Applying Comprehensive Analysis to EPA Method 1613B Samples — Discover Those Compounds Usually Discounted in Environmental Samples
Green Foodomics
Polychlorinated dibenzo-p-dioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs) are ubiquitous environmental pollutants that are persistent and toxic. The historical quantitative analysis of these compounds has been achieved with high-resolution targeted analysis using magnetic sector instruments, with subsequent lower resolution analysis to identify other contaminants. Advances in technology have led to comprehensive time-of-flight (TOF) mass spectrometers that can quantify PCDDs or PCDFs while simultaneously acquiring data on other contaminants in the samples. Samples that had been analyzed using EPA Method 1613B conditions on a sector instrument were analyzed with a high performance TOF mass spectrometer and a low-resolution comprehensive two-dimensional gas chromatography (GC×GC) TOF instrument. The quantitative results from the sector, the high-resolution TOF, and the GC×GC-TOF systems are compared in this article.
Polychlorinated dibenzo-p-dioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs) are ubiquitous environmental pollutants formed as by-products of industrial and thermal processes. They form part of a class of compounds known as persistent organic pollutants (POPs), as defined in the Stockholm Convention (SC), that are persistent, geographically widely distributed, bioaccumulative, and have the potential to cause adverse health and environmental effects (1). The historical analysis of these compounds has required the utilization of two technologies; magnetic sector instruments and lower resolution mass spectrometry (MS) (that is, quadrupole or ion trap) with multiple stages of analysis. High-resolution targeted analysis on a magnetic sector instrument is applied for quantitation of dioxins followed by lower resolution analysis to characterize other possible contaminants (2). The quantitative standard by which all other analyses for these compounds are compared is EPA Method 1613B (3) and its derivatives. Recent advances in MS technology have led to comprehensive time-of-flight (TOF) mass spectrometers that can provide quantitation of dioxins through much of the mandated concentration range while simultaneously acquiring data on other contaminants in the samples.
This article compares results from samples prepared and analyzed according to EPA Method 1613B with those from a high-resolution, accurate mass TOF mass spectrometer and a low-resolution TOF mass spectrometer interfaced to a comprehensive two-dimensional gas chromatography (GC) system (GC×GC–TOF-MS). The TOF systems were able to detect dioxins across a broad concentration range covering more than 75% of the reported results obtained under EPA Method 1613B conditions. Quantitation was established in all instances using commercial standards and confirmed that the TOF systems could successfully achieve or surpass (4) detection at the mandatory lower limit required by EPA Method 1613 (TCDD at 500 fg) (3). (Note: Throughout this article the relevant PCDD and PCDF will be described using the following abbreviations: XCDD and XCDF, where X = T [tetra], P [penta], Hx [hexa], Hp [hepta], and O [octa]). The quantitative results obtained from the GC–high-resolution TOF (GC–HRTOF-MS) system and the GC×GC–TOF-MS systems were correlated with those obtained under EPA Method 1613B conditions.
More significantly, while providing comparable quantitative results, the TOF systems simultaneously allowed for the identification of a diverse set of other POPs not detected under the highly selective, selected ion monitoring (SIM) conditions defined in EPA Method 1613B. SIM analyses require prior knowledge of the compounds to be analyzed. The retention times and windows for mass-to-charge are highly constrained to limit interference and provide selective detection with correlation to labeled standards. Because of the constrained conditions for mass analysis under Method 1613B, other analytes would not be detected, while the TOF systems, not suffering the same constraints, always yield full range mass spectra and provide considerable additional sample information. The absence of constraints (that is, comprehensive analysis) enables a single experiment to obtain good quantitative correlation with regulated methods while data for prospective or retrospective analysis of unknowns is also acquired. The isotopic abundance of the analyte ions, mass accuracy, and resolving power achieved during the comprehensive experiment on the GC–HRTOF-MS system provide for both robust quantitative information and confident analyte identification. In the case of the GC×GC–TOF-MS system, the added chromatographic resolving power and cryofocusing effect of thermal modulation provides added sensitivity and selectivity.
