Coupling Supercritical Fluid Chromatography with Evaporative Light Scattering Detection: A Study of the Relevant Parameters Ruling Response
LCGC North America
The mobile-phase flow rate and the amount of modifier mixed with carbon dioxide are the main parameters affecting peak area.
The complex phenomena involved in the use of evaporative light scattering detection (ELSD) with high performance liquid chromatography (HPLC) are fairly well described. Because the mobile phase is nebulized to produce droplets, however, the use of supercritical fluids as mobile phase with this low-pressure detection method is favored because of the fluid depressurization, before the detector entrance, which allows the aerosol formation. Despite the use of ELSD coupled with supercritical fluid chromatography (SFC) for various applications, however, there has been no study of the ELSD response variation with supercritical carbon dioxide–based mobile phases. Nor has any comparison with HPLC in terms of response (peak area) been reported. These points are partially addressed in this article.
The use of evaporative light scattering detection (ELSD) with high performance liquid chromatography (HPLC) overcomes some of the drawbacks and limitations of using ultraviolet (UV) detection — ELSD can be used for the detection of compounds lacking a chromophore group, provides no baseline deviation during gradient elution, and can be used with solvents having a high degree of UV absorption (1,2). The principle of ELSD is based on nebulization of the mobile phase, a process by which an aerosol is produced by the addition of a nebulizer gas (nitrogen) (3–8). This process takes place in a nebulizing chamber that can have various dimensions and volumes depending on the manufacturer. It can also be adapted to the type of chromatography used — HPLC, ultrahigh-pressure LC (UHPLC), or micro-LC. The droplets, consisting of the analyte molecules and an unknown amount of solvent, are introduced into a drift evaporative tube and heated at varied temperatures. The droplet size is reduced by further solvent evaporation as the droplets pass through this tube. Finally, the droplets go through a light source, and the deflected light is collected by a photomultiplier.
The response depends on the particle (droplet) size, which seems to increase with the increasing compound concentration and which is related to numerous physical properties of the solvents composing the mobile phase: viscosity, surface tension, and volatility. This explains that the response (A) variation with the concentration (m) is generally nonlinear for noncapillary columns, and follows the relation:

Consequently, to follow a linear trend, calibration curves should be plotted with double logarithmic coordinates:
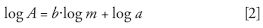
Equation 2 also can be written for identical injected volumes:
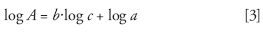
Consequently, using a gradient elution in reversed-phase HPLC, that is, increasing the organic solvent content in the mobile phase, leads to changes in the droplet size that favor the response of the compounds in relation to their retention time (4,9,10). A compensation gradient added after the column makes it possible to limit this drawback by keeping the mobile-phase composition at the detector entrance constant (10,11).
Other parameters induce great response changes, such as the atomization pressure (3,4), which is related to the flow rate of the nebulizer gas; the geometry of the nozzle (mainly the capillary diameter) (7,8); and the location of the mixing point of the mobile phase with this gas. The wavelength of the light source used also plays a role in the response intensity (6).
On the other hand, because of the nebulization process required, coupling supercritical fluid chromatography (SFC) with ELSD has been successfully done in the past (12–18). The specificity of the carbon dioxide–modifier (an organic solvent) mobile phase used in SFC is the pressure required at the column outlet to ensure the dense state of the fluid. This back-pressure is achieved with a back-pressure regulator or a restrictor, located after the column. However, after the back-pressure regulator or restrictor, carbon dioxide returns back to a gaseous state, thus favoring aerosol formation in the nebulizer chamber. However, this carbon dioxide depressurization cools the outlet capillary, which can induce dry-ice formation and possibly plug the capillary. Heating, provided either by the back-pressure regulator or by an additional transfer line, avoids this drawback.
Beyond these studies addressing this coupling of SFC to ELSD, however, few studies are available to describe the varied effects of the numerous chromatographic parameters affecting SFC separations. The difficulty of interfacing SFC with a low-pressure detector, given the phase separation between the liquid solvent and gaseous carbon dioxide, has been addressed (19), but no clear comparison has been performed between SFC and HPLC to evaluate how the use of carbon dioxide instead of a liquid affects ELSD responses in terms of area or sensitivity. That comparison is the topic of this paper. In this study, we used a Sedex 85 ELSD system (Sedere, Orléans, France) with a slightly adapted nebulizer, mainly with carbon dioxide–ethanol mobile phases, with the goal of using green chromatographic conditions.
