Translating HPLC Performance Gains of Core-Shell Media to LC–MS Applications
Special Issues
Adapting the use of "ultrahigh" performance chromatography for liquid chromatography–mass spectrometry (LC–MS) applications requires specific considerations in integrating the instrument platforms. Mobile phase options are limited to volatile buffers, and slow MS sampling rates can limit throughput advantages that such next-generation media offer. High-throughput LC–MS methods of different relevant pharmaceutical and environmental mixtures were developed using ultrahigh performance core-shell media. Such methods were developed using standard HPLC systems and back pressures, showing the ease and utility of using core-shell media for increasing throughput of LC–MS methods.
Coupling mass spectrometry (MS) to high performance liquid chromatography (HPLC) methods has become commonplace over the last two decades as many take advantage of both the sensitivity and the identity aspects of such instrumentation. The specificity of tandem MS using parent–daughter combinations (multiple reaction monitoring [MRM], for example) allows for quantifying individual compounds in a complex mixture, even when such compounds are not fully resolved from other analytes in a mixture. However, some resolution between analytes and ion-suppressing impurities is required for good MS quantitation. Combining shorter HPLC columns with MS has been the solution for developing sensitive methods for quantitating several analytes in a single analysis.
Over the last five years, several instrument and column manufacturers have invested much in marketing and advertising about ultrahigh-pressure LC (UHPLC) and the advantages of switching to higher efficiency columns for analytical separations. Showing performance gains for HPLC applications using UV detection by switching to UHPLC columns and instruments has been a rather straightforward process. Sampling rates of newer UV detectors are such that a large number of data points per second can be acquired, allowing one to visualize and quantitate the improved chromatography that UHPLC columns deliver. With LC–MS methods, visualizing improvements in chromatography are less apparent. The scan rate for MS instrumentation can sometimes be limited to a few points per second. This rate can be even slower for methods that scan large molecular weight ranges or when numerous parent–daughter pairs are being acquired. In addition, extracolumn volume in the MS interface or detectors postcolumn can mask any separation gain from a UHPLC column. As a result, realized performance gains for LC–MS methods using UHPLC have been significantly less than standalone LC methods.
Recently, a new type of UHPLC column has been introduced using core-shell media. These media use unique particle geometry versus the standard fully porous silica media; a nonporous core particle is surrounded by a thin porous layer of functionally bonded silica. The improved porosity and mass transfer of such media are much higher than that of fully porous media of the same particle size, resulting in performance on par with fully porous media much smaller than the core-shell media. In the case of the major core-shell products on the market, the core-shell media are slightly larger than 2.5 µm in size with performance on par with the major sub-2-µm media on the market with back pressures more in line with 3-µm fully porous HPLC media. The greater implication of such core-shell media is that one could get UHPLC performance on their existing HPLC system without having to buy a new ultrahigh pressure system.
Efforts were undertaken to adapt various existing LC–MS methods using standard HPLC systems to using the new core-shell media. Examples of high-throughput applications for pharmaceutical and environmental analysis were developed using core shell media. Key in developing such methods include maintaining reasonable resolution of key analytes from ion suppressing impurities while balancing the limitations of the MS being used for the analysis.
Materials and Methods
Chromatography standards (amphetamine, aflatoxin, sulfa drugs, and antibiotic mixtures), urine, and plasma were purchased from Sigma Chemical (St. Louis, Missouri). Various buffers and solvents were purchased from EMD (San Diego, California). Gemini 5 µm C18 (150 mm × 3.0 mm) and Kinetex 2.6 µm C18 and PFP (both 50 mm × 2.1 mm) HPLC columns were obtained from Phenomenex (Torrance, California). StrataX, StrataX-C, and Strata Florisil solid-phase extraction (SPE) tubes (50 mg/3 mL tubes) also were used. Food matrices were obtained from local grocery sources. Test samples were spiked into separate sample matrices. For the pharmaceutical–toxicology sample, amphetamines were spiked into urine. For environmental analysis, aflatoxin was spiked into peanut butter, sulfa drugs in honey, and an antibiotic mixture was spiked into a meat sample. While every SPE procedure was unique to the application, all had several common features: sample is diluted then loaded on their specific SPE cartridge; cartridge is washed, then analyte is eluted with an organic mixture. Samples are then dried down and reconstituted in aqueous mobile phase before injection on HPLC.

