Determining Zeranol in Bovine Tissues Under Nontoxic Conditions
LCGC North America
The authors have developed a method of determining zeranol residues in bovine tissues without using toxic chemicals, organic solvents, and reagents in sample preparation and reversed-phase HPLC separation.
The authors have developed a method of determining zeranol residues in bovine tissues (muscle, kidney, liver, and fat) without using toxic chemicals, organic solvents, and reagents in sample preparation and reversed-phase high performance liquid chromatography (HPLC) separation. Isolation is achieved by homogenization using a handheld ultrasonic homogenizer, which is easy to use and portable, followed by solid-phase extraction with an anion exchanger. For determination and identification of zeranol, the HPLC method uses an isocratic mobile phase and a photodiode-array detector. The authors obtained average recoveries of zeranol > 84% with relative standard deviations around 3.0%–4.8% in spiked samples. The total analytical time and quantitation limit were < 40 min/sample and 0.04 μg/g, respectively.

Zeranol is one of the Food and Drug Administration (FDA)-approved growth hormones used for fattening cattle and is the most-used synthetic growth hormone in beef production in the United States and other countries. However, the growth hormones are banned in other countries, including all the member states of the European Union. In the U.S., zeranol resiudues of 0.15, 0.3, 0.45, and 0.6 μg/g in bovine muscle, liver, kidney, and fat, respectively, are considered safe. FDA has set the previous concentrations as "the maximum safe tissue residue levels" for the routine residue monitoring of zeranol (1,2). Beef is an important source of nutrition in human living. A rigid residue monitoring of zeranol in beef, therefore, is an important specific activity to guarantee food safety. The acceptable analytical method for the routine monitoring of zeranol residues in beef must be reliable, economical in cost and time, capable of determining residues below the safe residue level, and nontoxic to the environment and analysts.
In recent years, there have been significant developments in techniques for determining residues of zeranol in bovine tissues (3–7). However, all of these methods require the use of toxic organic solvents, such as acetonitrile, chloroform, diethyl ether, and methanol, or toxic reagents, such as metaphosphoric acid (8,9), which are directly toxic to both humans and the environment. The risks associated with these chemicals extend beyond the direct implications for the health of humans and wildlife to affect our environment and the ecosystem (10–14). For most methods, the sample preparation involves several time-consuming and expensive analytical steps. Presently, there is no desired method for routinely monitoring zeranol residue in bovine tissues.
The determining technique described here is an economic method that enables a rapid and inexpensive determination of zeranol in bovine tissues without the use of toxic chemicals, organic solvents, and reagents in sample preparation and HPLC.
Experimental
Materials: Zeranol and other chemicals were obtained from Wako Pure Chemicals International, Osaka, Japan. Anhydrous sodium sulfate was of pesticide residue grade. Distilled water, n-heptane, and ethanol were of HPLC grade. As solid-phase extraction (SPE) syringe-columns, three anion-exchange silica-based sorbent types (sorbent mass: 500 mg; reservoir 10 mL) of Bond Elut LRC series, Bond Elut LRC-NH2 (aminopropyl), -SAX (quaternary amine), and -DEA (diethylaminopropyl) (Varian, Inc., Palo Alto, California) were used. These SPE columns were prepacked with 0.5 g of anhydrous sodium sulfate to remove the negligible moisture in heptane or ethanol as the eluent. The columns were preconditioned by washing with 5 mL of heptane.
Six types of C1 or C4 nonpolar sorbent columns (3- or 5-μm dp) (150 mm X 4.6 mm) with their guard columns (5 mm X 4.6 mm) for HPLC analysis were used: column A, Daisopak SP-200-3-C1-P (Daiso Co., Ltd., Osaka, Japan); column B, Kaseisorb LC C1-100-5 (Tokyo Kasei Kogyo Co., Ltd., Tokyo, Japan); column C, Wakosil 5TMS (Wako); column D, Develosil TMS-5 (Nomura Chemical Co., Ltd., Aichi, Japan); column E, Develosil TMS-3 (Nomura Chemical); column F, Mightysil RP-4 (Kanto Chemical Co., Inc., Tokyo, Japan).
Sample preparation: Extraction: Bovine muscle, kidney, and liver were minced fully and used for blank samples. An accurately weighed 0.5-g sample was placed in a 10-mL centrifuge tube and mixed sufficiently with 2 g of anhydrous sodium sulfate. The mixture was homogenized with a handheld ultrasonic homogenizer (model HOM-100, Iwaki Glass Co., Ltd., Funabashi, Japan) for 30 s with 5 mL of heptane. After the homogenization step, the capped tube was centrifuged at 3000g for 5 min. The supernatant liquid was applied to an SPE syringe-column. Bovine fat was premelted at 50 °C and used for the blank sample. An accurately weighed 0.5-g sample was dissolved in 1 mL of heptane and was directly applied to an SPE syringe-column.
