Understanding the Origins of Siloxane Ghost Peaks in Gas Chromatography
Siloxane contamination can interfere with low-level analysis of target compounds whether they are discrete peaks or manifested as high background bleed. They are one of the most common types of “ghost peaks” in gas chromatography (GC) and can come from a variety of sources such as inlet septa, columns, valves, solvents, and vial cap seals. While both column bleed and other sources of contamination contain siloxanes, the composition is different. Examining the mass spectrum and structure provides clues to the source of the contamination. This article analyzes both septa bleed and column bleed fragments and reviews best practices in reducing siloxane “ghost peaks”, in addition to understanding the differences in composition.
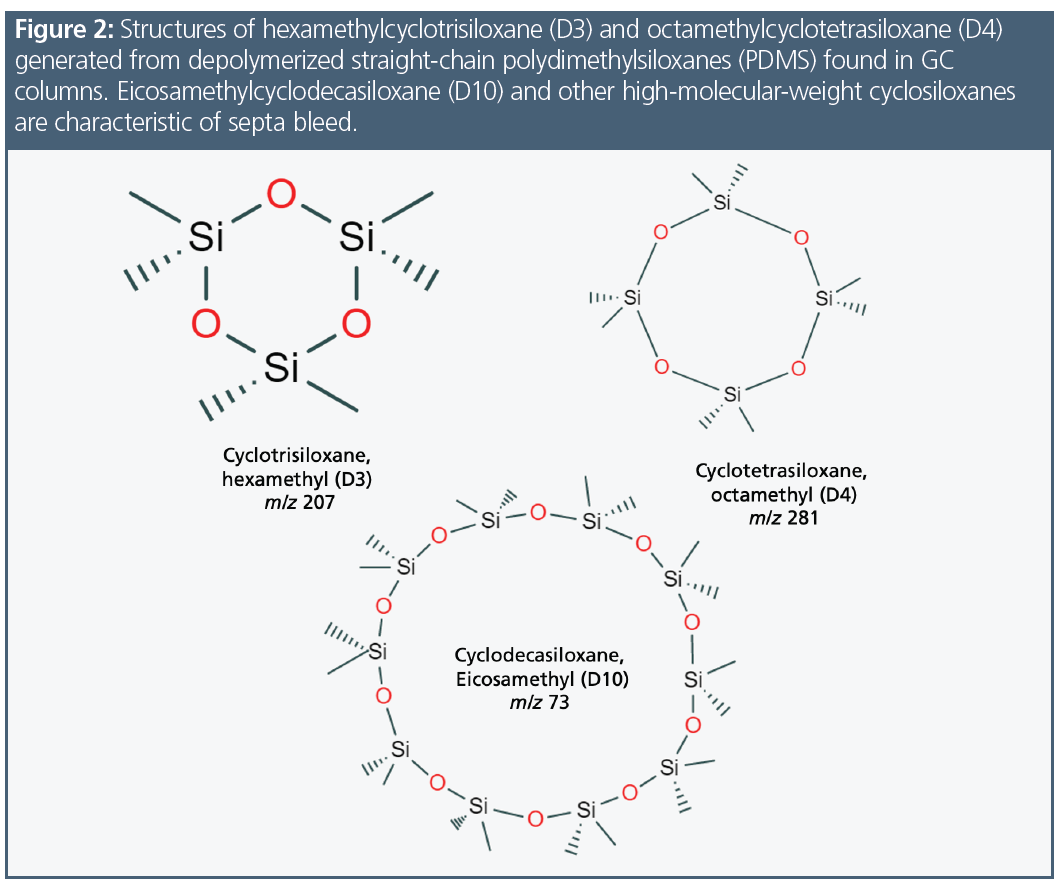
Siloxanes or polysiloxanes are molecules representing [-Si-O-Si-]n, where the Si has two other organic groups attached. For gas chromatography (GC) stationary phases, those side groups could be methyl (“1”‑type column), phenyl (“5”‑type column), combinations such as phenyl/cyano (“624”-type column), or a variety of other functional groups. These long siloxane chains can fold back upon themselves as temperature increases and depolymerize, yielding cyclosiloxanes, predominately trimers (D3), tetramers (D4), and pentamers (D5) (1). It is important to understand these molecules because they can impact a gas chromatogram in several ways.