Experimental
Chromatographic separations were performed using equipment manufactured by Jasco (Tokyo, Japan). Two model 980-PU pumps were used, one for carbon dioxide and another for the modifier. Control of the mobile-phase composition was performed by the modifier pump. The pump head used for carbon dioxide was cooled to -2 °C by a cryostat (Julabo F10c, Seelbach, Germany). When the two solvents (modifier and carbon dioxide) were mixed, the fluid was introduced into a dynamic mixing chamber (model PU 4046, Pye Unicam, Cambridge, UK) connected to a pulsation damper. The injector valve was supplied with a 20-μL loop (model 7125, Rheodyne, Cotati, California).
The columns were thermostated by an oven (Jetstream 2 Plus, Thermotechnic, Langenzersdorf, Germany). A UV detector with a high pressure–resistant cell was set to control the injection repeatability and volume (HP 1050, Hewlett-Packard). The detection wavelength was 210 nm. After the detector, the outlet column pressure was controlled by a Jasco 880-81 back-pressure regulator. The outlet regulator tube (internal diameter 0.25 mm) was heated to 60 °C to avoid dry ice formation during the carbon dioxide depressurization. The ELSD system (Sedex 85) was connected to the outlet capillary. The nebulizer tubing was slightly different from the one used with HPLC, in that the capillary diameter was 150 μm. The nebulizer gas (nitrogen) pressure was 2 bar and the drift temperature was 50 °C. Chromatograms were recorded using Azur software (Datalys, France).
The main column used was a 250 mm × 4.6 mm, 5-μm dp Synergi Fusion RP column (Phenomenex, Torrance, California), the mobile phase was 90:10 carbon dioxide–ethanol (except for the study of the modifier percentage), the temperature was 25 °C, the outlet pressure was 15 MPa, and the flow rate was 3 mL/min. Caffeine was provided by Sigma-Aldrich (St. Louis, Missouri). The injected volume was 5 μL for most studies, and the concentration was 2.5 mg/mL. Other columns used included a 250 mm × 4.6 mm, 5-μm dp Cyano column (Princeton, Cranbury, New Jersey) and a 250 mm × 4.6 mm, 5-μm dp Uptisphere HDO column (Interchim, Montluçon, France).
Results and Discussion
A first series of experiments was performed to study basic parameters: injected volume, additional gas pressure, outlet column pressure, drift temperature, and flow rate of the mobile phase. To ensure accuracy of the quantitative value studied, no split was used for these experiments. That means that the entire volume of supercritical mobile phase eluted from the column was introduced into the detector. By using a Jasco back-pressure regulator, the transfer capillary (around 10 cm in length) was heated to 60 °C. This was sufficient to avoid dry-ice formation and plugging of the transfer line.
Figure 1a shows the variation of the caffeine peak area with the injected volume from 2 to 15 μL, with both the UV and evaporative light scattering detectors. As expected, the UV response displays a linear variation, showing the accuracy of tested volumes. The variation of the ELSD response appeared to be exponential. Figure 1b shows, by using double logarithmic coordinates, that the relationship between the response and the injected volume is linear. This behavior (Figure 1a) is classical in comparison to the one observed in HPLC (1). The standard deviation measured for 10 injections of 10 μL was 0.5% for ELSD and 1.0% for UV detection.

Figure 1
Figure 2 shows the detector response compared to the nebulizer gas pressure, from 0 to 4 bar (5 μL injected). For this study, the nebulizer chamber of a Sedex 55 ELSD system was also tested with the same nebulizer, because this model had been used for analysis of carbohydrates by SFC in the 1990s (13). The difference between the two chambers is mainly the length, with the chamber of the Sedex 85 system being about twice as long as that of the Sedex 55 system.
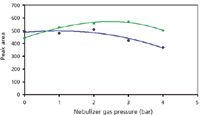
Figure 2
The highest response values were obtained with the longer nebulizing chamber (Sedex 85) at 3 mL/min, which seemed to be the optimal value. With this longer chamber, the addition of the nebulizer gas improved the response. Finally, the optimal nebulizer gas pressure was close to the pressure usually used in HPLC, despite the gaseous carbon dioxide introduced into the chamber in SFC. As reported elsewhere (20,21), the increase in the nebulizer gas flow rate induces both the shift of the droplet size toward smaller droplets and the increase of the number of droplets. These two effects should provide opposite response changes, but the second effect must be greater to explain the observed result (20). The strong effect of gas pressure on response was noticed also with HPLC (4). In contrast to our results, other studies report a rapid response decrease with an increase in the additional gas flow rate (22,23), but the design of the nebulizing chamber, and the interface section between the chamber and the drift evaporative tube are not identical from one detector to another (24) and those differences may well be responsible for the opposite observations.
