How to Tackle an Unknown: Notes from the Fourth Method Development Olympics at CoSMoS
LCGC North America
Put yourself in their spot: How would you tackle analyzing a bag of gummy bears that showed up on your lab bench? Here, we offer some insights from the very capable finalists at The Conference on Small Molecule Science (CoSMoS), which was held in August 2015 in San Diego, California.
How would you analyze a bag of gummy bears that showed up on your laboratory bench? This was the challenge taken on by teams of analysts in advance of the Conference on Small Molecule Science (CoSMoS) that was held in August 2015 in San Diego, California. This article shares insights from how the finalists approached the question.
An increasingly popular event each year at the Conference on Small Molecule Science (CoSMoS) is the Method Development Olympics. The program was initiated in 2011 by two conference committee members, Damian Morrison and Ken Fountain. Since then, it has evolved each year because a changing committee, often involving past winners, has lent its expertise to the next year’s challenge. This year’s committee consisted of Karen Alsante, Ruchi Mehta, and William Farrell, all of Pfizer, along with Kevin Gauger of Catalent Pharma Solutions, and was led by Jeff Kiplinger of Averica Discovery. The committee devised a sample that met a variety of requirements: It had to be nontoxic, be able to be sent to candidates, and remain stable. And of course, the sample had to be intriguing for those keen to challenge their analytical prowess.
How the Method Development Olympics Work
The process of the Method Development Olympics is simple. A challenge is posted on the CoSMoS website early in the year. Scientists register their interest in participating, and the sample is sent to them. Once they believe they have solved the question, they submit their answer, and the process they followed to get to the answer, to the committee. The committee then chooses three finalists, from among those who submitted correct answers, to present their approaches at CoSMoS in August. Conference attendees then vote, based on the novelty of the solutions, which teams should receive the gold, silver, and bronze medals.
The competition reflects what CoSMoS has been about for the past 12 years. CoSMoS is an analytical conference integrating discussions of tools, separations, chemistry, and experimental design, where practitioners gather to debate the best ways to solve complex mixture analysis problems in fields such as pharmaceuticals and health sciences, petroleum, food, and forensic toxicology. The same analytical tools are used in each industry but often with subtly different approaches. Discussing the differences provides a distinct benefit to everyone, giving them practical take-away knowledge.
In brief, the instructions for this year’s Method Development Olympics stated, “Each participant will be sent a container of gummy bears adulterated with an unknown compound. The goal is to identify and quantitate the unknown compound and discover any other pertinent information.” The participants had to figure out that the samples had been coated with indoprofen (2-[4-(1-oxo-1,3-dihydro-2H-isoindol-2-yl) phenyl]propanoic acid). Jeff Kiplinger, this year’s committee lead, cleverly included that last phrase of the instructions, “and discover any other pertinent information,” to test participants’ analytical skills at yet another level-by including chirality as a less obvious aspect of the adulterant.
All participants had to fit the challenge around their work schedules. The competition is designed to weigh heavily on the participants’ abilities to design the experiments and on their skills as analytical scientists. As the competition guidelines say, judging is based on “accuracy of the results, the amount of ancillary information discovered, as well as the creativity of the method development approach.”
Each competitor had access to different analytical tools and support. Gold medalist Ryan Cohen of Merck in Rahway, New Jersey, led a team with expertise in high-throughput analysis, mass spectrometry (MS), nuclear magnetic resonance (NMR) spectroscopy, metals analysis, and drug product analysis.
Lucas Westling, the bronze medalist, works at the Genomics Institute of the Novartis Research Foundation. For him, a really positive part of the competition was exposure to instruments and techniques that he doesn’t normally use in his day-to-day responsibilities. In his case, those were NMR and accurate mass. “I hadn’t used NMR for several years and am still quite a beginner, so the initial sample prep and instrument queuing took some time,” he said. “The accurate mass instrument is used in our lab for metabolite identification but, luckily, I was able to have its operators sit down with me and quickly run a few injections of my crude sample solution.”
Matt Lochansky and colleagues at Catalent Pharma Solutions in Research Triangle Park, North Carolina, entered the competition to showcase their diverse scientific talents and technical capabilities. They also looked forward to the opportunity to work together as a team to tackle an interesting, challenging problem and have fun.
