Simultaneous Determination of Plant Growth Regulators in Bean Sprouts by UHPLC Coupled with MS/MS
The Column
A rapid and sensitive ultrahigh-pressure liquid chromatography tandem mass spectrometry (UHPLC–MS/MS) method was developed to simultaneously determine six plant growth regulators in bean sprouts with a simple preparation. Analyte extraction from samples was effectively performed using liquid–liquid extraction (LLE) by acetonitrile. Chromatographic separation was conducted on a C18 reversed-phase column with gradient elution. The analytes were detected by tandem quadrupole MS after negative electrospray ionization by multiple reaction monitoring. The developed method was validated by testing method specificity, matrix effect, sensitivity, linearity, accuracy, and precision.
Photo Credit: MAHATHIR MOHD YASIN/Shutterstock.com

Cheng-Yuan Cai1, Li-Zhong Yang1, Yong-Ming Xie1, and Feng Qin2,1PerkinElmer Inc., Shanghai, China, 2PerkinElmer Inc., Toronto, Canada
A rapid and sensitive ultrahigh-pressure liquid chromatography tandem mass spectrometry (UHPLC–MS/MS) method was developed to simultaneously determine six plant growth regulators in bean sprouts with a simple preparation. Analyte extraction from samples was effectively performed using liquid–liquid extraction (LLE) by acetonitrile. Chromatographic separation was conducted on a C18 reversed-phase column with gradient elution. The analytes were detected by tandem quadrupole MS after negative electrospray ionization by multiple reaction monitoring. The developed method was validated by testing method specificity, matrix effect, sensitivity, linearity, accuracy, and precision.
Bean sprouts are a commonly used ingredient in Asian cuisine and are highly valued for their nutritional properties. In recent years, there have been concerns about the presence of exogenous plant growth regulators in bean sprouts (1,2), which can accelerate their growth. Though this accelerated growth is great for production, the addition of growth regulators is known to be harmful to the health of consumers. To effectively control the residues of these compounds in food, the Ministry of Agriculture of China has banned the use of gibberellin, benzyladenine, and 4-chlorophenoxyacetate in bean sprouts since 2015 (3). It is therefore of great importance to develop a simple and sensitive method for the simultaneous determination of plant growth regulators in bean sprouts. The traditional method uses liquid chromatography–ultraviolet (LC–UV) to separate and detect those compounds and has low sensitivity. Tedious sample concentration and cleanâup steps are needed to meet the sensitivity requirements based on new regulations. Furthermore, the selectivity of UV is very limited and the results generated might be questionable. In this study, we developed an ultrahigh-pressure liquid chromatography tandem mass spectrometry (UHPLC–MS/MS) method for the determination of those analytes. The method was proven to have high selectivity and sensitivity and was suitable for residue contaminants analysis in bean sprouts.
Experimental
Sample Preparation: All samples were obtained from a local market in Shanghai. After being homogenized, 2.5 g of sample was weighed into a centrifuge tube. Five mL of acetonitrile and 1.5 g of sodium chloride were added to the tube, which was then mixed for 1 min and placed in an ultrasonic bath for 5 min. The tube was then centrifuged at 6000 rpm for 5 min. The final solution was diluted fivefold with water and filtered through a 0.22-μm membrane filter.
Calibration Standards: Stock solutions (1 mg/mL) of analytical standards were prepared by dissolving 10 mg of the compounds in 10 mL of methanol. The calibration standard solutions were prepared by serial dilution (0.005, 0.01, 0.02, 0.05, 0.1, 0.2, 0.5, 1, 2, 5, 10, 20, 50, 100 ng/mL) of the stock solution with an 80:20 (v/v) water–acetonitrile mixture.
Liquid Chromatographic Operating Conditions (4): The analytes were separated with a high performance liquid chromatography system (Altus A-30 UPLC, PerkinElmer Inc.) (4) using a SPP C18 column (2.1 × 100 mm, 2.7-μm, PerkinElmer Inc). The temperature of the column oven was set at 40 °C. The mobile phase consisted of water containing 0.01% formic acid (A) and acetonitrile (B). A reversed phase separation with a 9-min gradient was used. The flow rate was set at 0.3 mL/min and the injection volume was 10 μL.
