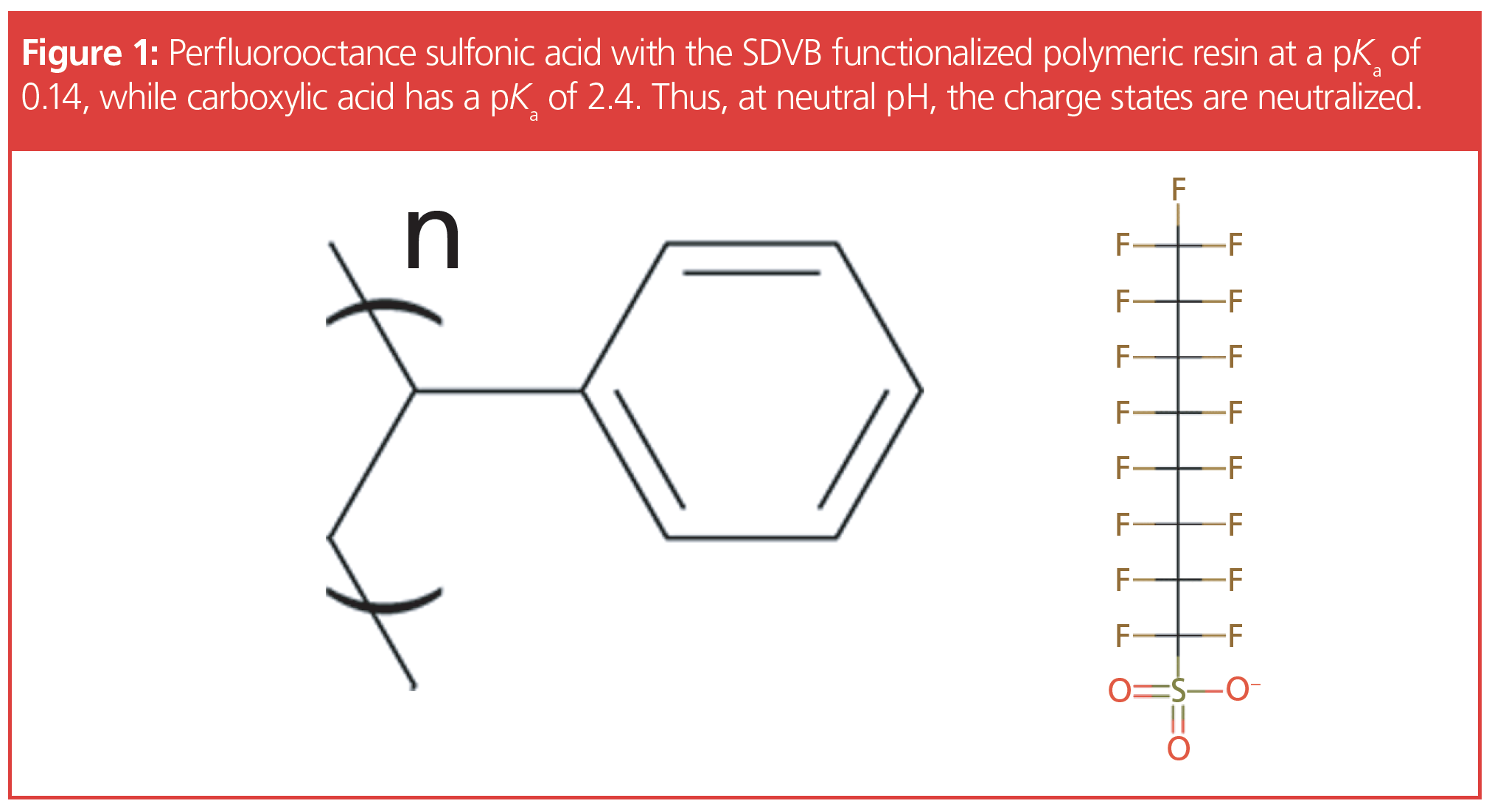
The Evolving Developments in Sample Preparation for PFAS in Environmental Samples
The ever-increasing knowledge concerning the toxicity, global scope, broad usage, and indestructible nature of per- and polyfluorinated alkyl substances (PFAS) has created a demand for extracting these compounds from many different matrices. In many cases, extraction is insufficient and additional matrix cleanup steps are required. This overview covers sample preparation primarily for water samples.