Oxygen and moisture can greatly accelerate linear siloxane breakdown, which is observed as bleed in GC. Siloxane compounds can originate from carrier gas line. To reduce this risk it is recommended that purification filters are used on carrier gas lines to reduce column bleed, minimize surface activity, and extend column lifetime. This was demonstrated by Linx Waclaski, who performed an experiment attaching different ball valves behind the GC system without purification filters, and set the oven at 35 °C to collect the off‑gassing from the ball valve lubricant. These silicone-based lubricants make opening and closing the valve easier, prevent leaks, and extend valve lifetime. There was a significant amount of contamination observed, which was eliminated by the addition of a triple filter (hydrocarbon, moisture, and oxygen) (2).
Septum bleed from vial caps is less common but can be difficult to isolate since chromatographically it looks identical to inlet septa bleed. Solvents such as methylene chloride can leach siloxanes from septa, especially if the vial has been penetrated more than once. Amanda Rigdon demonstrated this phenomenon by injecting derivatized amphetamines five consecutive times using the same vial and cap septa. In this case, the septum bleed peaks were almost as large as the analyte peaks (3). In another study, repeated use of the same vial cap and vial with a 50-µL insert resulted in endrin and dichlorodiphenyltrichloroethane (DDT) breakdown, in addition to contamination of the inlet (4). Most vial cap septa are lined with a protective layer of polytetrafluorethylene (PTFE) to prevent direct contact with solvent vapour. Without a protective layer, vial cap septa bleed can be detected after 24 h at ambient temperature (3).
The septum on the top of the injection port of the GC system requires care when handling since the septum can adsorb phthalates, finger oils, perfumes, makeup, skin creams, and hand soap residue. GC inlet septum bleed occurs when volatile degradation products enter the column and is seen as a late eluting series of evenly spaced peaks in a programmed GC analysis. During continuous instrument use, change the septum daily to avoid coring and to stop small pieces of septum from entering into the GC liner (5).
Further advice includes the selection of the correct syringe needle to help minimize coring and septa fragments. One popular choice is the tapered cone style 23s–26s needle. Furthermore, understanding the thermal profiles of injection ports for setting inlet temperatures will help to reduce inlet septa bleed. The temperature at the septum is affected by the heating element and the overall inlet design, which varies significantly among manufacturers. For instance, on most GC systems, an inlet set point of 250 °C results in a septa temperature of 100 °C lower. Compare this to some manufacturers of older instruments where the inlet set point is similar to the septa temperature (6,7,8).
If GC column bleed and septa bleed are both made up of siloxanes, what makes them different? At first glance septa bleed has a base peak of m/z 73, while column bleed has a base peak of m/z 207. But keep in mind both column bleed and septa bleed are made up of cyclosiloxanes. These differences are investigated below.
Experimental Design
The first part of this experiment was the extraction and concentration of both septa bleed and column bleed. A 30 m × 0.25 mm, 1.0-µm polydimethylsiloxane stationary phase column (“1-type”) (Restek) was installed into the GC inlet, with the detector end of the column protruding from the GC oven into 1 mL of methylene chloride. By operating the column at its maximum temperature (340 °C), bleed could be captured and concentrated in the methylene chloride, which was placed in a beaker of ice water to minimize evaporative loss. Bleed was collected for 6 h, with methylene chloride being added to the sample as necessary to maintain a volume of approximately 1 mL. Bleed from GC inlet septa was collected by submerging approximately 5 g of septa in 5 mL of methylene chloride for 6 h. Both septum and column bleed were concentrated 10-fold by evaporation at 60 °C. Samples were analyzed individually and combined for a single analysis to evaluate bleed profiles (Figure 1).