Mass Spectrometric Operating Conditions (5): The UHPLC system was coupled to a triple quadrupole mass spectrometer (QSight 210, PerkinElmer Inc.) equipped with an electrospray ionization source operating in negative ion mode (5). The mass spectrometer operating conditions were as follows: ElectroSpray: -5000 v; heating gas temp.: 500 °C; HSID temp.: 320 °C; dry gas setting: 70; nebulizer gas setting: 160. Detection of analytes by tandem mass spectrometry was conducted in multiple reaction monitoring (MRM) mode. Two MRM transitions were optimized for each analyte. The ones that show higher signal were used as quantifier, and the ones that show lower signal were used as qualifier. The MRM dwell time was set at 30 ms for each transition.
Validation of the method was conducted by characterizing method specificity, matrix effect, sensitivity, linearity, accuracy, and precision.
Results and Discussion
Specificity of analysis was tested by spiking trace level concentration of a mix of growth hormones into bean sprout matrix and matched against the blank matrix, as shown in Figure 1. The results suggested that there was no matrix interference and the method could be successfully applied to samples with no additional sample cleanup.
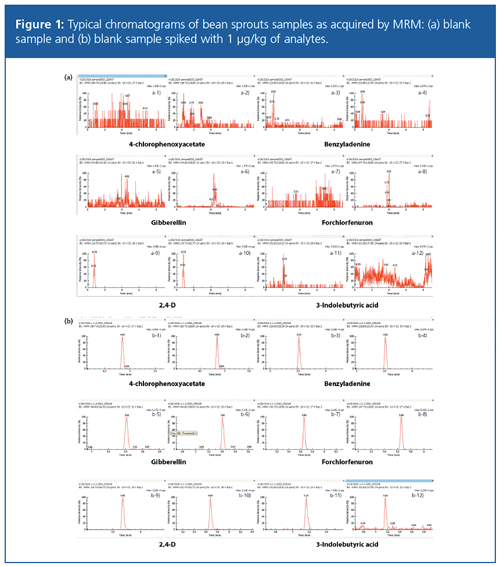
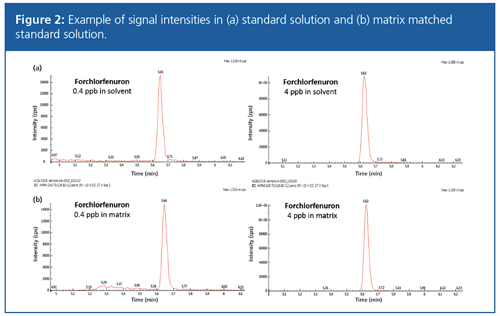
To evaluate ion suppression from matrix effects, the signal intensity of the standard solution (Figure 1[a]) was compared with that of the matrixâmatched standard solution (Figure 1[b]) at different concentrations. A negative matrix effect (b/a × 100%) of <100% indicates matrix suppression, whereas a positive matrix effect (b/a × 100%) of >100% indicates ionization enhancement. This is demonstrated in Figure 2, which shows an example of signal intensities of a (a) Forchlorfenuron standard solution and (b) matrix matched standard solution. The matrix effect was calculated for all six compounds in mung bean sprouts and soybean sprouts. Except for 3-indolebutyric acid, the five other analytes showed no significant matrix effect. 3-indolebutyric acid showed strong matrix suppression. In this study, matrixâmatched calibration curves were used for quantification to compensate for any matrix effect during the experiment.
Recovery study of the analytes during sample preparation was conducted by spiking standards into the sample at three different levels-0.4 ppb, 1 ppb, and 4 ppb. Overall, spiked recoveries were between 66–104%.
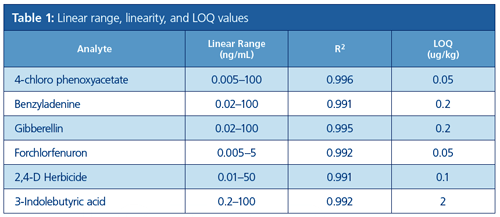
Linear dynamic range was evaluated by matrix-matched calibration standards prepared as described in the experimental section. Table 1 represents the linear dynamic range (LDR) and limits of quantification (LOQ) values for the analytes. LDR range was from 2 to 4 orders of magnitude. The correlation coefficients (R2) were all above 0.990. LOQ in real samples were between 0.05–2 µg/kg.