Legislative Considerations
The analysis of PCDDs and PCDFs is regulated globally and concentrations are determined using a variety of EPA methods (such as 1613B and 8290A) (3,5), and by Council Directives in the European Union (6). In most other countries requiring dioxin analysis, modified forms of these methods govern testing protocols. These methods prescribe the use of GC coupled with high-resolution mass spectrometry (HRMS) and stipulate minimum quantifiable levels of the analyte (TCDD at 500 fg) for an analytical system and the testing to be deemed acceptable. Any alternative technology should be able to achieve the levels of detection required by these methods and the capabilities must be verified through testing.
Analytical Considerations
The limitation of the sector HRMS approach lies in its targeted approach that provides accurate information on quantitative levels of PCDDs and PCDFs, but gives no information on the presence of other POPs in the sample. This has to be determined in separate runs, and is generally done using low-resolution mass spectrometry. To be able to do both quantification and sample component investigation in a single analysis holds obvious advantages.
Samples
Six samples were investigated in this study. These were sourced from the Ministry of the Environment Laboratory Services Branch in Toronto, Canada. All samples were prepared according to standard methods used for the preparation of samples for PCDD and PCDF analysis by HRMS (7).
Instrumentation
Conditions for EPA Method 1613B acquisition were achieved using a Waters Autospec magnetic sector mass spectrometer at a resolving power of 10,000 interfaced to an HP6890 gas chromatograph (Agilent Technologies) using a 40 m × 0.18 mm, 0.18-μm df DB-5 column (J&W Scientific).
The GC×GC–TOF-MS system consisted of a Pegasus 4D time-of-flight mass spectrometer (LECO Corporation) coupled to an Agilent 7890 GC system equipped with an Agilent 7683B autosampler, a secondary oven and a dual-stage, quad-jet thermal modulator. Liquid nitrogen was used for the cold jets and synthetic air for the hot jets. The liquid nitrogen levels were maintained using an AMI model 186 liquid level controller.
The HRTOF-MS system was a Pegasus HRT (LECO Corporation) using Folded Flight Path technology (8,9). The system included an Agilent 7890 GC system equipped with an Agilent 4513A autosampler.
Experimental
EPA Method 1613B calibration and verification solutions (EPA-1613CVS), labeled calibration solutions (EPA-1613LCS), internal standard spiking solution (EPA-1613ISS), and cleanup standard stock solution (EPA-1613CSS) were selected for spiking and calibration purposes. These solutions were purchased from Wellington Laboratories and contained the 17 native and corresponding mass-labeled PCDD and PCDF congeners in nonane.
All instrument functions and data processing for the GC×GC–TOF-MS were managed with the LECO ChromaTOF software (version 4.44). All instrument functions and data processing for the Pegasus HRT system were managed using LECO ChromaTOF software (version 1.61). Manual review of all peak identifications and integrations was performed using this software. Library searching was performed using a PCDD and PCDF user library compiled from the PCDD and PCDF standards. GC–HRMS data were obtained using conditions and processing as specified in EPA Method 1613B.
Experimental conditions used for the analysis of the samples are shown in Table I.
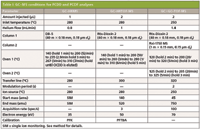
Table I: GCâMS conditions for PCDD and PCDF analyses
Results
Limits of Detection
The results obtained from the EPA Method 1613B (3) standard set provides an estimate of the limits of detection (LOD) possible using the GC×GC–TOF-MS system and GC with HRTOF-MS detection, with the understanding that matrix interference and sample preparation effectiveness will contribute significantly. The low-level standard (CS1), which contains 2,3,7,8-TCDD at 0.5 pg/μL, was used to provide an estimated LOD using GC×GC–TOF-MS. Using the methodology described in EPA Method 1613B, the signal-to-noise ratio (S/N) for the ion of m/z 322 for 2,3,7,8-TCDD was calculated as 20 (EPA Method 1613 requires this ratio to be >10 [3]).
