Real-Time Response to Bacteria Infection of Bioethanol Fermentation Using a Short Rezex™ ROA Column
The Application Notebook
Political priorities as well as economic interests have fueled a dramatic growth in the biofuel industry, due much in part to research funding and tax incentives. Currently the world's ethanol production is estimated to be over 16 billion gallons a year. This number is expected to increase in the next few years, reaching an estimated yearly production of 20 billion gallons by 2012. Today, many ethanol producers add fermentors to expand their production capacity. In order to continue using existing HPLC equipment for the increased monitoring, increased analytical throughput is needed.
Michael McGinley*, Ngoc Nguyen*, and Jim Mott**
*Phenomenex, Inc., and **Shimadzu Scientific Instruments Inc.
Political priorities as well as economic interests have fueled a dramatic growth in the biofuel industry, due much in part to research funding and tax incentives. Currently the world's ethanol production is estimated to be over 16 billion gallons a year. This number is expected to increase in the next few years, reaching an estimated yearly production of 20 billion gallons by 2012. Today, many ethanol producers add fermentors to expand their production capacity. In order to continue using existing HPLC equipment for the increased monitoring, increased analytical throughput is needed.
The current standard analytical procedure for monitoring ethanol production uses an ion exclusion column, such as the Phenomenex Rezex ROA, 300 × 7.8 mm. However, several plants have begun to use the shorter Rezex ROA 150 × 7.8 mm column in place of, or in addition to, the Rezex ROA 300 × 7.8 mm column. The shorter column increases throughput by reducing the analysis time by 50% (from 24 to 12 min run time). In this article, the advantages and disadvantages of using a shorter Rezex ROA will be discussed.
Materials and Methods
Crude samples of various bioethanol fermentation timepoints, generously provided from ICM (Colwich, Kansas), were filtered using a 0.20 μm Phenex™-RC syringe tip filter (Phenomenex, Torrance, California). Ethanol HPLC testing standard was obtained from Midland Scientific (Omaha, Nebraska). Filtered aliquots of 10 μL were injected on an HPLC operating at a flow rate of 0.6 mL/min and the HPLC column was heated to 65°C. SecurityGuard cartridges were regularly changed every 100 runs or whenever static backpressure increased 10% above initial backpressure values. 50% methanol was used in the autosampler needle wash to avoid bacterial contamination.
All analyses were run on a Shimadzu LC-20AT LC system (Shimadzu Scientific Instruments, Columbia, Maryland) equipped with a SIL-10AF autosampler, degasser, and a RID-10A RI detector; data was collected using CLASS-VP Version 7 Software. Two dimensions of Rezex ROA columns (Phenomenex Inc., Torrance, California) were used: 150 × 7.8 mm and 300 × 7.8 mm. SecurityGuard99 guard column and Carbo-H+ 4 × 3.0 mm cartridges (Phenomenex, Torrance, California) were used to prolong the Rezex column life. Aqueous mobile phase (0.005 N Sulfuric Acid in water) was purchased from CHATA (Fort Collins Colorado).
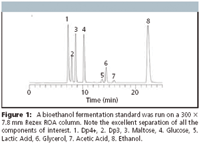
Figure 1
Results and Discussion
Figure 1 shows the HPLC run of a fermentation standard using the Rezex ROA 300 × 7.8 mm column. Baseline resolution is achieved for all peaks in about 24 min. Runs of two bioethanol fermentation samples at the 18 and 39 h timepoints were done using the 300 × 7.8 mm column (Figure 2). These runs show that early in the fermentation process the oligosaccharide and saccharide peaks are very abundant. Later in the production, as sugars are converted into ethanol, the oligosaccharide and saccharides are depleted as the concentration of ethanol, glycerol, and organic acid peaks increases.

Figure 2
As some producers have increased the scale of their ethanol production and may now be operating several fermentors in parallel, there is an interest in reducing the analysis time for fermentation monitoring. Quick analysis of the production process would not only increase throughput, but would help prevent the loss of an entire fermentation batch due to a bacteria infection. Lactic and acetic acid are byproducts produced by contaminated bacteria and these acids can inhibit or kill the yeast that converts the sugar to ethanol. Because these acids may potentially have detrimental effects on the fermentation process, producers closely monitor their levels throughout the entire production process. Regular monitoring of the process by HPLC allows operators to assess if microbial contamination is affecting the fermentation process and if remediation steps such as antibiotic addition are necessary to maximize ethanol yield (1).
One method for reducing HPLC run time is by reducing the length of the Rezex column used. In Figure 3, the fermentation standard is run on a Rezex ROA 150 × 7.8 mm (half the length of the typical 300 mm column used). As expected, the run time using a shorter column is significantly reduced from 24 to 12 min. Compared to the 300 mm column, the resolution of the early eluting oligosaccharide and saccharide peaks is reduced. However, baseline resolution is still achieved for the late eluting organic acids (lactic acid and acetic acid), glycerol, and alcohol peaks.
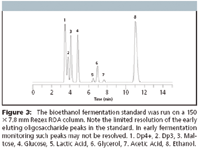
Figure 3
Using the Rezex ROA 150 × 7.8 mm column will increase sample throughput two-fold, producing faster results that allow for more real-time responses. In addition, the shorter column length utilizes less solvent. With most producers operating around the clock, this will significantly help to lower solvent usages and production cost.
Choosing optimal Rezex ROA column length is dependent on whether it is critical to have an accurate quantitation of the Dp4+ and Dp3 peaks. Since the separation of oligosaccharides is based primarily on a gel filtration mechanism, there is a limitation on how much the column length can be shortened and still maintain resolution of key saccharide peaks. The early eluting peaks (Dp4+, Dp3, Maltose, Glucose) represent the different degrees of polymerization of the various saccharides present in the sample. Monitoring of these peaks during early time points of the fermentation run gives operators a good indication as to the progression of the various amylases used to break down starches to simple sugars, and dictate when yeast is added to the fermentor to start generating ethanol.
Producers that focus on the later eluting peaks (Lactic acid, Glycerol, Acetic acid, Ethanol) are looking for an indication of the fermentation endpoint, as well as when bacterial contamination is severe enough to warrant addition of an antibiotic to limit bacterial by-products that may inhibit ethanol production. This is still achieved when using the shorter Rezex ROA 150 × 7.8 mm column (Figures 3 and 4).
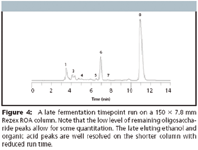
Figure 4
Conclusion
The standard HPLC column used for fermentation monitoring of bioethanol production is a Rezex ROA 300 × 7.8 mm column. This column is good for monitoring saccharides, organic acids, and alcohols generated during bioethanol production all in one run. However, with the increase of bioethanol production, producers may consider using the shorter Rezex ROA 150 × 7.8 mm column to reduce analysis time and to allow for more real-time responses of bacteria infection.
References
(1) "Using the HPLC System in a Fuel Grade Ethanol Production Laboratory" J.Mott, Shimadzu Scientific Application Note, 2006.
(2) "HPLC of Carbohydrates with Cation- and Anion-Exchange Silica and Resin-Based Stationary Phases" C.G.Huber and G.K. Bonn in Carbohydrate Analysis; Journal of Chromatography Library, 1995, Vol. 58, 147–180.

Phenomenex, Inc.
411 Madrid Ave.
Torrance, CA 90501
(310) 212-0555; (310) 328-7768
