Figure 1
All samples were injected on an HP1100 HPLC system with an autosampler, solvent degasser, and multiwavelength detector (all from Agilent Technologies, Santa Clara, California). HPLC-UV data were collected using Chemstation software (Agilent Technologies). The aqueous mobile phase used was either 0.1% formic acid in water or a mixture of formic acid with ammonium formate. For organic mobile phase, either acetonitrile or methanol was used. Various gradient methods were used based upon the analyte mixture and columns used. For LC–MS analysis, the HPLC instrument was linked postcolumn (excluding the UV detector) directly to the electrospray ionization (ESI) interface of an API3000 tandem mass spectrometer (Applied Biosystems, Foster City, California). While individual MS parameters differ based upon the application, in most cases, the MS system was run in positive ion mode using MS-MS multiple reaction monitoring, in which analytes were quantitated using specific parent– daughter ion transitions. In many cases, a multiply deuterated internal standard was used for quantitation.
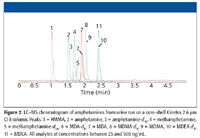
Figure 2
Results and Discussion
The promise of UHPLC has been to shorten existing HPLC methods using 3-µm and 5-µm media up to 10-fold or more using new UHPLC systems and associated sub-2-µm media. While such dramatic increases in throughput are conceivable for methods using UV detection, for LC–MS detection, one is limited by the sampling rate of the MS detector. Depending upon the dwell time needed for sensitivity and number of transitions being monitored, one can potentially be limited to only a two- to threefold decrease in analysis time simply because the mass spectrometer might not be able to collect enough data points across an analyte peak for accurate quantitation. This has become more readily apparent with the introduction of core-shell media with performance on par with sub-2-µm columns while operating at standard HPLC back pressures. Such versatility allows for increased throughput with existing LC–MS system without having to purchase a new system that can tolerate higher backpressures. An example of this is shown in Figure 1 and Figure 2. In Figure 1, a mixture of amphetamines was run on a fully porous 5-µm C18 column (150 mm × 3.0 mm) using an 8-min run time with good resolution between individual components (the d5 internal standards obviously are eluted closely to their natural counterparts). Transferring the method to using a core-shell media column (Kinetex 2.6 µm C18, 50 mm × 2.0 mm, Figure 2), both the column and gradient time are shortened resulting in an analysis time around 3 min, even with the additional analytes in the mixture. The increased efficiency (plates/m) of the core-shell column allows for shortening column length while maintaining resolution; the van Deemter plot for 2.6-µm core-shell columns more closely resemble the plot for a 1.7-µm fully porous media, resulting in better performance at higher mobile phase linear velocities.

Figure 3
A similar example of the utility of using core-shell columns to decrease the run time of an existing LC–MS method is shown in Figure 3 and Figure 4. In this case, a sulfa drug mixture is extracted from honey using SPE and injected on a fully porous 5-µm C18 column (150 mm × 3.0 mm) with a run time of 10 min (Figure 3). The gradient and column are again shortened (a 2.5-min gradient on a core-shell Kinetex 2.6 µm C18 column 50 mm × 2.1 mm) with concurrent increase in mobile phase linear velocity (Figure 4). Similar to what was shown in the previous example, the analysis time was decreased around threefold, and critical resolution between critical components is maintained without having to use a specialized UHPLC system. In addition to the platform compatibility that lower operating back pressure of core-shell media provides, one could conceivably operate at even higher linear velocities to further reduce run times. However, flow rates must be balanced with the sampling rate of the mass spectrometer. If peak widths are narrowed excessively by faster separation methods, then peak shape distortion will occur, resulting in poor method quantitation. In Figure 4, this is only slightly apparent as negative dips due to peak smoothing. In Figure 5, peak distortion is slightly more noticeable. Figure 5 is an example of an aflatoxin LC–MS method using a core-shell Kinetex 2.6 µ PFP column (50 mm × 2.1 mm). Pentafluorophenol phases are particularly popular for separation of geometrically unique polyaromatic analytes. In the case of microtoxins, PFP phases do a particularly good job at separating different aflatoxins. In this LC–MS example, all the major aflatoxins are well separated in under 2 min with good quantitation at the 50-ng level, however, very slight distortion is observed due to a slightly low sampling rate of the mass spectrometer. Such results emphasize the balance needed in developing LC–MS methods using core-shell and other high performance media: analysis speed, analyte sensitivity, and quantitation all must be factored into an LC–MS method based upon the capabilities of the mass spectrometer. The use of a PFP column for this separation also emphasizes the utility of using alternate selectivities for LC–MS applications. The use of a C18 column for this application would require a much longer analysis time because of the limited selectivity that C18 has for aflatoxins. For many applications, an alternate selectivity media can allow for shorter run times due to their greater selectivity for specific compound mixtures.