The GC column chosen to examine both septa and column was a “1-type” 30 m × 0.25 mm, 0.25-µm (Restek) installed into a GC–MS system (Agilent 7890 GC, 5975 GC–MS) with a split/splitless inlet and a 10:1 split, flow 2 mL/min, and a GC program of 40 °C (hold 1 min) 15 °C per min to 330 °C (hold for 20 min). The MS scan rate was m/z 45 to 550 with a 3-min solvent delay. Compounds associated with bleed profiles were identified using a mass spectra database from the National Institute of Standards and Technology (NIST).
Discussion
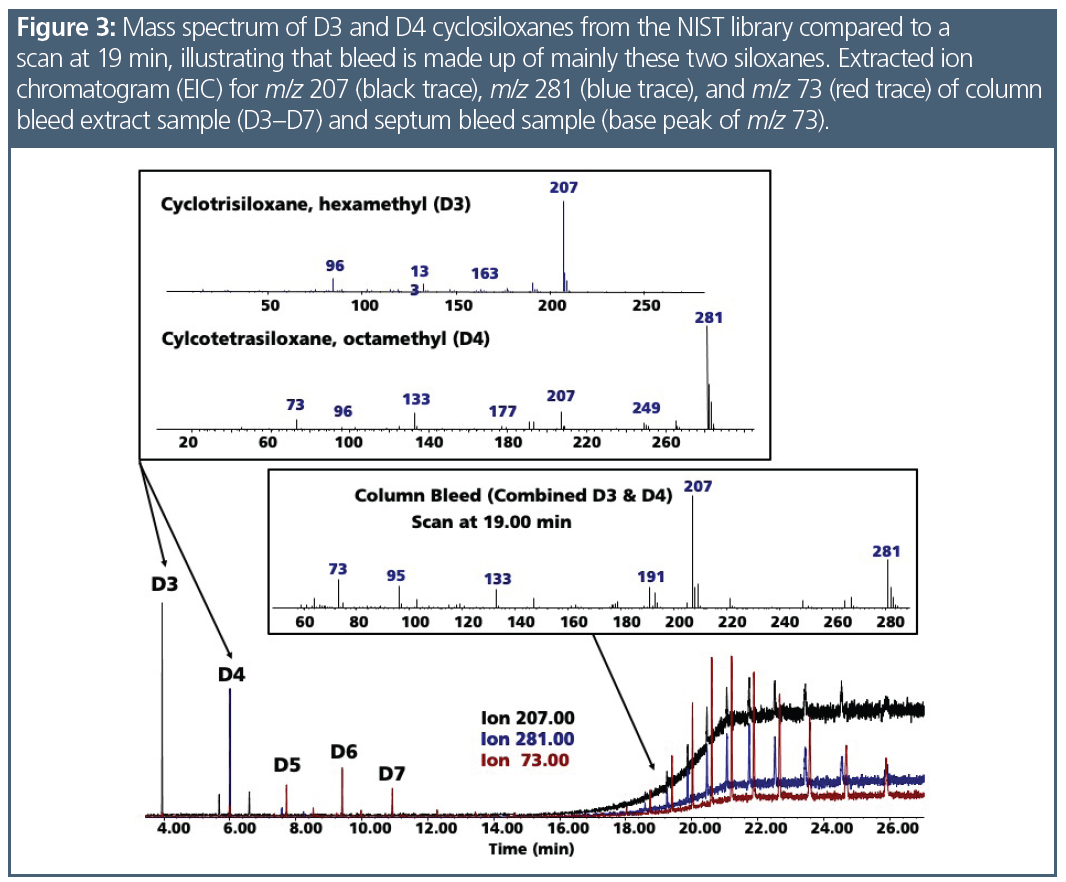
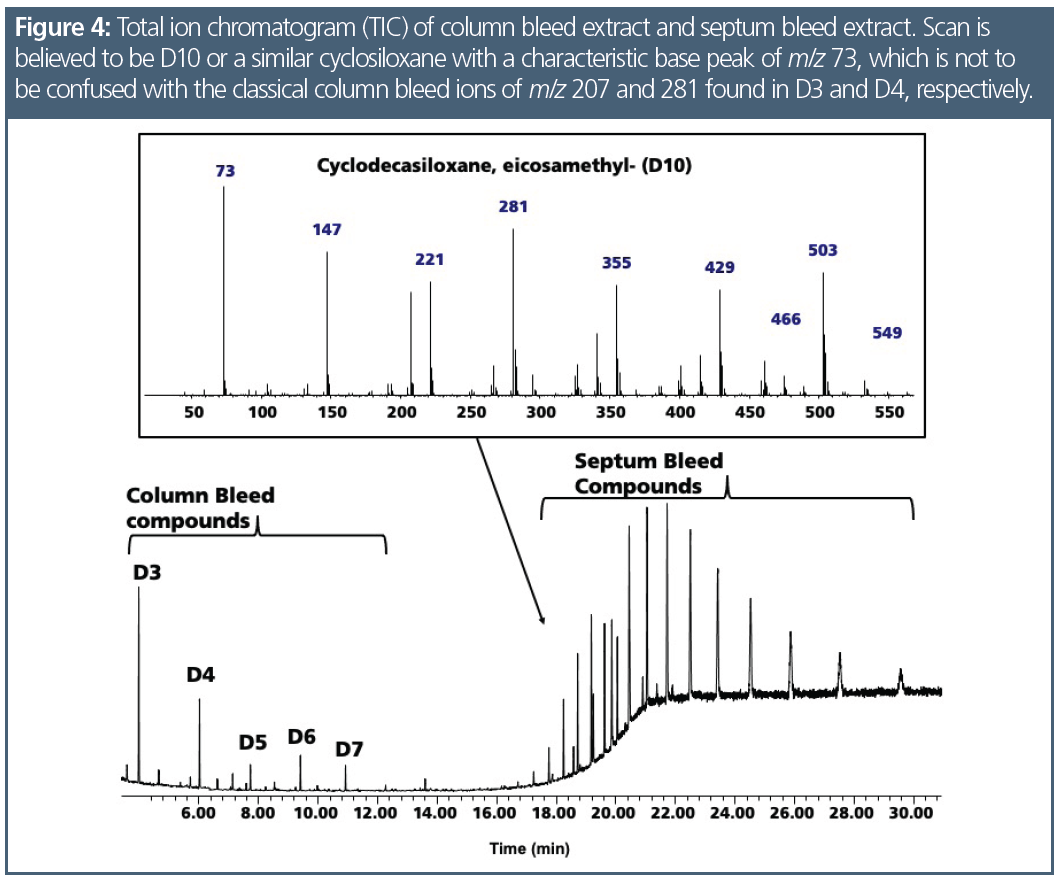
Polydimethylsiloxanes (PDMS), the stationary phase in many GC columns, are long‑chain polymers that slowly decompose into short-chain cyclic molecules. Column bleed had two prominent ions, m/z 207 and m/z 281, which are the base peaks for hexamethylcyclotrisiloxane (D3) and octamethylcyclotetrasiloxane (D4), respectively. Since bleed occurred throughout the chromatogram (Figure 2) and was a mix of these compounds that were not focused, it was seen as background. To confirm the specific compounds in the bleed profile, it was necessary to collect the column bleed and analyze it. Septa bleed concentrated at the head of the column was seen as chromatographic peaks of cyclosiloxanes, but they were in fact much larger molecules. There were over a dozen of these compounds, such as eicosamethylcylcosiloxane (D10), which has a less stable structure and produces a base peak of m/z 73 (Figure 2). Figure 3 presents the spectra of D3 and D4 separately and examines the chromatogram bleed spectrum at 19.00 min, confirming that the bleed is a combination of both compounds. While column bleed also contains D5, D6, and D7, the concentrations of these compounds were less compared to D3 and D4. Since the scan rate was limited to 550 amu, compounds with spectral fragments higher than this scan rate were not able to be correctly identified; however, the fragmentation pattern suggested that they were larger cyclosiloxanes. Figure 4 demonstrates how septum-related bleed compounds were still eluting after 15 min at 330 °C. If the column was not baked out at this temperature, the septum compounds would carryover into the next analysis and would be observed as a bump in the baseline or an increase in background.
