Simultaneous, Automated Extraction of 96 Urine Samples for Drugs of Abuse Analysis by LC–MS/MS Using the Microlab® NIMBUS® Workstation
Special Issues
Utilizing Hamilton’s CO-RE® disposable tips with DPX technology provides a fast, accurate, and simple extraction method for analyzing drugs of abuse in urine. The Microlab NIMBUS equipped with a CO-RE 96-channel Multi-Probe Head (MPH) allows for high-throughput, automated sample processing.
Kaylee McDonald*, Evan DiVirgilio*, William Brewer*, Jose Ocampo†, and Michael Mouradian†, *DPX Labs, †Hamilton Robotics
Utilizing Hamilton’s CO-RE® disposable tips with DPX technology provides a fast, accurate, and simple extraction method for analyzing drugs of abuse in urine. The Microlab NIMBUS equipped with a CO-RE 96-channel Multi-Probe Head (MPH) allows for high-throughput, automated sample processing. Using the NIMBUS96 platform, two sample plates can be ready for LC–MS/MS injection in approximately 15 min.
Matrix effects are a major concern in LC–MS/MS analysis. Proper sample preparation can be very time consuming and is generally the bottleneck for sample throughput and analysis of many laboratories. In-tip Dispersive Pipette eXtraction (DPX) is a new solid phase extraction (SPE) technology that minimizes sample processing steps and decreases reagent use compared to typical SPE plate-based techniques. Hamilton’s CO-RE tips containing DPX chemistry, now offer a fully automated, high-throughput extraction method that can be used on any Hamilton liquid handling platform. Hamilton automated DPX methods include a variety of applications: therapeutic and abused drugs in oral fluid, urine and blood, and vitamins and steroids in serum. The versatility of DPX on the Hamilton automated liquid handling platforms allows for customized laboratory workflow solutions. The method reported herein demonstrates a urine extraction method for the analysis of therapeutic and abused drugs with high reproducibility, and provides the necessary sensitivity for target analyte quantification and analysis for forensic and clinical laboratories.
NIMBUS with CO-RE 96 MPH

Microlab NIMBUS

CO-RE Tip with DPX Technology


Workflow
Microplates containing hydrolyzed urine were loaded on to the NIMBUS96. The automated NIMBUS method started by filling a 96-well microplate with 200 μL of water (for the wash step) and 150 μL of 1% FA (formic acid) in methanol (for the elution step). The DPX tips (mixed mode with RP/WAX) were then conditioned by aspirating 30% methanol from a solvent reservoir. After conditioning, the sample solutions, containing 150 μL urine + 100 μL mixture of buffer, enzyme and internal standards, were aspirated and dispensed three times in order to bind the drugs of abuse targets to the sorbent. Water was then aspirated and dispensed to remove sample matrix components such as salts, urea, and creatinine. The analytes of interest were eluted by aspirating and dispensing 1% FA in methanol three times. The eluent is simply diluted until an appropriate percentage of methanol is reached for injection. In this case, 1050 μL of water was added (to make the final solution 12.5% methanol). Analysis was performed on a Thermo TSQ Vantage triple quadrupole instrument with an Agilent 1260 HPLC using an Agilent Poroshell ECC18 column (3.0 x 50 mm, 2.7 μm) with a 10 μL injection.
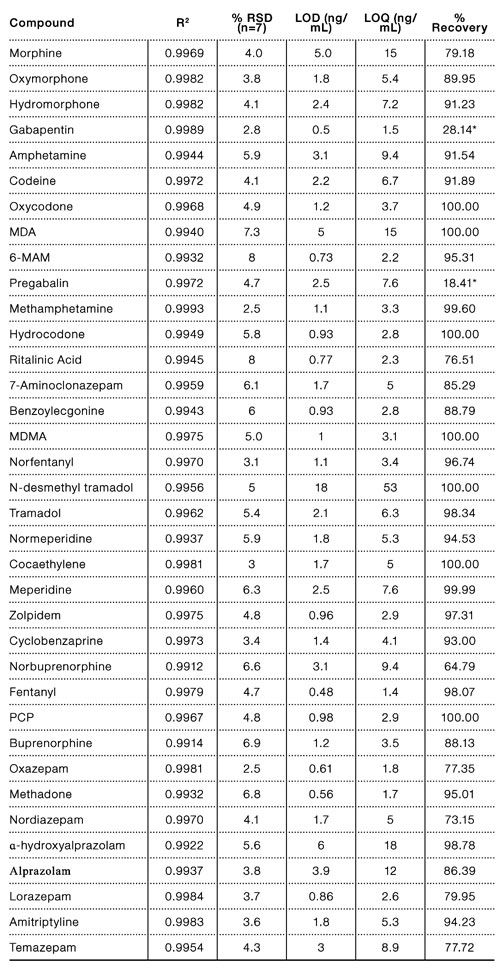
Results and Discussion
From start to finish, this fully automated dispersive SPE method takes approximately 15 min to prepare two 96-well microplates of samples. Results from this method are linear, accurate, and reproducible. All correlation coefficients were greater than 0.99 for the range of at least 12.5–400 ng/mL, with most analytes being linear from 6.25–800 ng/mL. Relative standard deviation was calculated using seven replicate extractions at 400 ng/mL and ranged from 1.6 to 8.0 ng/mL. Limits of detection (LOD) were calculated as 3.3*σ/m, where σ is the standard deviation of the lowest non-zero calibrator and m is the slope of the calibration line. Limit of quantitation (LOQ) was calculated as 10*σ/m. LOD ranged from 0.50 to 18 ng/mL and LOQ ranged from 1.5 to 54 ng/mL. While it should be emphasized that confirmatory tests and cutoff levels are highly dependent on LC-MS/MS instrumentation and procedures, for improved sensitivity and lower LODs and LOQs, solvent evaporation can be employed to concentrate the extracts. Elution volume can also be increased to 500 μL to increase recovery as well.
Conclusion
The method described herein demonstrates adequate recoveries, sensitivity, and reproducibility for reliable sample preparation in a high-throughput setting. Complete reliable automated sample preparation method provides a less tedious and faster process compared to manual processing for analysis of drugs of abuse; two 96-well microplates are processed in 15 min.

Hamilton Company
4970 Energy Way, Reno, NV 89502
tel. (775) 858-3000, (800) 648-5950
Website: www.hamiltoncompany.com

Determining the Effects of ‘Quantitative Marinating’ on Crayfish Meat with HS-GC-IMS
April 30th 2025A novel method called quantitative marinating (QM) was developed to reduce industrial waste during the processing of crayfish meat, with the taste, flavor, and aroma of crayfish meat processed by various techniques investigated. Headspace-gas chromatography-ion mobility spectrometry (HS-GC-IMS) was used to determine volatile compounds of meat examined.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)