“Because we are a contract development and manufacturing organization, most of our projects are confidential and well-defined in scope, so our scientists rarely have freedom in the selection of techniques and approaches to solve a problem or the opportunity to publish the results,” said Lochansky. “The CoSMoS Method Development Olympics provides a unique opportunity for the scientists to fully leverage their expertise and creativity and compete against the best in the industry.” When his team learned that this year’s challenge was to identify and quantitate an unknown adulterant in gummy bears, they started calling the project “CSI: Catalent.”
The Samples Arrive
Rarely do scientists have either the full range of freedom in their jobs or the necessity to investigate samples in such broad scope. More often, there is some context, such as known starting materials, expected outcomes, and even historical data. Investigators engaged in the Method Development Olympics would likely agree the first step with an unknown is to characterize its gross morphology in true forensic style, allowing one’s experience as an analytical scientist to dictate the next steps while not prejudging outcomes based on scant initial data.
Westling’s response to receiving the samples was typical of the competitors. “There was an initial burst of energy or urgency to get started,” he said. “I had a map in my head of how I was going to proceed, so within several days most of the initial qualitative analyses were finished.” (See Figure 1.)
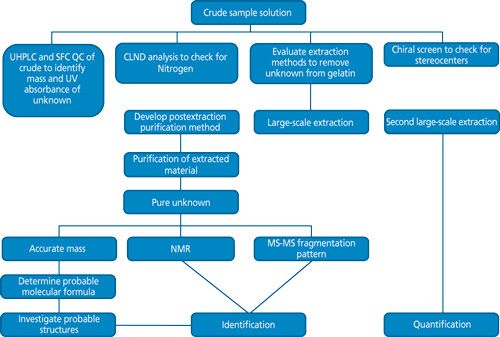
Figure 1: A prospective approach to characterizing an unknown. (Courtesy of L. Westling.)
The Merck team employed spectral imaging to compare the sample with a putative control (Figure 2) based on the information readily at hand and appearance of the sample. The Merck team examined 19 wavelengths using a videometer (Videometer A/S, Hørsholm, Denmark) at a spatial resolution of 45 µm. They noted that no regional differences were seen on the adulterated red seahorse-shaped gummy bear sample. Therefore, at least lengthwise, they were dealing with a homogenous sample.
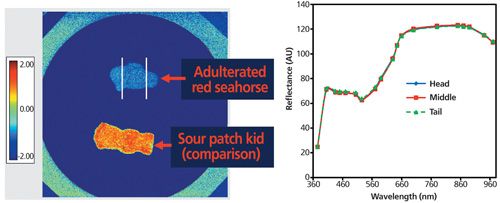
Figure 2: Initial spectral analysis of intact unknown in the gummy bear sample. Left: a heat map showing reflection differences; right: diffuse reflectance of an adulterated red seahorse. (Courtesy of R. Cohen.)
Participants received only five gummy bears, so sample conservation was a big concern, particularly during the initial extraction and quantitation portion of the competition. “I wanted at least two full gummies (ideally three) for the final quantitation,” noted Westling.
The Catalent team was also careful to preserve sample. To do so, they carefully examined the gummy bears under a microscope to look for potential sites where the adulterant could have been injected. Then they carefully dissected and examined the first gummy bear. “We did not find any evidence of injection sites on the surface or the cross sections of the dissected gummy bear,” said Lochansky.
The Initial Analytical Attempts
The initial visual examinations, hypotheses, and suppositions must lead to testing, of course. The results from broad-spectrum analytical tools used in the first phase were typically supplemented by correlative, orthogonal approaches.
Merck developed a series of reversed-phase liquid chromatography–mass spectrometry (LC–MS) analyses to rapidly assess sample composition. Their first step was an achiral ultrahigh-pressure LC (UHPLC) screen. They used six columns: Eclipse Plus C-18, SB-CN, SB-C8, and SB-Phenyl (all from Agilent) and Gold AQ and Gold PFP (from Thermo Scientific), along with six mobile phases. The weak eluents were 0.1% phosphoric acid, 0.1% phosphoric acid + 35 mM potassium hexafluorophosphate, 0.02% perchloric acid with 150 mM sodium perchlorate, and 5 mM ammonium phosphate (dibasic). The strong eluents were acetonitrile and methanol.