However, for GC–HRTOF-MS the prescribed approach is not applicable. Modern high-resolution mass spectrometers show little chemical noise on the plot of an exact mass ion owing to the elimination of noise during processing and acquisition. This is demonstrated in Figure 1, which shows a plot of the ion at m/z 321.893 for TCDD at 500 fg/μL (CS1 standard). As can be seen, the compound is readily detected at the lowest level (500 fg/μL) required by EPA Method 1613B indicating a capability of achieving the minimum levels. It is, however, often desirable to detect lower in routine analyses to ensure compliance and analytical capability.
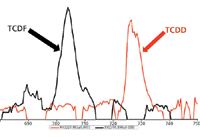
Figure 1: Selected ion chromatogram (m/z 321.893) for TCDD at 500 fg/μL (CS1 standard) on the HRT system.
Quantitative
The quantitative values for the 17 PCDDs and PCDFs whose levels are regulated using Method 1613B are shown in Table II. Values obtained using GC×GC–TOF-MS and GC–HRTOF-MS are compared to the levels acquired using 1613B conditions. At the lowest levels, the GC×GC–TOF-MS system is not capable of detecting analytes, but this may be partially offset using the concurrent solvent recondensation technique of large-volume splitless injection (10). This technique, originally described by Magni and Porzano (10), can be done in an unmodified split–splitless injection port, provided that a guard column is used to protect the analytical column and that the matrix is clean to minimize fouling of the inlet and columns. For dioxin analysis in which the sample has been subjected to considerable cleanup, this latter condition is met, and the technique is advantageous when trying to analyze the trace-level PCDDs and PCDFs in the sample on the low-resolution system.
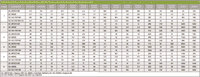
Table II: Quantitative values (pg/g) for PCDDs and PCDFs (T = tetra; P = penta; Hx =Hexa; Hp = hepta; O = octa)
The data show strong correlation between Method 1613B results and those from GC×GC–TOF-MS analysis, suggesting its viability as a screening tool for dioxin levels in complex samples prepared using Method 1613B protocols.
Similarly, when compared to one another, the results from the two high-resolution systems are generally in excellent agreement, showing the high-resolution TOF system to provide quantitative results comparable to those obtained under Method 1613B criterion when concentrations are above a threshold near 500 fg/μL.
Comprehensive
Targeted dioxin analysis, as obtained with Method 1613B, fails to provide a comprehensive picture of the samples. Only the targeted PCDDs and PCDFs are detected, and additional POPs and analytes with dioxin-like properties present in the samples are transparent to detection. The capability of TOF instrumentation to generally reach or approach the required low levels, while still acquiring full-range mass spectra, means that these systems can be used not only to quantify the PCDD and PCDF components, but also to detect and identify additional components in the sample in the same run. The ability to detect and identify analytes not specifically monitored in Method 1613B is a feature demonstrated by both the GC×GC–TOF-MS and GC–HRTOF-MS systems.

Figure 2: (a) GCÃCâTOF-MS surface plot showing the m/z 360 extracted ion. A dibromopyrene congener is one of a cluster of peaks in the top right corner. (b) GCâHRTOF chromatogram showing the m/z 359.896 extracted ion and the 2,7-dibromopyrene peak on the right side.
