Making Method Development Faster for the Analysis of Natural and Artificial Flavourings
The Column
A simple, rapid, and robust ultrahigh-performance liquid chromatography (UHPLC) method has been developed for the simultaneous determination of natural and artificial vanilla flavouring substances as well as some precursors has been developed using an automated method scouting or method optimization workflow. The most suitable mobile phase and stationary phase combination was identified in a scouting run. These conditions were used to create a two-dimensional model in computer simulation software. Temperature and gradient time were varied to establish the optimum fast and robust separation conditions. This approach resulted in a 5.5 min gradient method that allowed for fast screening of 11 compounds of interest.
Photo Credit: amriphoto/Getty Images

A simple, rapid, and robust ultrahigh-performance liquid chromatography (UHPLC) method has been developed for the simultaneous determination of natural and artificial vanilla flavouring substances as well as some precursors has been developed using an automated method scouting or method optimization workflow. The most suitable mobile phase and stationary phase combination was identified in a scouting run. These conditions were used to create a two-dimensional model in computer simulation software. Temperature and gradient time were varied to establish the optimum fast and robust separation conditions. This approach resulted in a 5.5 min gradient method that allowed for fast screening of 11 compounds of interest.
Vanilla is one of the most important flavours worldwide and is widely used in foods, beverages, and perfumes. Natural vanilla extract contains up to several hundred substances with vanillin, vanillic acid, 4-hydroxybenzoic acid, and 4-hydroxybenzaldehyde the major components. As a result of continuously increasing demand and the resulting high costs of natural vanilla extracts, artificial flavourings are often used instead. Vanillin can be obtained through various methods such as chemical synthesis, biotransformation, or degradation of waste sulphite liquors, as well as extraction of natural vanilla pods. These artificial flavours can contain synthetic vanillin, ethyl vanillin, eugenol, guaiacol, vanillin mandelic acid, and others.
As authenticity criteria for vanilla, the ratios of the major components vanillin, 4-hydroxybenzaldehyde, vanillic acid, and 4-hydroxybenzoic acid are frequently used. In order to monitor the composition and therefore quality of vanilla flavours contained in food, an analytical method needs to enable individual quantification of any of these ingredients as well as possible ingredients like the precursors from the synthesis of vanillin.
The article describes the computer-assisted development and optimization of a rapid ultrahigh-performance liquid chromatography (UHPLC) screening test for the separation and quantification of natural and artificial vanilla flavouring substances as well as some precursors for the quality control of vanilla products. This article illustrates the effectiveness and speed of software-assisted method development tools.
Experimental
Equipment and Chromatographic Methods: For UHPLC method scouting, a Nexera X2 Method Scouting System (Shimadzu) was used, consisting of two quaternary solvent pumps, autosampler, and column oven including a six-column switching valve. The system was also equipped with a high resolution photo diode array detector. Acetonitrile and water + 10 mM HCOONH4 (pH 2.8) were used as mobile phase. The different stationary phases applied for method scouting for the separation of 11 analytes relevant to vanilla products are displayed in Table 1.
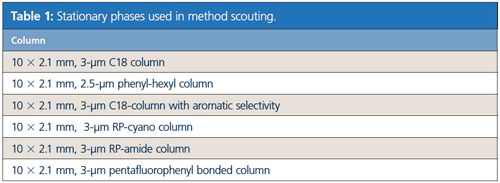
Method scouting was performed in a sequence using 7.5 min gradient runs at 40 °C with combinations of organic and aqueous mobile phases (acetonitrile/H2O + 10 mM HCOONH4; gradient: 5–95%) on the six different columns, which cover a wide spectra of diverse stationary phase materials for reversed phase chromatography.
The phenyl-hexyl-column showed the most promising results with all analytes of interest at least partly separated using a mobile phase consisting of A: 10 mM ammonium formate in H2O (pH 2.8) and B: acetonitrile. These conditions were used to create a two-dimensional DryLab model (Molnár Institute) using 2 min and 6 min gradient runs at 25 °C and 50 °C as input data. These experiments resulted in a colour-coded resolution map for simple identification of the optimum separation conditions (Figure 1). The figure shows the calculated resolution (colour-coded) for the different simulated combinations of retention time (x-axis) and oven temperature (y-axis).