Nevertheless, the responses obtained without additional gas (Figure 2) are not dramatically lower than those obtained with additional nebulizer gas, showing a rather good production of droplets as a result of the carbon dioxide depressurization.
The outlet pressure (Pout) applied to the chromatographic system was also studied from 9 to 19 MPa (Figure 3). With both detection methods, but mainly for ELSD, the response was enhanced by an increase in outlet pressure. Increasing Pout enhances the fluid density before the back-pressure regulator, leading to a reduction in the linear speed of the mobile phase that enhances the residence time of the compounds in the cell; the increased residence time explains the response increase, mainly from 11 to 16 MPa (a refractive index change in the cell also can modify the UV response) (25). The greater amount of carbon dioxide before the back-pressure regulator also results in an enhanced fluid speed in the nebulizer (after the back-pressure regulator), which seems to favor the ELSD response.
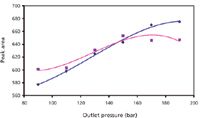
Figure 3
The temperature of the drift evaporative tube was also studied from 40 °C to 70 °C (Figure 4). With caffeine, a semivolatile compound, the increase in this evaporative temperature leads to a regular decrease in response. This classical behavior has been reported with HPLC (3) and results from the reduction of particle size by vaporization with increasing temperature.
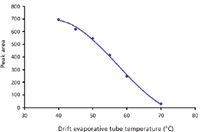
Figure 4
The flow rate of the mobile phase was also studied from 2 to 5 mL/min (Figure 5). With both detectors the response decreases when flow rate increases. For UV, a concentration detection method, this behavior is expected; the residence time in the cell decreases as the mobile-phase flow rate increases. As reported previously, the nonlinear decrease of the UV response also could be caused by varied effects related to the fluid density change. However, for ELSD, a mass detection method, these flow-rate changes would not modify the amount of compound going through the detection system.
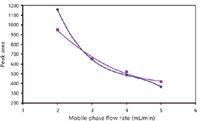
Figure 5
In HPLC, varied trends have been observed. An increase in response with the flow rate (from 0.1 to 1 mL/min) was reported for a concentric nebulizer by direct solute injection (no chromatographic column) (23), whereas the opposite was seen with different mobile phases, different compounds, and drift temperatures (6,21,26).
From the Nukiyama–Tanasawa equation (27), the increase in the mobile-phase flow rate should improve the average particle diameter. Larger droplet diameters are supposed to enhance ELSD response (up to a limit value) (6,28), but they also favor droplet condensation and precipitation on the walls of the nebulizing chamber, thus decreasing the number of droplets that can enter the drift evaporative tube. Nonetheless, this equation describes the primary droplet size distribution (the distribution obtained just after the nebulizer), whereas the measured response is that of the ternary distribution (after evaporation and loss of larger droplets by impaction) (24,29), limiting the relevance of this equation to describe the phenomena encountered. Moreover, this equation does not take into account the flow conditions, which are more often turbulent than laminar (29). Consequently, several works report the poor calculation of the droplet size distribution from this equation in some cases (30). In particular, investigating the effect of a flow rate increase on the droplet size, it was observed that the droplets were smaller than estimated based on the equation (31).
Whatever the cause, this decrease is observed in packed-column SFC, where the total liquid flow rate entering the nebulizer is 0.2–0.5 mL/min (the mobile phase was 90:10 [v/v] carbon dioxide–ethanol, 2–5 mL/min). The fluid depressurization induces a cooling effect, which increases with the flow-rate increase. As a result, the condensation of droplets increases, meaning that the amount of compound going through the light beam decreases.
