Liposome Size, Concentration, and Structural Characterization by FFF-MALS-DLS
Liposomes are often used as nanocarriers to encapsulate in their cores hydrophobic or highly toxic drugs, and deliver these drugs safely to target tissue. During drug nanocarrier research and development as well as production quality control, it is of great importance to monitor liposome size distributions accurately while also verifying drug encapsulation. FFF-MALS-DLS, consisting of field-flow fractionation (FFF) combined with multi-angle light scattering (MALS) and dynamic light scattering (DLS), is a powerful tool for characterizing the size, concentration, and structure of large nanoparticle ensembles.
Wyatt Technology
Liposomes are often used as nanocarriers to encapsulate in their cores hydrophobic or highly toxic drugs, and deliver these drugs safely to target tissue. During drug nanocarrier research and development as well as production quality control, it is of great importance to monitor liposome size distributions accurately while also verifying drug encapsulation. FFF-MALS-DLS, consisting of field-flow fractionation (FFF) combined with multi-angle light scattering (MALS) and dynamic light scattering (DLS), is a powerful tool for characterizing the size, concentration, and structure of large nanoparticle ensembles.
Method
Encapsulation might cause changes in liposomal dimensions, but that is not always the case and other effects could cause such changes. Therefore, a more sophisticated analysis is warranted than mere size. Here, we report the analyses of two liposome samples, one empty and one filled with drug, by means of an Eclipse® FFF system followed by a DAWN® MALS detector with embedded WyattQELS™ DLS module. The WyattQELS detector was placed at approximately 143° to extend the rh measurement to well over 60 nm. The FFF separation method was optimized with the aid of Wyatt’s proprietary FFF simulation software. ASTRA software was used to collect and analyze the light scattering data to determine size and concentration (number density).
Results and Discussion
FFF separates particles according to hydrodynamic radius. Thanks to upstream separation, quantitative size distributions by FFFâMALSâDLS provide far more resolution and quantification than batch (unfractionated) DLS. Online DLS directly measures the hydrodynamic radius, rh, sequentially for each eluting size fraction, while MALS measures simultaneously the root-mean square radius, rg. The shape factor ρ, which is defined as the ratio rg : rh, provides important structural information: it can discriminate between empty and filled shells or quantify the axial ratio of a uniform ellipsoid.
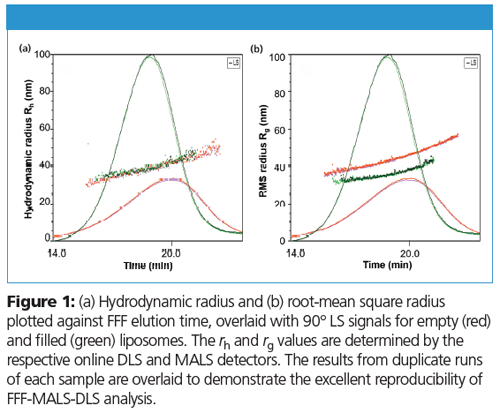
Both rg and rh are plotted against elution time in Figure 1. The results from duplicate runs demonstrate clean separation and excellent reproducibility of the FFF-MALS-DLS method. Figure 1a shows that the rh values for both empty and filled liposomes are well-overlaid, suggesting that drug loading does not impact hydrodynamic size. However, as shown in Figure 1b, rg values for these two liposomes do not overlay, which indicates different internal structures. Figure 2a plots rg against rh; ρ is the slope of the linear fit. The values of ρ for these two populations correlate precisely to empty and filled liposomal structures.
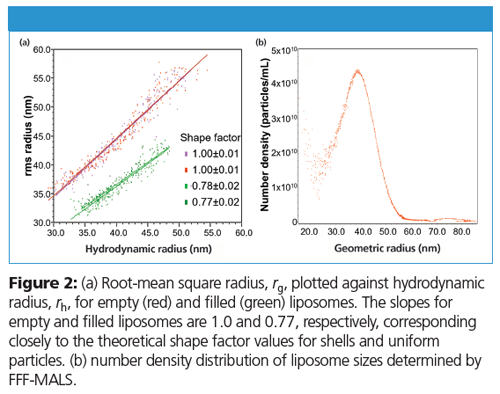
In addition to size and structure, FFF-MALS can determine quantitative size distributions (number density versus size) of sizeâfractionated nanoparticles if the refractive index of the constituent material is known. For lipids this is quite straightforward and Figure 2b provides the quantitative nanoparticle concentration analysis.
Conclusion
For liposomes or other nanoparticles, FFF-MALS-QELS provides an easily adaptable yet powerful characterization tool to obtain information on particle size, size distribution, particle count, as well as structure-all without making assumptions about the particles or their composition. FFF-MALS-DLS instrumentation is essential for robust drug nanocarrier development and quality control.

Wyatt Technology
6330 Hollister Avenue, Santa Barbara, California 93117, USA
Tel.: +1 (805) 681 9009
Website:www.wyatt.com • E-mail:info@wyatt.com

Altering Capillary Gas Chromatography Systems Using Silicon Pneumatic Microvalves
May 5th 2025Many multi-column gas chromatography systems use two-position multi-port switching valves, which can suffer from delays in valve switching. Shimadzu researchers aimed to create a new sampling and switching module for these systems.
Studying Cyclodextrins with UHPLC-MS/MS
May 5th 2025Saba Aslani from the University of Texas at Arlington spoke to LCGC International about a collaborative project with Northwestern University, the University of Hong Kong, and BioTools, Inc., investigating mirror-image cyclodextrins using ultra-high performance liquid chromatography–tandem mass spectrometry (UHPLC–MS/MS) and vibrational circular dichroism (VCD).

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)