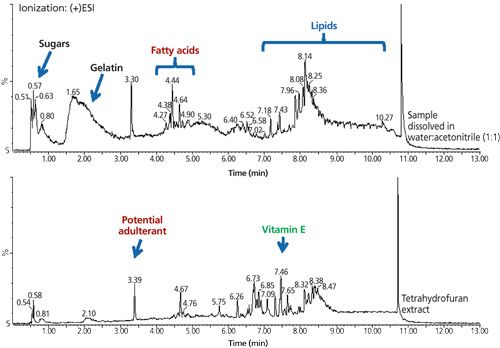
Figure 3: LC–MS and initial method survey by the Merck team. Column: 100 mm x 2.1 mm, 1.7-µm dp BEH-C4-300 Å; mobile-phase A: 0.1% trifluoroacetic acid in water; mobile-phase B: 0.1% trifluoroacetic acid in acetonitrile; flow rate: 0.7 mL/min; gradient: 5-95% B in 8 min, then hold for 2 min; detection: UV absorbance at 210 nm; temperature: 45 °C; injection volume: 3 µL. (Courtesy of R. Cohen.)
They followed with a large-molecule screening step. They used six columns (BEH-C18-135A, C-18-300A, and C4-300A [from Waters Corporation]; SEC125, SEC200-Unix [from Bio-Rad Laboratories]; and SEC300-Zenix [from Sepax]) and six mobile phases. The weak eluents were 0.1% trifluoroacetic acid (aq), 50 mM ammonium acetate, and 50 mM ammonium formate. The strong eluent was 0.1% trifluoroacetic acid in acetonitrile.
According to Lochansky’s colleague and Catalent team captain Wei Pan, who is the director of Strategy and Analytical CMC, the team brainstormed a hypothesis after receiving the description of the challenge. “We thought that the most likely approach to adulterate the candy with a drug is by injecting the drug,” Pan said. The team admitted that their thinking was influenced by the urban legend of adulterated Halloween candy in the Unites States. The team decided to dissolve the gummy bear in water. They also thought it was critical to remove the gelatin to avoid clogging the MS detector (which would result in the team’s being banned from access to the MS instrument in the future). A centrifugal molecular membrane filter was proposed as an easy solution, assuming the adulterant was a small molecule (a safe assumption considering the host conference). Gummy Bear brand candies were purchased from a local grocery store to test the procedure and the resulting solution was analyzed by both gas chromatography–mass spectrometry (GC–MS) and LC–MS. No peaks were observed in the profile. “At this point, we thought we were ready for the real sample,” said Pan.
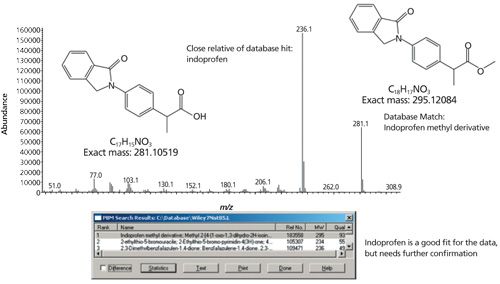
Figure 4: GC–MS yielded a direction but needed further corroborative effort. (Courtesy of M. Lochansky.)
When the real sample gummy bear was placed in cold water, a white precipitate was observed immediately. “This observation was very different from that of the purchased gummy bear,” said Pan. Next, the gummy bear was removed from the suspension, rinsed, and placed into another beaker and dissolved with heated water. The resulting solution was filtered through the centrifugal molecular filter, divided into three portions, and extracted with methylene chloride at neutral, acidic, and basic pH levels. They then analyzed the three portions by GC–MS and LC–MS. “The extract at neutral pH showed vitamin E and fatty acids esters by GC–MS,” the team noted. “At this point, we thought vitamin E was the adulterant and we ordered a vitamin E standard for confirmation by Fourier transform infrared (FT-IR) and Raman spectroscopy.”
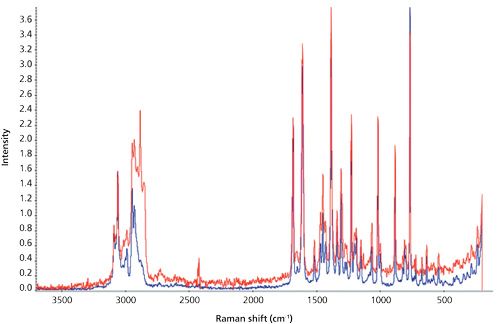
Figure 5: Overlay of outer surface Raman spectrum extracted from CH2Cl2 (red trace) and reference spectrum of suspected adulterant indoprofen (blue trace). (Courtesy of M. Lochansky.)
