Tips & Tricks GPC/SEC–Light Scattering: Garbage In, Garbage Out
This instalment of Tips & Tricks shows the influence of analytical parameters on gel permeation chromatography/size-exclusion chromatography (GPC/SEC)–light scattering results when artificially wrong parameters reflecting typical experimental errors are used.
Photo Credit: Dominik Pabris/ Getty Images

This instalment of Tips & Tricks shows the influence of analytical parameters on gel permeation chromatography/size-exclusion chromatography (GPC/SEC)–light scattering results when artificially wrong parameters reflecting typical experimental errors are used.
Hyphenation of gel permeation chromatography/size-exclusion chromatography (GPC/SEC) with light scattering can help both methods to overcome the limitations of each single method. Light scattering benefits from the separation power of GPC/SEC, and when combined with GPC/SEC it is possible to determine the complete molar mass distribution of a sample and not just the weight average molecular weight. GPC/SEC benefits from the absolute detection principle of the light scattering detector, which reduces the need to rely on elution volume and calibration with narrow standards.
However, when combining two techniques the prerequisites of both methods need to be fulfilled. Data obtained using an inappropriate GPC/SEC method will provide false distribution information. Results obtained using the wrong parameters or settings for light scattering detection will result in the wrong molar mass distributions.
Why is the Precise and Accurate Concentration Needed for GPC/SEC–Light Scattering Measurements?
The scattering intensity of a substance is a product of an instrument-specific constant (light scattering detector constant), a sample-specific constant (refractive index increment, [dn/dc]2), the molar mass, and the concentration. Both parameters contribute – the higher the molar mass and/or concentration, the higher the signal intensity. If the molar mass needs to be determined the concentration needs to be known.
Batch light scattering experiments (only light scattering, no GPC/SEC fractionation prior to detection) are easy to evaluate because the sample concentration can be determined using a balance. In this case all parts of the sample are present at the same time in the cell and contribute to the measured intensity of the scattered light.
This is different for on-line experiments. Although the complete sample will be injected with a known concentration, the sample parts (molecules of different sizes) are fractionated in the column. They reach the light scattering detector cell at different times and the light scattering data are obtained for fractions with unknown concentrations.
Concentration Determination for GPC/SEC–Light Scattering Measurements
A typical solution to overcome the concentration problem is to use a concentration detector (typically refractive index, RI, or UV/DAD) in combination with the light scattering detector. This allows the concentration of each single slice or chromatographic fraction to be measured on-line.
There are two approaches to determine the slice concentration, ci.
1) 100% mass recovery/injected mass:
Here it is assumed that the area underneath the signal peak corresponds to the injected mass (concentration and injection volume dependent). This means that all parts of the sample are soluble, that everything is injected properly, and that everything elutes from the column. The parameters that mostly influence the results are dn/dc, concentration, and injection volume. If these parameters are wrong, the obtained molar masses are also wrong.
2) Calibrated concentration detector and known sample-specific constant for the concentration detector:
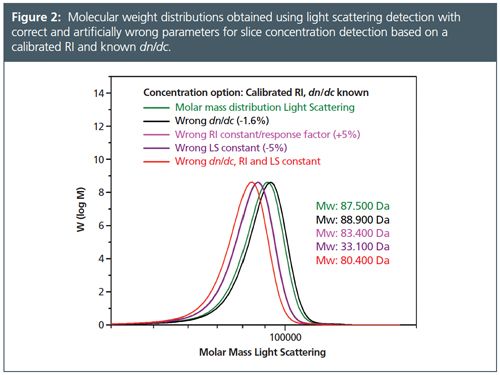
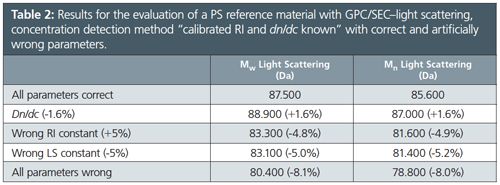
â¨In this case a response factor/detector constant is determined for the concentration detector, similar to high performance liquid chromatography (HPLC) data evaluation processes. In case of RI detection this constant and the dn/dc of the sample are used to determine the slice concentration from the signal intensity of the concentration detector. The parameters that mostly influence the results are dn/dc and the detector constants of the concentration detector and the light scattering detector. If these parameters are wrong the obtained molar masses are also wrong.
Influence on Results from dn/dc and Concentration Determination
Using a calibrated concentration detector and the matching sample-specific constant is the more robust method and leads to less deviation and higher reproducibility of the results.