The description and properties of per- and polyfluorinated substances (PFAS) have recently become a widely researched topic with many intricacies. PFAS owe their persistence to the carbon-fluorine bond, which is one of the shortest and strongest chemical bonds and makes these chemicals highly resistant to breakdown in the environment, earning them the nickname “forever chemicals”. The fluorinated carbon chains varying in length for C1 to C18 instill hydrophobic properties, while the acidic end groups impart a hydrophilic character, making these chemicals a unique chemical composition. The wide variety and complexity of PFAS compounds has resulted in many PFAS groups, including carboxylic acids, sulfonic acids, ethers, chlorinated, sulfonamides, alcohols, and telomers. What these all have in common is the fluorination and varying degrees of hydrophobicity/hydrophilicity.
Separation and Detection
The most common method for detecting the presence of PFAS in a sample uses a reversed‑phase liquid chromatography (LC) separation followed by tandem mass spectrometry (MS/MS) detection. Gas chromatography (GC)–MS/MS methods have also been published, and some methods even recommend ion chromatography (IC). Using mass spectrometry combines the need for quantitation, confirmation, and sensitivity that is required for low detection and matrix interferences. LC column phases that are often used include C8, C18, phenyl-hexyl, and F5; in addition to many peer-reviewed articles, the US. Dept. Of Agriculture, EPA Office of Ground Water and Drinking Water, the EPA Office of Water, the Department of Defense, US FDA, and ISO have all published methods using a combination of these columns. To date, most have migrated towards a C18 approach for good resolution and reliability. While analysis may be relatively straightforward for PFAS detection, the cleanup using sample preparation has evolved and continues to be an area of optimization for environmental laboratories.
The Evolving Sample Preparation for PFAS Compounds
There are several methods developed by government agencies that utilize solid-phase extraction (SPE) followed by LC–MS/MS detection. EPA Method 537, originally drafted in 2009 and updated to EPA 537.1 in 2020 (1), involves preconcentration of a water sample on a polystyrenedivinylbenzene (SDVB) cartridge followed by methanol extraction. These methods rely on the hydrophobicity of PFAS to interact with the SDVB by cartridge conditioning and extraction with methanol.
As can be seen in Figure 1, an SPE strategy that relies on a hydrophobic approach alone does not take into account the complete chemical structure of the majority of PFAS compounds. In addition, scientists that tried to identify shorter chain PFAS, such as perfluorobutanoic acid (C4) and PFAS precursors, such as hexafluoropropylene oxide dimer acid (HFPO-DA), met with poor recoveries, which is not surprising since these shorter length compounds would have a lower affinity for a hydrophobic interaction. For this reason, the EPA Office of Ground Water and Drinking developed EPA Method 533 (2,3), which states in the disclaimer: “This analytical method may support a variety of monitoring applications, which include the analysis of multiple short-chain per- and polyfluoroalkyl substances (PFAS) that cannot be measured by Method 537.1.”