Cleanup using an anion-exchange SPE syringe-column: After the application to the Bond Elut LRC-DEA column, the barrel was washed with 5 mL of heptane, and then zeranol was eluted with 5 mL of 10:90 (v/v) ethanol–heptane (flow rate < 5 mL/min). The eluate was evaporated to dryness, and the residue was dissolved in 1 mL of the HPLC mobile phase. The solution was filtered through a 0.45-μm disposable syringe filter unit (hydrophilic cellulose acetate membrane, Advantec, Toyo Roshi Co. Ltd., Tokyo, Japan). The filtrate was injected into an HPLC system.
HPLC: The HPLC system included a model PU-980 pump and a model DG-980-50 degasser (Jasco Corp., Tokyo, Japan) equipped with a model CO-8010 column oven (Tosoh Corp., Tokyo, Japan) and a model SPD-M10A VP photodiode array (PDA) detector (Shimadzu, Kyoto, Japan).
The operating conditions were as follows: the analytical column was a 150 mm X 4.6 mm Mightysil RP-4 (endcapped, 4.3-μm dp, 361 m2/g surface area, 119-Å pore diameter, 1.1-mL/g pore volume, 4.7% carbon content, Kanto Chemical Co., Inc., Tokyo, Japan) equipped with a guard column (5 mm X 4.6 mm) containing the same packing material; the isocratic mobile phase consisted of 35:65 (v/v) ethanol–0.01 M citric acid; the pump flow rate was 1.0 mL/min; the column temperature was 40 °C; the injection volume was 20 μL; and the analysis time was < 7 min. The absorption spectrum of a zeranol standard solution was measured by a photodiode-array detector, and the obtained maximum absorption wavelength was 222 nm. The monitoring wavelength was adjusted to 222 nm. The peak identification of zeranol was established by comparing the retention time and its absorption spectrum in real samples with that obtained by injection of the standard.
Results and Discussion
One goal of this work was to develop a technique for determining zeranol in bovine tissues without the use of any toxic organic solvents and reagents. The environmental and human toxicity of ethanol, heptane, and citric acid used here are negligible (8,9).
Sample preparation: The extraction operation used a portable handheld ultrasonic homogenizer and effected extraction of zeranol from a small bovine tissue sample in a small volume of heptane. The extract did not form an emulsion.
Anion-exchange SPE-NH2 (6) or –DEA (15) cartridges have been used in a valid cleanup technique of zeranol, an acidic growth hormone, in extracts from beef or bovine liver. The authors used three types of Bond Elut LRC anion-exchange SPE syringe-columns for the further cleanup technique. As a preliminary study, a 50:50 (v/v) mixture of ethanol and heptane was used as the eluent and the recoveries of zeranol from these columns were compared. Bond Elut LRC-DEA and -SAX gave a satisfactory recovery (> 96%) and reproducibility (RSD = 2%, n = 3) for zeranol when the elution volume was standardized at 5 mL. The recovery from Bond Elut LRC-NH2 was 64% with a relative standard deviation (RSD) of 4% (n = 3).

Table I: Effect of the ethanol concentration in the eluent on the recovery of zeranol from selected anion-exchange SPE columns*
Table I presents the effects of an ethanol concentration in the eluent (ethanol–heptane, v/v) on the recovery of zeranol from the selected two SPE syringe-columns when the elution volume was standardized at 5 mL. The zeranol in 5 mL of each collected fraction was determined by HPLC. The better eluents that recovered zeranol from the DEA and SAX sorbents were a 10% (v/v) solution of ethanol in heptane and a 20% (v/v) solution of ethanol in heptane, respectively. As can be seen in Table II, the elution volume required for zeranol was 5 mL. Because the extracts that were treated by the SAX column showed significant chromatographic interferences that hindered the confirmation of zeranol, the DEA column was used in this study.