Conclusion
One overlooked area for septa bleed is the vial cap septa, and, while less common, it is characterized as sharp repetitive peaks even after several blanks have been analyzed (3). With vial inserts, the issues can be greatly exaggerated and can also degrade target compounds; an example of this would be endrin or DDT (4). Coring, whether from the sample vial or the injection port septum, will result in an increased number of siloxanes that will manifest themselves as “ghost peaks” (5). Whether it is valve bleed, septum vial bleed, or GC inlet septum bleed, there will be late eluting compounds, with m/z 73 for many of them. Identifying the source of siloxanes makes troubleshooting much easier. While it’s true that both septa bleed and column bleed are the same—that is, they are both made up of cyclosiloxanes—the molecular weights are quite different, resulting in different spectra.
Acknowledgements
Special thanks to Jaap de Zeeuw and Erica Pack (Restek) for their technical advice and review.
- References
- D. Allan, S.C. Radzinski, M.A. Tapsak, and J.J. Liggat, Silicon 8, 553–562 (2016) DOI: 10.1007/s12633-014-9247-6
- L. Waclaski,Are My Shut-off Valves Contaminated?www.restek.com/en/chromablography/chromablography/are-my-shut-off-valves-contaminated
- A.Rigdon,How to Diagnose GC Septum Bleed Contamination Sources: Could it be Your Vial Cap?www.restek.com/en/technical-literature-library/articles/how-to-diagnose-gc-septum-bleed-contamination-sources-could-it-be-your-vial-cap
- J. Cochran,Silicone Autosampler Vial Septa Cause Endrin Breakdown and Sample Contaminationwww.restek.com/en/chromablography/chromablography/silicone-autosampler-vial-septa-cause-endrin-breakdown-and-sample-contamination
- M. Badger and S. Grossman, Preventing GC Septum Problemswww.restek.com/globalassets/pdfs/literature/gnar2671-unv.pdf
- L. Waclaski, Thermo Trace 1310 Inlet Temperature Profile vs Agilent 7890 for Split/Splitless Injectors www.restek.com/en/chromablography/chromablography/thermo-trace-1310-inlet-temperature-profile-vs-agilent-7890-for-splitsplitless-injectors
- S. Grossman, It’s a Matter of Degrees, but Do Degrees Really Matter? https://www.restek.com/globalassets/pdfs/literature/gnar1937-unv.pdf
- S. Grossman,How Hot is Your Septum? https://pdf2.chromtech.net.au/580134_ADV07-2_22.pdf
Chris English has managed a team of chemists in Restek’s innovations laboratory since 2004. Before taking the reins of the laboratory, he spent seven years as an environmental chemist and was critical to the development of Restek’s current line of volatile GC columns. Prior to joining Restek, he operated a variety of gas chromatographic detectors conducting method development and sample analysis. Chris holds a B.S. in environmental science from Saint Michael’s College, USA.
E-mail: chris.english@restek.com
Website: www.restek.com

Detecting Hyper-Fast Chromatographic Peaks Using Ion Mobility Spectrometry
May 6th 2025Ion mobility spectrometers can detect trace compounds quickly, though they can face various issues with detecting certain peaks. University of Hannover scientists created a new system for resolving hyper-fast gas chromatography (GC) peaks.
Altering Capillary Gas Chromatography Systems Using Silicon Pneumatic Microvalves
May 5th 2025Many multi-column gas chromatography systems use two-position multi-port switching valves, which can suffer from delays in valve switching. Shimadzu researchers aimed to create a new sampling and switching module for these systems.
New Study Reviews Chromatography Methods for Flavonoid Analysis
April 21st 2025Flavonoids are widely used metabolites that carry out various functions in different industries, such as food and cosmetics. Detecting, separating, and quantifying them in fruit species can be a complicated process.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)