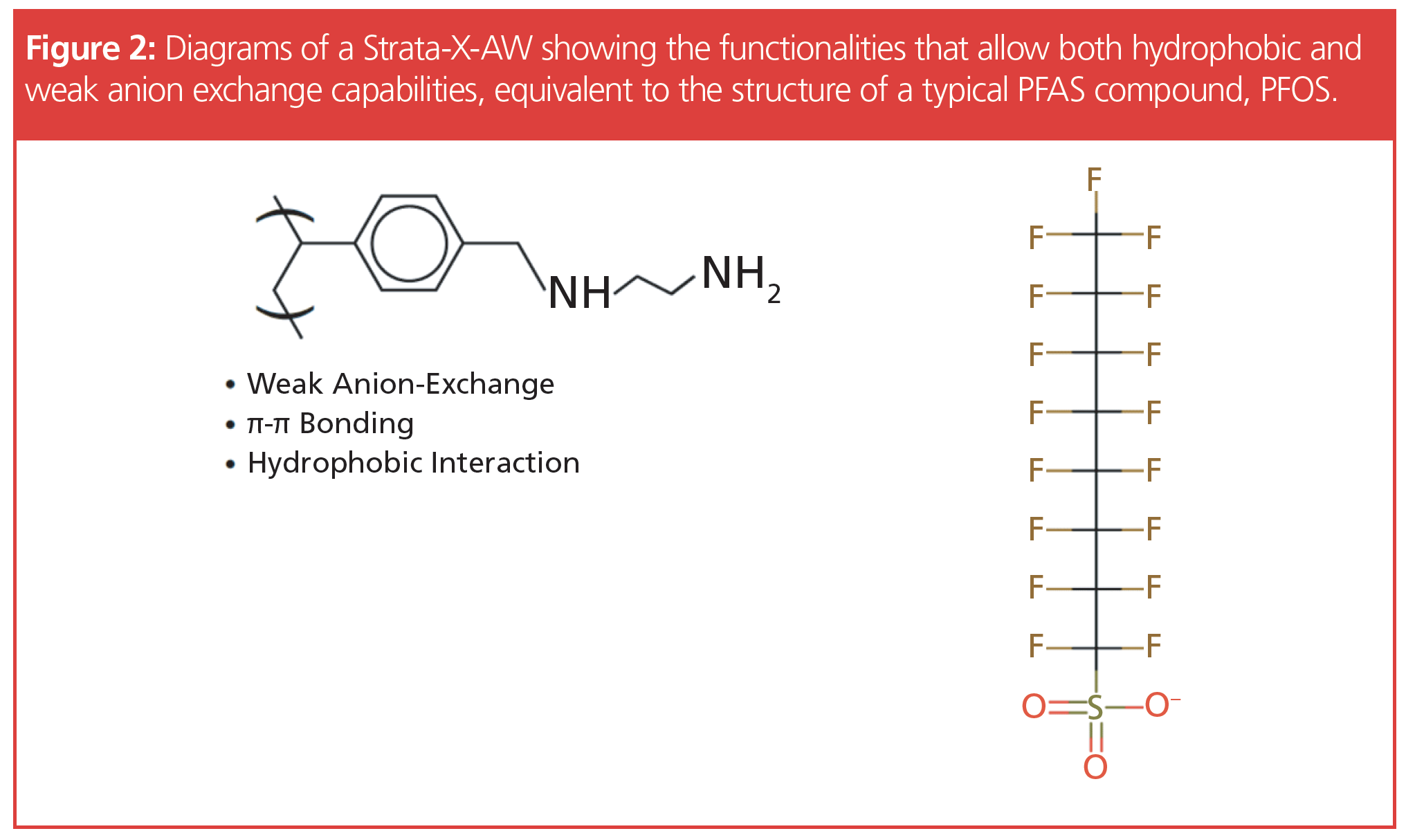
However, this method is virtually identical to EPA 537.1: “The sample is passed through an SPE cartridge containing polystyrene divinylbenzene with a positively charged diamino ligand to extract the method analytes and isotope dilution analogues. The cartridge is rinsed with sequential washes of aqueous ammonium acetate followed by methanol, then the compounds are eluted from the solid phase sorbent with methanol containing ammonium hydroxide”. Figure 2 shows the functionality for a WAX SPE. The diamino cation has a selective affinity for anionic compounds such as PFAS required for short-chain extraction efficiency. This format also has a hydrophobic interaction that when combined is similar to the structure of the PFAS molecules.
Combined, EPA methods 537.1 and 533 allow 29 PFAS compounds to be measured in drinking water. The data concerning the requirements for quality control and method performance are documented in the methods and not a part of the scope here. The precision and accuracy shown are within 10% for the majority of the analytes at the lower spike, 10 ppt levels, except for the short-chain (C4) PFBS.