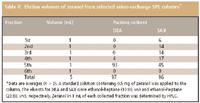
Table II: Elution volumes of zeranol from selected anion-exchange SPE columns*
HPLC operating conditions: The C18 nonpolar sorbent column is used most frequently for reversed-phase HPLC analysis for veterinary drugs including zeranol. All of these analyses require larger volumes of acetonitrile and methanol, toxic organic solvents, as the elution solvent. Of the packed nonpolar sorbents in the column for reversed-phase HPLC separation, the C1 and C4 sorbents are less retentive than C18 sorbent when retention is based upon nonpolar interactions alone. To reduce the volume of elution solvent required and provide a more rapid separation, we tested six columns in total — five types of the C1 sorbent columns and one type of the C4 sorbent column. The physical–chemical specifications are listed in Table III. We used a mixture of ethanol and 0.01 M citric acid as the mobile phase. Citric acid is also a nontoxic reagent. We examine mobile phases with the ethanol concentrations of 10%–40% (v/v), column temperatures ≥ 25 °C, and HPLC follow rates ≥≥0.5 mL/min. We compared the six columns with regard to how the analysis separated the interfering peaks and the sharpness of the zeranol peak obtained upon injection of equal amounts. The observed chromatographic zeranol peaks formed within the condition ranges examined also are presented in Table III.

Table III: Physical/chemical specifications of the C1 and C4 columns* used and chromatographic zeranol peak forms obtained under the HPLC condition ranges examined.
Columns A and B provided significantly weakened retention of zeranol throughout the condition ranges. They had difficulty separating zeranol and the interference of the resulting bovine tissue extract and preventing the tailing peak. Columns C, D, and E separated zeranol as a significant leading peak, rounded peak, and broadening peak, respectively. An ideal chromatogram with complete separation of zeranol, a symmetrical, sharp zeranol peak and a short retention time, was obtained by using column F and 35:65 (v/v) ethanol–0.01 M citric acid with a flow rate of 1.0 mL/min and a column temperature of 40 °C. The low-pH environment of the citric acid mobile phase used here was needed to obtain these findings. From the data shown in Table III, it is difficult to prove the critical parameter in the column with regard to the retention of zeranol and its peak form.
Figure 1 displays typical chromatograms for a spiked bovine liver sample and for a blank bovine liver sample obtained under the procedure developed here, with the photodiode-array detector set at 222 nm (giving maximum absorption spectrum for zeranol). This figure demonstrates that the present method can reliably provide a quantitation and identification of zeranol. Similar chromatograms were obtained from the muscle, kidney, and fat samples. The present method of HPLC with photodiode-array detection easily confirmed the peak identity of the target compound. Zeranol was identified in the sample by its retention time and absorption spectrum. In the HPLC system, separation, identification, and quantification were achieved in a mere 7-min run without requiring a gradient system to improve the separation and precolumn washing after an analysis. Because the HPLC separations were performed serially, the time per run became critical in routine residue analysis. The short run time not only increased sample throughput for analysis but also affected the method-development time.
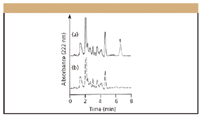
Figure 1: Chromatograms obtained from HPLC analyses of (a) a bovine liver sample spiked with 0.3 μg/g zeranol and (b) a blank bovine liver sample. Peak 1 = zeranol (retention time = 6.6 min).
The authors established a toxic, chemical-free method for determining zeranol in bovine tissues, one that is nontoxic to the environment and humans. The procedure has a minimal number of steps and uses a small sample size, resulting in rapidness and simplicity with considerable savings in analytical cost. The total time and nontoxic solvent consumption required for the analysis of one sample were < 40 min and < 20 mL (heptane and ethanol), respectively.
Method qualification: Table IV summarizes the analytical performance parameters (that is, linearity, precision, accuracy, and sensitivity) of the complete method. The calibration curve was generated by plotting the peak area of fortified sample extracts in the 0.05–1 μg/g range for zeranol. The resulting correlation coefficient in bovine muscle, liver, kidney, fat samples were ≥ ≥0.997 (P < 0.01). The accuracy and precision were expressed as the average recoveries from spiked bovine tissue samples and their RSDs. The recoveries and their RSDs were ≥ 84% and ≤ 4.8%, respectively. We calculated the quantitation limit by analyzing control bovine tissue samples (free zeranol); the samples were spiked with concentrations of zeranol. The quantitation limit should correspond to the concentration for which the signal-to-noise ratio is > 10. The limit was 0.04 μg/g for zeranol in all bovine tissue samples, which was considerably less than the maximum safety tissue residue levels, as described previously.
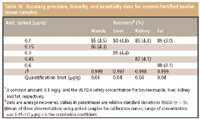
Table IV: Accuracy, precision, linearity, and sensitivity data for zeranol-fortified bovine tissue samples
Monitoring Residue in Marketed Beef
A total of 33 different beef samples that were available in Osaka, Japan were analyzed by the present method. No samples contained detectable concentrations of zeranol. The resulting chromatograms were free from interference.