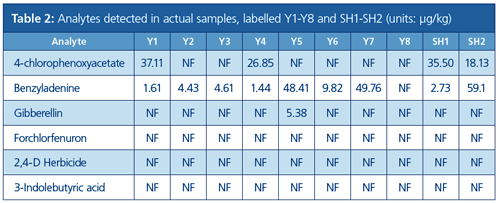
The developed method was applied to the analysis of 10 samples obtained from different local markets. The results are summarized in Table 2. Based on the results, the occurrences of 4-chlorophenoxyacetate, benzyladenine, and gibberellin were 40%, 90%, and 10%, respectively, with benzyladenine the most commonly detected plant growth regulator in bean sprouts. This is consistent with the information from Ministry of Agriculture (3). The detected concentrations were within 1–50 µg/kg range, which was very close to the maximum residue level (10 µg/kg).
Conclusion
This study has demonstrated a rapid, sensitive, and reproducible UHPLC–MS/MS method for the analysis of six plant growth regulators in bean sprouts. Compared to the traditional LC–UV method, the sample preparation steps are greatly reduced. The MRM scan provided much better selectivity and sensitivity, achieving limits of quantitation for all analytes within 0.05–2 μg/kg. The methodology also provides a robust platform for analysis of trace level plant growth regulators and can easily meet limits set down by the Chinese regulatory authorities (5).
References
- S.-K. Cho, A.M. Abd El-Aty, K.H. Park, et al., Food Chemistry 136, 1414–1420 (2013).
- L. Ma, H. Zhang, and W. Xu. Food Anal. Methods6, 941–951 (2013).
- Ministry of Agriculture of the Peoples Republic of China, Document No. 1 of 2003, dispatched by Ministry of Agriculture of China; http://english.agri.gov.cn/
- Altus HPLC Product Note: PerkinElmer Inc., http://www.perkinelmer.com/lab-solutions/resources/docs/BRO_012040_01_Altus_HPLC.pdf
- QSight Triple Quad LC/MS/MS Product Note: PerkinElmer Inc., Shelton, CT, http://www.perkinelmer.com/product/qsight-220-multi-opt-dual-source-system-bc003382?searchTerm=&pushBackUrl=
Feng Qin is a Product Manager at PerkinElmer. He has more than ten years of experience in LC–MS for food and environmental applications. In his career, his responsibility progressed from field application specialist to applications scientist (in R&D), lead customer for software product, and product manager for food safety solutions. Prior to joining PerkinElmer, Feng did a postdoctoral fellowship at the University of Alberta for three years, and worked for Sciex for
seven years.
Cheng-Yuan Cai is a Mass Spectrometry Product Specialist at PerkinElmer in China. He has over eight years of experience in LC–MS applications. Prior to joining PerkinElmer, he worked for Sciex for three years. He holds a Masters Degree from the Chinese Academy of Agricultural Sciences.
Li-Zhong Yang is a Mass Spectrometry Product Specialist at PerkinElmer in China. Prior to joining PerkinElmer, he worked for Sciex for three years. He holds a Master Degree from Jimei University.
Yong-Ming Xie is a Strategic Business Development Leader at PerkinElmer. He has over 20 years of experience in LC–MS applications and in various roles, including marketing development, applications support, and sales across multiple mass spectrometry companies.
E-mail: feng.qin@perkinelmer.comWebsite:www.perkinelmer.com

Characterizing Plant Polysaccharides Using Size-Exclusion Chromatography
April 4th 2025With green chemistry becoming more standardized, Leena Pitkänen of Aalto University analyzed how useful size-exclusion chromatography (SEC) and asymmetric flow field-flow fractionation (AF4) could be in characterizing plant polysaccharides.
Investigating the Protective Effects of Frankincense Oil on Wound Healing with GC–MS
April 2nd 2025Frankincense essential oil is known for its anti-inflammatory, antioxidant, and therapeutic properties. A recent study investigated the protective effects of the oil in an excision wound model in rats, focusing on oxidative stress reduction, inflammatory cytokine modulation, and caspase-3 regulation; chemical composition of the oil was analyzed using gas chromatography–mass spectrometry (GC–MS).



![IMG_4773[1].jpg IMG_4773[1].jpg](/_next/image?url=https%3A%2F%2Fcdn.sanity.io%2Fimages%2F0vv8moc6%2Fchroma%2F6db719ea80519dad8e948ad595b96c3fa3731826-200x207.jpg%3Ffit%3Dcrop%26auto%3Dformat&w=3840&q=75)