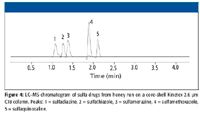
Figure 4
While there can be limitations on a high-speed LC–MS method based upon the instrument platform used, Figure 6 shows an example of the tremendous power of LC–MS analysis. In this example, a mixture of 30 different antibiotics are extracted from a meat sample by SPE and run on a core-shell Kinetex 2.6 µm C18 column (50 mm × 2.1 mm) and individual MRM traces are overlaid. The 30 different analytes are partially resolved in a gradient of 8 min, and either positive or negative ion mode was used depending upon the analyte being detected. Because of the large number of analytes and the slow MS sampling rate, it is obvious that the peak shape of several analytes are distorted significantly, resulting in compromised quantitation. In addition, coeluted analytes can potentially suppress the electrospray efficiency, resulting in further inaccuracies in quantitation. However, such a broad method can be very effective for widespread detection of analytes in which exact quantitation is not necessary.
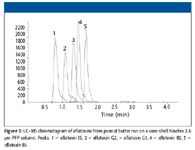
Figure 5
Conclusion
UHPLC media have allowed for greatly reduced run times, resulting in significant improvements in throughput for analytical methods. These improvements have been especially profound when using core-shell media, in which improved throughput and performance can be realized with existing HPLC systems without the need for dedicated ultrahigh-pressure instruments. While adapting existing HPLC methods can be fairly easy, with LC–MS methods, the specific limitations of the mass spectrometer being used must be considered. For complex MS acquisition programs, sampling rates can approach the peak widths of some analytes separated on a core-shell column. In such cases, peaks can be distorted, resulting in poor quantitation. As a result, one might not be able to completely realize the full benefit in throughput that core-shell columns provide for LC–UV methods. With a balanced view of scan rates, sensitivity requirements, and chromatographic peak widths, one can develop rapid and accurate LC–MS methods using core-shell and other UHPLC media. In addition, the different phases of core shell media (C18, PFP, or hydrophilic interaction chromatography) offer selectivity options that should be considered when developing optimal high-speed LC–MS methods.

Figure 6
Michael McGinley and Carl Sanchez are with Phenomenex, Inc., Torrance, California.

Accelerating Monoclonal Antibody Quality Control: The Role of LC–MS in Upstream Bioprocessing
This study highlights the promising potential of LC–MS as a powerful tool for mAb quality control within the context of upstream processing.
Using GC-MS to Measure Improvement Efforts to TNT-Contaminated Soil
April 29th 2025Researchers developing a plant microbial consortium that can repair in-situ high concentration TNT (1434 mg/kg) contaminated soil, as well as overcome the limitations of previous studies that only focused on simulated pollution, used untargeted metabolone gas chromatography-mass spectrometry (GC-MS) to measure their success.
Prioritizing Non-Target Screening in LC–HRMS Environmental Sample Analysis
April 28th 2025When analyzing samples using liquid chromatography–high-resolution mass spectrometry, there are various ways the processes can be improved. Researchers created new methods for prioritizing these strategies.
Potential Obstacles in Chromatographic Analyses Distinguishing Marijuana from Hemp
April 28th 2025LCGC International's April series for National Cannabis Awareness Month concludes with a discussion with Walter B. Wilson from the National Institute of Standard and Technology’s (NIST’s) Chemical Sciences Division regarding recent research his team conducted investigating chromatographic interferences that can potentially inflate the levels of Δ9-THC in Cannabis sativa plant samples, and possible solutions to avoid this problem.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)