Figure 6 shows the response variation for a diastereoisomer separation at two mobile-phase flow rates. As expected, for the two signals, the retention and peak width are lower, and the height of the peak is quite constant with UV. However, for the ELSD response, both the peak height and area are dramatically reduced for the higher flow rate, suggesting a strong modification in the droplet size distribution. Figure 7 shows the separation and detection of various pharmaceutical compounds in carbon dioxide–ethanol mobile phase. Theophylline, which is included in this set of compounds, displays a log P equal to –0.32, explaining why this compound is often studied by hydrophilic interaction chromatography (HILIC) (32–34). The other compounds display higher log P values (1.16 for dexamethasone, 1.20 for cortisone, and 1.66 for hydrocortisone) (35). The separation of these four compounds is achieved in carbon dioxide–ethanol mobile phase without any additives (acidic or basic), with good peak symmetry. The response of at least three compounds (cortisone, hydrocortisone, and dexamethasone) is higher with ELSD detection than with UV detection, regardless of the flow rate used, 3 or 5 mL/min.
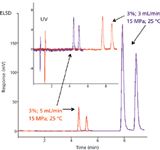
Figure 6
As expected, the peak intensity decreases with ELSD when increasing the mobile phase flow rate, but the ELSD response is still higher than the UV response, despite the very low wavelength used (210 nm) with the SFC mobile phase. The amount of organic solvent added to carbon dioxide was also studied (3–20% ethanol).

Figure 7
As reported previously, the mobile- phase composition induces response changes, both in HPLC (9) and SFC (10), leading to the use of an additional inverse gradient to maintain a constant response (10). The use of organic solvent is reported to shift the droplet size toward smaller values (36).
Figure 8 shows that the increase in ethanol favors the compound elution (diastereoisomers), but dramatically reduces their ELSD response. This means that isocratic conditions should be preferred for SFC–ELSD and that the stationary phase should be carefully selected to allow good separation with the smallest modifier proportion. Classifications of stationary phases in SFC favoring this choice are available (37,38).
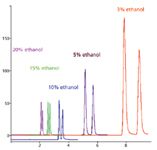
Figure 8
To compare the HPLC and SFC responses, studies were carried out for caffeine with different mobile phases (methanol–water, ethanol–water, acetontrile–water, and carbon dioxide–ethanol), having close eluting power to maintain identical retention factors (k).
With all mobile phases, the column provided symmetrical peaks. Figure 9 shows a log–log plot of peak area versus concentration for these mobile phases. Two flow rates (3 and 1 mL/min) were studied in SFC. Satisfactory linearity is reported up to 50 ppm in all cases (log–log coordinates). The response increases from methanol–water to acetonitrile–water, and ethanol–water, then SFC at 3 mL/min. The response is multiplied by three from methanol to acetonitrile or ethanol in HPLC, and by three again from acetonitrile or ethanol in HPLC to SFC with 10% ethanol (3 mL/min). Moreover, at 1 mL/min, the signal is dramatically higher in SFC. The b coefficient regularly decreases in the order methanol<ethanol<acetontrile<SFC at 1 mL/min. Numerous studies report that this coefficient is related to the droplet size (39). The higher the value of b, the lower the droplet size. Does that mean that in SFC, as a result of the lower b value, the droplets are bigger? Another study reported the reverse effect in packed-capillary SFC by using 2-propanol as modifier (40). However, the use of capillary columns, that is, of very low flow rates and injected volumes, seems to affect the response significantly because of the direct linear relationships between the peak area and the concentration observed in such conditions (8,40–42).

Figure 9
Whatever the reasons, the peak area in SFC is dramatically improved. Lower droplet condensation is observed in SFC conditions because of the lower amount of liquid in the mobile phases (10% for our studies), apparently favoring the transfer of analyte from the nebulizer to the drift evaporative tube.
The difference observed for the two flow rates in SFC could be a result of varied cooling effects of the nebulization chamber — greater at higher flow rates — which induces a greater droplet condensation on the wall (the overall signal decreases from 1 to 3 mL/min) and a larger droplet size of the ternary aerosol, which explains the lower b value at 1 mL/min.
