HPLC Method for the Separation of Nine Key Components of Milk Thistle
Silymarin, derived from the milk thistle plant, has been used as a natural remedy for the treatment of a number of liver diseases as well as in the protection of the liver from potential toxins. More recently, silymarin has been connected to antitumor promoting activity (1). It is therefore of interest to have available analytical methods for the analysis of silymarin components in various matrices.
Silymarin, derived from the milk thistle plant, has been used as a natural remedy for the treatment of a number of liver diseases as well as in the protection of the liver from potential toxins. More recently, silymarin has been connected to antitumor promoting activity (1). It is therefore of interest to have available analytical methods for the analysis of silymarin components in various matrices.
The silymarin complex consists of several closely related, biologically active components. The objective of this study was to investigate several modern stationary phase chemistries for optimal selectivity toward the development of an efficient method for the analysis of silymarin. The method would optimally provide baseline resolution for nine of the known components of silymarin in less than 15 min.
Initial screening experiments were used to select a single combination of stationary phase and organic modifier that demonstrates the greatest potential for the separation. The information from subsequent optimization experiments was processed with simulation software to predict an optimized set of conditions. Finally the predicted conditions were confirmed using the analysis of an herbal supplement obtained from a local drug store.
Experimental
The screening protocol utilized five Ascentis Express stationary phases including C18, C8, RP-Amide, Phenyl-Hexyl, and pentafluorophenylpropyl (F5). Gradient elution with two distinct organic modifiers was performed. Injections of a standard containing nine of the known constituents of silymarin were made under each condition.
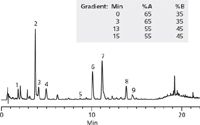
Figure 1
The HPLC screening method employed gradient elution from 5–95% of either methanol or acetonitrile. The aqueous component of the mobile phase was 0.1% formic acid (pH 2.6).
Results and Discussion
Using a simple peak counting approach, the combination of the C18 stationary phase along with methanol as the organic modifier resulted in the most visible peaks with nine. Other combinations of stationary phase and organic modifier produced between six and eight peak responses.
Four separate chromatographic runs using the C18 stationary phase and methanol modifier were conducted where the gradient slopes were varied (5% and 10% ramp) at two different temperatures (30 °C and 60 °C). The data was then analyzed using ACD LC-Simulator to predict the most suitable optimized conditions. Because the suggested conditions ran to just 45% organic, an additional step to 100% organic was included to elute potential hydrophobic components from a natural sample.
Finally an herbal supplement labeled as containing milk thistle along with dandelion, fennel, and licorice was extracted using water:ethanol 50:50, v/v and analyzed. As shown, all nine of the targeted components could be observed in the herbal supplement material.
Conclusions
Stationary phase screening at the onset of method development, especially when dealing with a complex set of analytes, provides a facile means of analytical method development. As shown in the study, a few short experiments, coupled with powerful prediction software provided chromatographic conditions suitable for the analysis of biologically active milk thistle components. The developed conditions should prove useful in natural supplement, raw material and/or biological monitoring of silymarin complex components.
Reference
(1) J.I. Lee, B.H. Hsu, D. Wu, and J.S. Barrett, J. of Chromatog.A 1116, 57–68 (2006).
Supelco/Sigma-Aldrich
595 North Harrison Road, Bellefonte, PA 16823
tel. (800) 359-3041, (814) 359-3041
Website: sigma-aldrich.com/analytical

The Benefits of Custom Bonded Silica
April 1st 2025Not all chromatography resins are created equal. Off-the-shelf chromatography resins might not always meet the rigorous purification requirements of biopharmaceutical manufacturing. Custom bonded silica from Grace can address a wide range of separation challenges, leading to real performance improvements. Discover more about the latest innovations in chromatography silica from Grace, including VYDAC® and DAVISIL®.
5 Things to Consider When Selecting a Chromatography Silica
April 1st 2025Particularly in the pharmaceutical industry, drug purity isn’t just a goal – it’s essential for achieving safety, stability and efficacy. However, purification is easier said than done, especially with challenging molecules like DNA and RNA “oligonucleotides,” due in large part to their diversity and the range of impurities that can be generated during production. Enter DAVISIL® chromatographic silica, with a wide range of pore diameters and particle sizes to meet your specific application, performance and sustainability requirements. Before you choose the chromatography resin for your next purification application, take a look at these 5 considerations.
MilliporeSigma: Ultrapure Water for Sensitive LC-MS Analysis of Pesticides
March 25th 2025The aim of the study was to illustrate the efficiency of Milli-Q® water purification systems in eliminating pesticides from tap water, thereby producing and delivering reliable and consistent-quality ultrapure water suitable for pesticides analysis
