For example, the GC×GC–TOF-MS chromatogram for sample 2 (Figure 2a) shows a high-boiling compound containing a prominent molecular ion cluster at m/z 360, not specified in the ions monitored in Method 1613B. The deconvoluted mass spectrum obtained for this compound can be library-searched to obtain the result shown in Figure 3. The compound is identified as 2,7-dibromopyrene, that can be confirmed against a standard to verify the congener, with a spectral match of 84% and would have gone undetected under Method 1613B conditions. Bromopyrenes have been shown to be irritating to the skin, eye, and respiratory systems (11) and while not the health concern of a dioxin, this compound represents what could be missed and may provide valuable insight should a contamination with dioxins occur. Although the toxicity of this specific dibromopyrene is not known, it may well contribute to the overall sample toxicity and so its presence in the sample may be of importance.
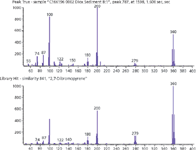
Figure 3: Mass spectrum of high boiling compound from sample 2, which gives an 84.1% match with dibromopyrene.
This same compound can be located in the chromatogram of sample 2 obtained when using the GC–HRTOF-MS system (Figure 2b). In this case, the relevant mass is measured as 359.89648, which can be used to determine the formula C16H8Br2 with a mass accuracy of -0.55 ppm. In the case of the GC×GC experiment, it is library match alone that provides identification. In the case of the GC–HRTOF-MS analyses, accurate mass provides a clear indication of the likely formula and confirms the library match.
Numerous other POPs, which would not have been detected in the sector data, have been found in the samples. Examples of these are shown in Table III. It should be noted that the compounds shown are a random selection among many. It is only possible to obtain a full picture of the sample when a comprehensive analysis is performed. By focusing just on the PCDDs and PCDFs, only a partial understanding of the total toxicity factor is obtained. It should also be kept in mind that the sample has undergone rigorous cleanup before analysis. It is likely that this cleanup would remove other potentially harmful components (for example, pesticides).
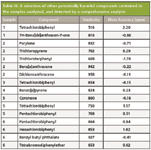
Table III: A selection of other potentially harmful compounds contained in the samples analyzed, and detected by a comprehensive analysis
Mass Accuracy and Isotope Abundance
An important part of the identification of unknown compounds is the measurement of accurate mass (see above) and the robust determination of isotope abundance. For example, one of the important POPs identified in sample 6 is tetrabromodiphenylether (TBDE), a member of a class of compounds known as brominated flame retardants (BFRs), with a chemical formula C12H6Br4O, and a molecular mass of 482. The measured molecular ion was 481.71496 Da and the calculated value is 481.71455 Da, with a Δm of 0.00041 Da (or 0.85 ppm) leading to confident confirmation of its identity.
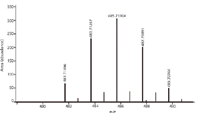
Figure 4: Measured molecular ion cluster for tetrabromodiphenylether.
In the case of compounds with pronounced molecular ion clusters (arising from the presence of chlorine or bromine in the molecule), measurement of the relative isotopic abundance in the molecular ion cluster is also an important confirmation of molecular formula. In the case of the tetrabromodiphenylether, a high-resolution mass spectrum is shown in Figure 4 and the calculated values comparing measured with theoretical are provided in Table IV.

Table IV: Ion abundance differences between measured and calculated relative abundances for tetrabromodiphenylether
In general, differences of up to 30% are considered acceptable (Method 1613B) when working with PCDDs and PCDFs at low levels. If that criterion is applied to the spectrum of the low-level tetrabromodiphenylether then, as can be seen from Table IV, all of the ions fall within the acceptable level except for the low-level ion at 481.71 Da. These values, coupled with the excellent mass accuracy obtained on the molecular ion provide strong confirmatory evidence of the proposed identification.