Table 1 and the corresponding Figure 1 compare results for a polymethylmethacrylate (PMMA) sample evaluated using artificially wrong evaluation parameters for the method 100% mass recovery.
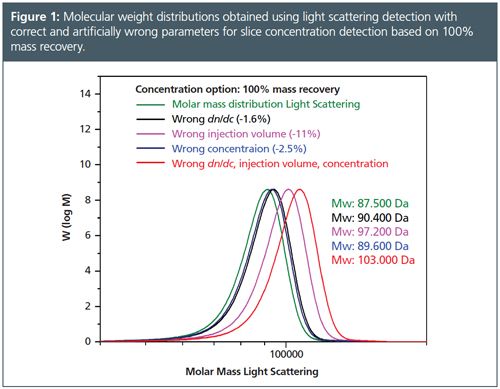
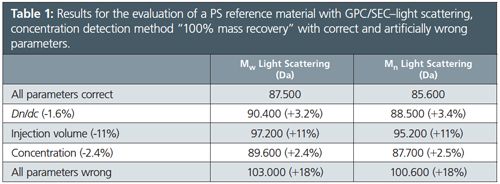
Table 2 and the corresponding Figure 2 compare results for the same PMMA sample evaluated using artificially wrong evaluation parameters for the method calibrated RI and known dn/dc.
Comparison of the data shows that if all parameters are correct and chromatographic conditions are good both concentration detection methods will yield the same results within experimental error. This would not be the case if, for example, the wrong parameters are used, parts of the sample are insoluble, if parts of the sample are retained by frits or on the column, or in the case of unreliable injection.
In general the influence of a wrong dn/dc on the results is less for the method “Calibrated concentration detector and known sample-specific constant”. Also this method is more robust with respect to the most common experimental error of an incorrectly determined injected mass or not completely eluted mass.
It is good practice to prepare samples in a way that it is possible to work with both concentration detection options. This means that all samples and solvents should be weighed to ensure a precise and accurate concentration. It is also then possible to check sample recovery.
Comparison to Conventional GPC/SEC and GPC/SEC-Viscometry (Universal Calibration)
One advantage of GPC/SEC alone is that absolute sample concentration does not influence the results. Changing any of the parameters above will not influence the results. Molar mass averages and molar mass distribution results will be reproducibly the same for all the curves shown if only standard detection is used. Even the constant for the concentration detector does not influence the results.
In contrast, the majority of the parameter changes above will also influence the results obtained with GPC/SEC–viscometry. As with GPC/SEC–light scattering, this method also relies on slice concentration determination. However, the shift of GPC/SEC–viscometry results will be in the opposite direction to GPC/SEC–light scattering. If a wrong parameter decreases the molar mass for light scattering evaluation it will increase the molar mass for viscometry/universal calibration. One of the most common experimental problems therefore leads to a strong deviation between results for the two methods that should measure true molar masses. Understanding the influence of analysis parameters is therefore essential for users of advanced multi-detection.
Summary
• While conventional GPC/SEC results are not affected by imprecise and inaccurate concentrations, GPC/SEC–light scattering results show a dependence.
• Two independent concentration detection methods can be applied for GPC/SEC–light scattering. The method to measure the slice concentration using a calibrated detector and a known sample-specific constant is more robust. This method is also less affected by a wrong dn/dc.
• Dn/dc needs to be considered every time that light scattering detection is applied for GPC/SEC. The dn/dc should be high enough and constant over the evaluated fractions to reduce the applicability for GPC/SEC–light scattering for copolymers.
• GPC/SEC–light scattering results and GPC/SEC–viscometry results are influenced in opposing directions by the same experimental error.
Daniela Held studied polymer chemistry in Mainz, Germany, and works in the PSS software and instrument department. She is also responsible for education and customer training.

Characterizing Plant Polysaccharides Using Size-Exclusion Chromatography
April 4th 2025With green chemistry becoming more standardized, Leena Pitkänen of Aalto University analyzed how useful size-exclusion chromatography (SEC) and asymmetric flow field-flow fractionation (AF4) could be in characterizing plant polysaccharides.
Investigating the Protective Effects of Frankincense Oil on Wound Healing with GC–MS
April 2nd 2025Frankincense essential oil is known for its anti-inflammatory, antioxidant, and therapeutic properties. A recent study investigated the protective effects of the oil in an excision wound model in rats, focusing on oxidative stress reduction, inflammatory cytokine modulation, and caspase-3 regulation; chemical composition of the oil was analyzed using gas chromatography–mass spectrometry (GC–MS).