PFAS Methods and Sample Preparation for Non-Drinking Water Matrices
Simultaneously, while these methods were being developed and validated, additional testing needs arose for non-drinking water matrices and the continued increase in PFAS compounds being identified. In order to address these issues, many laboratories turned to the Department of Defense Quality Systems Manual (QSM) as a starting point (4). This document realizes the utility of weak anion exchange (WAX), not SDVB, for the SPE material, and addresses several issues, including the baseline requirements for dirtier matrices along with many of the traditional method aspects such as quality assurance/quality control (QA/QC) criteria. The EPA’s Office of Water, in partnership with the Department of Defense’s (DoD) Strategic Environmental Research and Development Program, published a single‑laboratory validated method in August 2021 (5). This method targets 40 PFAS compounds for wastewater, surface water, groundwater, soil, biosolids, sediment, landfill leachate, and fish tissue. It can be incorporated into various applications, including National Pollutant Discharge Elimination System (NPDES) permits. Both the DOD QSM and EPA 1633 (6) mention a requirement for the use of graphitized carbon black (GCB) in a dispersive (powder) or cartridge format prior to injection into the LC–MS/MS system. Each sample and associated QC sample require a cleanup, thus requiring a two-step SPE process that adds time, cost, and chances for error. The purpose of the additional SPE cleanup step is to eliminate matrix that can cause interference and reduce bias. GCB has been shown to remove organic acids (such as humic and cholic acids), which can suppress ionization (TDCA and PFOS). However there is an issue with GCB and as stated in the Draft EPA Method 1633, “…It is important to minimize the time the sample extract is in contact with the carbon.” The draft EPA Method 1633 was developed in a joint collaborative effort between the US Department of Defense and the US EPA.
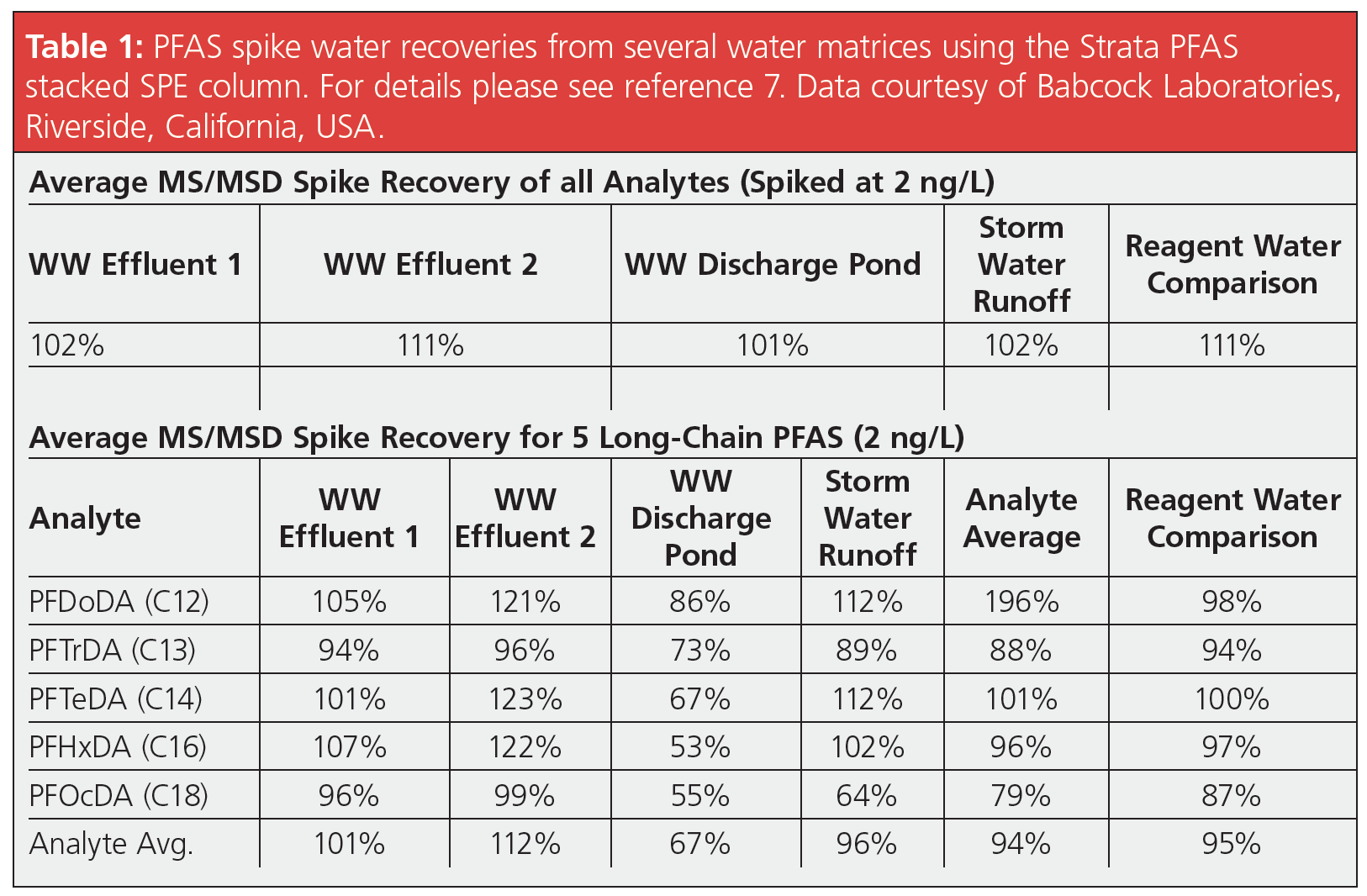