Conclusion
The proposed sample preparation technique followed by HPLC is useful as a routine method for residue monitoring of zeranol in bovine muscle, liver, kidney, and fat samples and has the following advantages: The sample preparation by means of the homogenization using a handheld ultrasonic homogenizer followed by an anion-exchange SPE column provides an easy, rapid, effective, and economical isolation of zeranol from the sample. The procedure uses no toxic chemicals (organic solvents and reagents) and has a total analytical time of less than 40 min/sample and nontoxic organic solvent consumption of less than 20 mL/sample.
Naoto Furusawa and Kunihiro Kishida Faculty of Food and Nutrition, Kyushu Nutrition Welfare University, Fukuoka 803-8511, Japan Graduate School of Human Life Science, Osaka City University, Osaka 558-8585, Japan. Please direct correspondence to Nao to Furusawa at furusawa@life.osaka-cu.ac.jp
References
(1) U.S. Meat Export Federation. http://www.usmef.org/
(2) Center for Veterinary Medicine, Summary of NADA 038-233: RALGRO (zeranol), Food and Administration, 1995. http://www.fda.gov/cvm/
(3) J. Jodlbauer, P. Zollner, and W. Lindner, Chromatographia 51, 681–687 (2000).
(4) .M. Fang, J.H. Chen, and D.H. Guo, J. AOAC Int. 85, 841–847 (2002).
(5) J. Seo, H.Y. Kim, B.C. Chung, and J.K. Hong, J. Chromatogr. A 1067, 303–309 (2005).
(6) S. Taguchi, S. Yoshida, Y. Tanaka, and S. Hori, J. Food Hyg. Soc. Jpn. 42, 226–230 (2001).
(7) M. Horie and H. Nakazawa, J. Chromatogr. A 882, 53–62 (2000).
(8) Merck Catalogue 2002–2003 Merck KGaA, Darmstadt, Germany, 2002
(9) Chemicals 32nd Edition, Wako Pure Chemical Industries, Ltd., Japan, 2002.
(10) M. Ishibashi, J. Food Hyg. Soc. Jpn. 38, J193 (1997).
(11) M. Ishibashi, J. Food Hyg. Soc. Jpn. 38, J194–195 (1997).
(12) M. Ogawa, J. Food Hyg. Soc. Jpn. 37, J289–290 (1996).
(13) P.T. Anastas and J.C. Warner, Green Chemistry: Theory and Practice, P.T. Anastas and J.C. Warner, Eds. (Oxford University Press, Oxford, United Kingdom, 1998).
(14) T. Yoshimura, T. Nishinomiya, Y. Honma, and M. Murabayashi, Green Chemistry — Aim for the Zero Emission-Chemicals (Sankyo Publishing Co. Ltd. Press, Tokyo, Japan, 2001).
(15) T. Miyazaki, T. Sasamoto, T. Hashimoto, and Y. Kokubo, J. Food Hyg. Soc. Jpn. 36, 748–753 (1995).

Thermodynamic Insights into Organic Solvent Extraction for Chemical Analysis of Medical Devices
April 16th 2025A new study, published by a researcher from Chemical Characterization Solutions in Minnesota, explored a new approach for sample preparation for the chemical characterization of medical devices.
Sorbonne Researchers Develop Miniaturized GC Detector for VOC Analysis
April 16th 2025A team of scientists from the Paris university developed and optimized MAVERIC, a miniaturized and autonomous gas chromatography (GC) system coupled to a nano-gravimetric detector (NGD) based on a NEMS (nano-electromechanical-system) resonator.
Miniaturized GC–MS Method for BVOC Analysis of Spanish Trees
April 16th 2025University of Valladolid scientists used a miniaturized method for analyzing biogenic volatile organic compounds (BVOCs) emitted by tree species, using headspace solid-phase microextraction coupled with gas chromatography and quadrupole time-of-flight mass spectrometry (HS-SPME-GC–QTOF-MS) has been developed.
A Guide to (U)HPLC Column Selection for Protein Analysis
April 16th 2025Analytical scientists are faced with the task of finding the right column from an almost unmanageable range of products. This paper focuses on columns that enable protein analysis under native conditions through size exclusion, hydrophobic interaction, and ion exchange chromatography. It will highlight the different column characteristics—pore size, particle size, base matrices, column dimensions, ligands—and which questions will help decide which columns to use.













