principal scientist at Waters Corporation. Her work focuses on the development of liquid and gas chromatography–mass spectrometry methods to address food, environmental, and various small molecule research analyses.
Adam Ladak is a senior strategic scientific marketing manager at Waters Corporation. He is responsible for food and environmental market development worldwide with a focus on environmental research.
E-mail:Adam_Ladak@waters.comWebsite:www.waters.com

Determining the Effects of ‘Quantitative Marinating’ on Crayfish Meat with HS-GC-IMS
April 30th 2025A novel method called quantitative marinating (QM) was developed to reduce industrial waste during the processing of crayfish meat, with the taste, flavor, and aroma of crayfish meat processed by various techniques investigated. Headspace-gas chromatography-ion mobility spectrometry (HS-GC-IMS) was used to determine volatile compounds of meat examined.
Accelerating Monoclonal Antibody Quality Control: The Role of LC–MS in Upstream Bioprocessing
This study highlights the promising potential of LC–MS as a powerful tool for mAb quality control within the context of upstream processing.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)