Conclusion
Analysis of samples suspected to contain PCDDs and PCDFs using GC–MS with appropriate selectivity and sensitivity requires a targeted approach. EPA Method 1613B uses GC–HRMS using SIM analysis. This requirement compromises the ability to identify other priority POPs that may be present in the samples in the same run using a single analytical technique. Time-of-flight mass spectrometry, implemented either as a high-resolution, accurate mass instrument, or as a low-resolution instrument coupled with comprehensive two-dimensional GC, provides an alternative to the traditional, regulated approach. TOF systems have the sensitivity to achieve the low detection levels mandated by regulated methods while showing a strong correlation to results obtained using the regulated methods, and in addition provide the flexibility to detect and identify other priority pollutants in the same analytical run. Although not able to achieve detection in 100% of the instances, the benefit of providing a comprehensive result and detection of EPA Method 1613B analytes at or above the limits provides advantages. These capabilities lead to the opportunity for considerable savings of time and money. As such, these comprehensive technologies form a potent weapon in the hands of the environmental analyst.
Jayne de Vos is with the National Metrolgy Institute of South Africa in Pretoria, South Africa. Jack Cochran is with Restek Corporation in State College, Pennsylvania. Eric J. Reiner is with Ontario Ministry of the Environment in Ontario, Canada. Peter Gorst-Allman is with LECO Africa in Pretoria, South Africa. Direct correspondence to: peter@lecoafrica.co.za
References
(1) Stockholm convention on persistent organic pollutants. Website: http://chm.pops.int (2011).
(2) E.J. Reiner, Mass Spectrom. Rev. 26, 526–259 (2010).
(3) EPA Method 1613: Tetra through octa-chlorinated dioxins and furans by isotopic dilution HRGC/HRMS. Website: http://www.epa.gov/region3/1613.pdf (1994).
(4) J. de Vos, P. Gorst-Allman, and E. Rohwer, J. Chromatogr., A 1218, 3282 (2011).
(5) EPA Method 8290A: Polychlorinated dibenzo-p-dioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs) by high-resolution gas chromatography/high-resolution mass spectrometry (HRGC/HRMS).
(6) Official Journal of the European Communities, L22⅛ (2002).
(7) Ontario Ministry of the Environment. The determination of polychlorinated dibenzo-p-dioxins, polychlorinated furans and dioxin-like PCBs in environmental matrices by GC-HRMS. Report number DFPCB-E3418 (2010).
(8) A.N. Verentchikov, M.I. Yavor, Y.I. Hasin, and M.A. Gavrik, Tech. Phys. 50, 73-81 (2005).
(9) A.N. Verentchikov, M.I. Yavor, Y.I. Hasin, and M.A. Gavrik, Tech. Phys. 50, 82-86 (2005).
(10) P Magni and T Porzano, J. Sep. Sci. 26, 1491 (2003).
(11) www.lookchem.com, Safety Data Sheet.

Thermodynamic Insights into Organic Solvent Extraction for Chemical Analysis of Medical Devices
April 16th 2025A new study, published by a researcher from Chemical Characterization Solutions in Minnesota, explored a new approach for sample preparation for the chemical characterization of medical devices.
Sorbonne Researchers Develop Miniaturized GC Detector for VOC Analysis
April 16th 2025A team of scientists from the Paris university developed and optimized MAVERIC, a miniaturized and autonomous gas chromatography (GC) system coupled to a nano-gravimetric detector (NGD) based on a NEMS (nano-electromechanical-system) resonator.
Miniaturized GC–MS Method for BVOC Analysis of Spanish Trees
April 16th 2025University of Valladolid scientists used a miniaturized method for analyzing biogenic volatile organic compounds (BVOCs) emitted by tree species, using headspace solid-phase microextraction coupled with gas chromatography and quadrupole time-of-flight mass spectrometry (HS-SPME-GC–QTOF-MS) has been developed.
A Guide to (U)HPLC Column Selection for Protein Analysis
April 16th 2025Analytical scientists are faced with the task of finding the right column from an almost unmanageable range of products. This paper focuses on columns that enable protein analysis under native conditions through size exclusion, hydrophobic interaction, and ion exchange chromatography. It will highlight the different column characteristics—pore size, particle size, base matrices, column dimensions, ligands—and which questions will help decide which columns to use.



