Finally, there is an experimental linear correlation in HPLC between the two coefficients, b and log a (24). Figure 10 shows this correlation for 14 varied compounds in SFC: palmitic and linolenic acid, tripalmitoyl- and trilinonenoylglycerol, cholesterol, hexadecanol, octadecanol, eicosanol, docosanol, theophylline, ketoprofen, hydrocortisone, caffeine, and 4-hydroxybutylparaben. A linear relationship is observed in SFC with 90:10 (v/v) carbon dioxide–ethanol mobile phase (blue diamonds). Furthermore, other data obtained in HPLC were added to Figure 10. With a Sedere 75 detector in a nonaqueous reversed-phase LC separation (acetontrile–dichloromethane) (39), the values for one compound (tricaprylin) with different post-column addition (red squares) are very close to the values obtained in SFC with various compounds. The slopes of the tendency curves are also very close for direct injection of ceramide with various pure organic solvents (pink line) (26) and for HPLC analysis of neomycin with aqueous mobile phases with a Sedere 75 detector (green line) (43). The main differences are noticed on the intercept, which appears to be strongly improved with aqueous mobile phases and lowered with direct injection in pure organic solvents.

Figure 10
Conclusion
The use of ELSD with SFC is favored by the nature of the mobile phase in SFC, which enables aerosol formation and thus improves the peak area, at least for the carbon dioxide–ethanol mixture. The b coefficient is smaller, possibly indicating that the particle size is greater. Conversely, the log a value is greater, explaining the higher response obtained in SFC conditions. From the numerous analytical parameters studied, the mobile-phase flow rate and the amount of modifier mixed with carbon dioxide are the main parameters affecting peak area. The outlet pressure also plays a role in response by affecting the speed of fluid in the nebulizer. Finally, these studies show that using a heated back-pressure regulator, the slightly modified nebulizer chamber of the Sedere 85 is well suited for SFC analysis.
References
(1) N.C. Megoulas and M.A. Koupparis, Crit. Rev. Anal. Chem. 35, 301–316 (2005).
(2) K. Petritis, C. Elfakir, and M. Dreux, J. Chromatogr. A 961, 9–21 (2002).
(3) J.M. Charlesworth, Anal. Chem. 50(11), 1414–1420 (1978).
(4) T.H. Mourey and L.E. Oppenheimer, Anal. Chem. 56, 2427–2434 (1984).
(5) A. Stolyhwo, H. Colin, and G. Guiochon, J. Chromatogr. 323, 1–18 (1983).
(6) P. Van der Meeren, J. Vanderdeelen, and L. Baert, Anal. Chem. 64, 1056–1062 (1992).
(7) R. Trones, T. Andersen, I. Hunnes, and T. Greibrokk, J. Chromatogr. A 81, 55–61 (1998).
(8) S. Hoffmann, H.R. Norli, and T. Greibrokk, J. High Resolut. Chromatogr. 12, 260 (1989).
(9) B.T. Mathews, P.D. Higginson, R. Lyons, J.C. Mitchell, N.W. Sach, M.J. Snowden, M.R. Taylor, and A.G. Wright, Chromatographia, 60(11/12), 625–633 (2004).
(10) A. de Villiers, T. Gorecki, F. Lynen, R. Szucs, and P. Sandra, J. Chromatogr. A 1161, 183–191 (2007).
(11) M. Lisa, F. Lynen, M. Holcapek, and P. Sandra, J. Chromatogr. A 1176, 135–142 (2007).
(12) A. Salvador, B. Hebreteau, M. Lafosse, and M. Dreux, J. Chromatogr. A 785, 195–204 (1997).
(13) M. Lafosse, B. Hebreteau, and L. Morin-Allory, J. Chromatogr. A 720, 61–73 (1996).
(14) J. Thompson, B. Strode III, L. Taylor, and T.A. van Beek, J. Chromatogr. A 738, 115–122 (1996).
(15) K. Gaudin, E. Lesellier, P. Chaminade, D. Ferrier, A. Baillet, and A. Tchapla, J. Chromatogr. A 883, 211–222 (2000).
(16) F.S. Deschamps, K. Gaudin, E. Lesellier, A. Tchapla, D. Ferrier, A. Baillet, and P. Chaminade, Chromatographia, 54, 607–611 (2001).
(17) E. Lesellier, K. Gaudin, P. Chaminade, A. Tchapla, and A. Baillet, J. Chromatogr. A 1016, 111–121 (2003).
(18) F.S. Deschamps, E. Lesellier, J. Bleton, A. Baillet, A. Tchapla, and P. Chaminade, J. Chromatogr. A 1040, 115–121 (2004).
(19) T.L. Chester and J.D. Pinkston, J. Chromatogr. A 807, 265–273 (1998).
(20) A. Stolyhwo, H. Colin, M. Martin, and G. Guiochon, J. Chromatogr. 288, 253–275 (1984).