Figure 3 shows the SPE cleanup cartridges applicable to EPA 1633 that require a WAX and a GCB cleaning step. A dual cartridge has been developed to simplify the extraction and cleanup in one step. Several laboratories have demonstrated that instead of a two-step SPE method, a single cartridge containing WAX/GCB in a stacked format is more than sufficient and achieves similar recoveries than a costly two-step process (7,8,9). Table 1 shows the average spike recoveries of 42 PFAS analytes at 2 ng/L for five water matrices after using a WAX/GCB stacked cartridge. The recoveries are excellent, from the short- (C4) to the long-chain (C18) PFAS compounds. If we look at the more long-chain compounds in isolation, there are still excellent recoveries. The dual WAX-hydrophobic interaction on the resin backbone was sufficient for optimal retention and the GCB did not interfere with recovery. Results showed that there was no difference in the spike recoveries between the dual and single SPE approaches, yet the dual cartridge saved significant time and cost. PFAS accuracy and precision results obtained using the single tube sample preparation procedure were equivalent to those obtained using the two-step WAX + a dispersive or cartridge GCB clean-up step.

WAX/GCB Single Cartridge SPE for Longer Chain PFAS Compounds?
In July 2021 the California Water Resources Control Board announced the order for “Sections 13267 and 13383 order for the determination of the presence of per- and polyfluoroalkyl substances at publicly owned treatment works order WQ 2020-0015-DWQ“ (10). This announcement specified the requirement for public-owned treatment works (POTWs) to monitor an extended list of PFAS compounds in wastewater discharges. Excellent recoveries of over 40 PFAS compounds, ranging in length from C4 to C18, from over four different water matrices, including wastewater influents, holding ponds, and discharge waters, have been demonstrated (6).
Conclusion
Strategies for sample preparation have evolved to address the increased number of PFAS that need to be analyzed from a wide variety of matrices. They have taken into account the analyte of interest, interfering matrices, as well as the need for simple, less costly steps to achieve accurate quantitation. This brief outline also highlights that these strategies can be incorporated into validated methods for compliance monitoring.