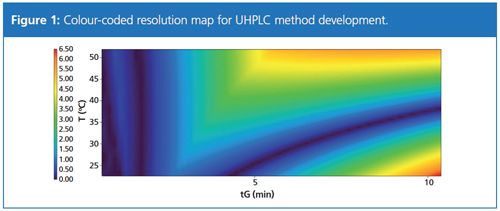
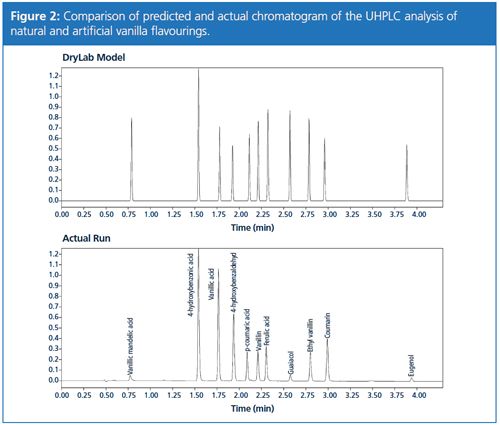
The software predicted an optimum separation with a minimum resolution of the critical peak pair of 2.1 in a gradient run from 5% to 70% B in 5.5 min at a flow rate of 0.5 mL/min at 50 °C. A comparison of the predicted chromatogram with an actual sample run is displayed in Figure 2.
The advantage of a computer-assisted method development approach compared to a more traditional one lies in the time savings because four gradient runs (see above) are enough input for the software to calculate the optimal conditions (gradient, oven temperature) for the method. This can mean lower costs because of fewer working hours and smaller solvent consumption. However, there are some limitations such as when developing a method with a two- (or more) step gradient. In such cases the simulations are not that precise as those with a linear gradient. This is because the data for the calculation are from a linear gradient run and a computer simulation is more precise the more similar the conditions from the input data and the simulation are.
Conclusion
A robust, fast, and sensitive UHPLC method for the simultaneous separation of natural and artificial vanilla flavouring substances as well as some precursors has been developed. The method scouting experiment and further optimization using computer simulation software was able to save time and offered visualization of the design space in a resolution map to establish the most robust separation method.
Philipp Jochems graduated in 2013 with a master of science in applied chemistry from the University of Applied Science in Krefeld, Germany. In 2014 he was a scientific assistant at the University Medical Center of the Johannes Gutenberg University Mainz, Germany, and since 2016 has worked as a HPLC product specialist in the analytical business unit of Shimadzu Europa in Duisburg, Germany.
Gesa Schad graduated with a diploma in chemical engineering from the Technical University, NTA in Isny, Germany, in 2004 and as a master of science in pharmaceutical analysis from the University of Strathclyde in Glasgow, UK, in 2005. Until 2006 she worked as a consultant in HPLC method development and validation in an analytical laboratory of the FAO/IAEA in Vienna, Austria. She gained her doctorate for research in pharmaceutical sciences at the University of Strathclyde in 2010 and was employed as an HPLC specialist in the R&D department at Hichrom Ltd. in Reading, UK, from 2009. Since 2013, she has worked as a HPLC product specialist in the analytical business unit of Shimadzu Europa in Duisburg, Germany.

University of Tasmania Researchers Explore Haloacetic Acid Determiniation in Water with capLC–MS
April 29th 2025Haloacetic acid detection has become important when analyzing drinking and swimming pool water. University of Tasmania researchers have begun applying capillary liquid chromatography as a means of detecting these substances.
Prioritizing Non-Target Screening in LC–HRMS Environmental Sample Analysis
April 28th 2025When analyzing samples using liquid chromatography–high-resolution mass spectrometry, there are various ways the processes can be improved. Researchers created new methods for prioritizing these strategies.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)