(21) J.W. Nowak Jr. and R.F. Browner, Anal. Chem. 52, 792–796 (1980).
(22) S. Almeling and U. Holzgrabe, J. Chromatogr. A 1217, 2163–2170 (2010).
(23) J.L. Robinson, M. Tsimidou, and R. Macrae, J. Chromatogr. 324, 35–51 (1985).
(24) M. Rigghezza and G. Guiochon, J. Liq. Chromatogr. 11, 1969–2004 (1988).
(25) T.A. Berger and B.K. Berger, J. Chromatogr. A 1218, 2320–2326 (2011).
(26) F.S. Deschamps, A. Baillet, and P. Chaminade, Analyst, 127, 35-41 (2002).
(27) S. Nukiyama and Y. Tanasawa, Trans. Soc. Mech. Eng. 5, 62–67 (1939).
(28) G. Guiochon, A. Moysan, and C. Holley, J. Liq. Chromatogr. 11, 2547–2570 (1988).
(29) R.F. Browner, A.W. Boorn, and D.D. Smith, Anal. Chem. 54, 1411-1419 (1982).
(30) R.A. Mugele and H.D. Evans, Ind. Eng. Chem. 43, 1317–1324 (1951).
(31) A. Canals, V. Hernandis, and R.F. Browner, J. Anal. Atomic Spectr. 5, 61-66 (1990).
(32) M. Lämmerhofer, M. Richter, J. Wu, R. Nogueira, W. Bicker, and W. Lindner, J. Sep. Sci. 31, 2572–2588 (2008).
(33) G. Shuster and W. Lindner, Anal. Bioanal. Chem. DOI 10.1007/s002 16-011-4745-5.
(34) A. dos Santos Pereira, A. Jimenez Giron, E. Admasu, and P. Sandra, J. Sep. Sci. 33, 834–837 (2010).
(35) K. Kurata, T. Shimoyama, and A. Dobashi, J. Chromatogr. A 1012, 47–56 (2003).
(36) J. Farino and R. Browner, Anal. Chem. 56, 2709–2714 (1984).
(37) C. West and E. Lesellier, J. Chromatogr. A 1191, 21–39 (2008).
(38) C. West and E. Lesellier, J. Chromatogr. A 1110, 191–199 (2006).
(39) S. Heron, M.G. Maloumbi, M. Dreux, E. Verette, and A. Tchapla, LCGC Europe 19 (12), 664–672 (2006).
(40) S. Hoffmann and T. Greibrockk, J. Micro. Sep. 1, 35–40 (1989).
(41) Z. Cobb, P.N. Shaw, L.L. Lloyyd, N. Wrench, and D.A. Barrett, J. Micro. Sep. 13, 169–175 (2001).
(42) J.N. Alexander IV, J. Micro. Sep. 10, 491–502 (1998).
(43) N.C. Megoulas and M.A. Koupparis, J. Chromatogr. A 1057, 125–131 (2004).
E.Lesellier and C. West are with the Institut de Chimie Organique et Analytique UMR 7311, at the Université d'Orléans in Orléans, France. Direct correspondence to: eric.lesellier@univ-orleans.fr.
T. Bizet, A. Valarché, and M. Dreux are with Sedere in Orléans, France.

Determining Enhanced Sensitivity to Odors due to Anxiety-Associated Chemosignals with GC
May 8th 2025Based on their hypothesis that smelling anxiety chemosignals can, like visual anxiety induction, lead to an increase in odor sensitivity, a joint study between the University of Erlangen-Nuremberg (Erlangen, Germany) and the Fraunhofer Institute for Process Engineering and Packaging (Freising, Germany) combined behavioral experiments, odor profile analysis by a trained panel, and instrumental analysis of odorants (gas chromatography-olfactometry) and volatiles (gas chromatography-mass spectrometry).
Investigating 3D-Printable Stationary Phases in Liquid Chromatography
May 7th 20253D printing technology has potential in chromatography, but a major challenge is developing materials with both high porosity and robust mechanical properties. Recently, scientists compared the separation performances of eight different 3D printable stationary phases.
Detecting Hyper-Fast Chromatographic Peaks Using Ion Mobility Spectrometry
May 6th 2025Ion mobility spectrometers can detect trace compounds quickly, though they can face various issues with detecting certain peaks. University of Hannover scientists created a new system for resolving hyper-fast gas chromatography (GC) peaks.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)