References
- EPA Method 537.1, Determination of Selected Per- and Polyfluorinated Alkyl Substances in Drinking Water by Solid Phase Extraction and Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS) Version 2.0 March 2020: https://www.epa.gov/dwanalyticalmethods/approved-drinking-water-analytical-methods
- EPA Method 533, Determination of Per- and Polyfluoroalkyl Substances in Drinking Water by Isotope Dilution Anion Exchange Solid Phase Extraction and Liquid Chromatography/Tandem Mass Spectrometry: https://www.epa.gov/dwanalyticalmethods/approved-drinking-water-analytical-methods
- Phenomenex Technote 0141, EPA Method 533, PFAS in Drinking Water (Torrance, California, USA).
- Department of Defense (DoD) Department of Energy (DOE) Consolidated Quality Systems Manual (QSM) for Environmental Laboratories, DoD Quality Systems Manual Version 5.4, 2021.
- EPA Announces First Validated Laboratory Method to Test for PFAS in Wastewater, Surface Water, Groundwater, Soils, 2 September 2011: https://www.epa.gov/newsreleases/epa-announces-first-validated-laboratory-method-test-pfas-wastewater-surface-water
- Draft Method EPA 1633, Analysis of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous, Solid, Biosolids, and Tissue Samples by LC-MS/MS.
- Phenomenex Technote TN-0144, Method Validation of a C4-C18 PFAS Panel from Water Extracts Using DualMode SPE followed by LC-MS/MS (Torrance, California, USA).
- Phenomenex Technote, TN74410520_W Per- and Polyfluoroalkyl Substances (PFAS) Extraction by LC-MS/MS Using Strata® PFAS for a Stacked Solid Phase Extraction (SPE) Solution (Torrance, California, USA).
- Phenomenex Technote 0145, Comparison of PFAS Recoveries Between Cartridge Format WAX/GCB vs. Dispersive GCB for DoD Compliance (Torrance, California, USA).
- APPROVAL TO EXTEND DEADLINES FOR SAMPLING AND REPORTING OF PERAND POLYFLUOROALKYL SUBSTANCES (PFAS) AT PUBLICLY OWNED TREATMENT WORKS SUBJECT TO THE JULY 9, 2020 STATE WATER RESOURCES CONTROL BOARD ORDER # WQ 2020-0015-DWQ, 24 November 2020: https://www.waterboards.ca.gov/pfas/docs/11242020_signed_es_pfas_potw_scap_extension_lt.pdf
Richard Jack is Global Market Development Manager for the food, cannabis, and environmental markets with Phenomenex Corporation. Richard has been in the analytical industry for many years as a product manager, and in market development with several companies. He has been active in global regulatory involvement and participated in several method validation projects. His participation with the US EPA Office of Ground Water and Drinking Water resulted in coauthoring EPA methods 557; 557.1. Richard is the second vice-chair for the ASTM D19 water committee and drafted several ASTM methods including D4327 and D6919. Richard has an M.S. in ecology and a Ph.D.
in biochemistry.
E-mail: richardj@phenomenex.com
Website: www.phenomenex.com/pfas

University of Rouen-Normandy Scientists Explore Eco-Friendly Sampling Approach for GC-HRMS
April 17th 2025Root exudates—substances secreted by living plant roots—are challenging to sample, as they are typically extracted using artificial devices and can vary widely in both quantity and composition across plant species.
Sorbonne Researchers Develop Miniaturized GC Detector for VOC Analysis
April 16th 2025A team of scientists from the Paris university developed and optimized MAVERIC, a miniaturized and autonomous gas chromatography (GC) system coupled to a nano-gravimetric detector (NGD) based on a NEMS (nano-electromechanical-system) resonator.
Characterizing Plant Polysaccharides Using Size-Exclusion Chromatography
April 4th 2025With green chemistry becoming more standardized, Leena Pitkänen of Aalto University analyzed how useful size-exclusion chromatography (SEC) and asymmetric flow field-flow fractionation (AF4) could be in characterizing plant polysaccharides.
