After the FT-IR and Raman spectra showed only a trace amount of vitamin E and an overwhelming amount of fatty acid, the team became less confident that vitamin E was the adulterant, especially knowing there are vitamin gummy bears available on the market. “This led us to examine the white precipitate,” said Pan.
The amount of precipitate was insufficient for filtering, so an aliquot of the suspension was extracted with methylene chloride and directly injected into a GC–MS instrument. A large peak was present in the profile and was later correctly identified as indoprofen. This result was confirmed by FT-IR and Raman spectral analyses of the surface of a second gummy bear, of reference indoprofen (Sigma-Aldrich), and of a sugar sample from the company break room. These results confirmed that indoprofen was present on the surface.
Even though most teams used NMR to confirm the chemical identity of the adulterant, the Catalent team chose FT-IR and Raman spectroscopy because these are the most commonly used nondestructive techniques in forensic laboratories and law enforcement. “In addition to the confirmation of the chemical identity of the extracted material, we also used Raman microscopy to confirm that the adulteration was indeed on the surface of the gummy bear, not in the interior as hypothesized initially.”
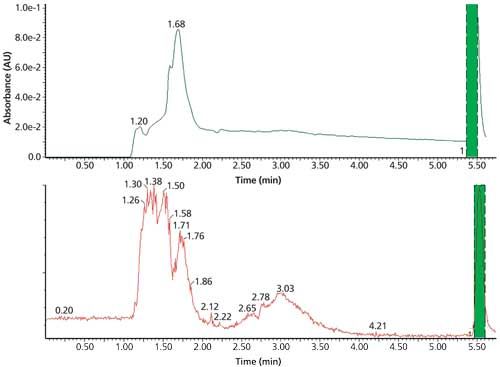
At Novartis, Westling used high-resolution MS as well as NMR to achieve a finely detailed characterization of the sample. In addition to using supercritical fluid chromatography (SFC) as an orthogonal method, and taking the novel approach of using chromatographic anion analysis to check for post-purification salt formation, Westling used evaporative light scattering detection (ELSD) and chemiluminescence nitrogen detection (CLND). Westling also invested in preparative chromatography work (Figure 6) using a mass-triggered preparative LC–MS system to develop scaled-up extracted material in view of possible matrix complexities and to compensate for having so little information about the sample at the outset.
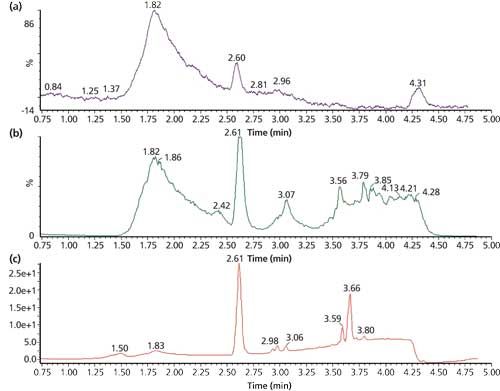
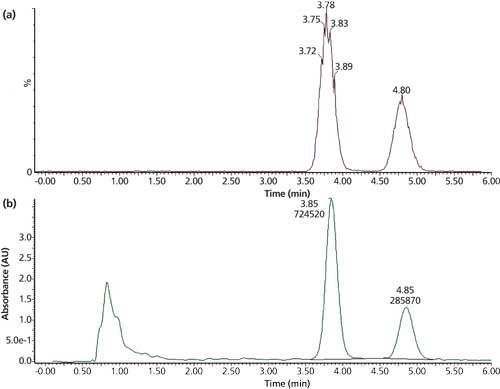
The team at Merck conducted a thorough and detailed examination of the samples, displaying their skills with the range of equipment they had access to. High-resolution MS-MS and chiral techniques were used for aspects of characterization in addition to NMR, which is classically used to confirm what might be found by other techniques. The Merck team’s results (Figure 9) further indicated that the indoprofen was not observed in the sample core but only on the surface.
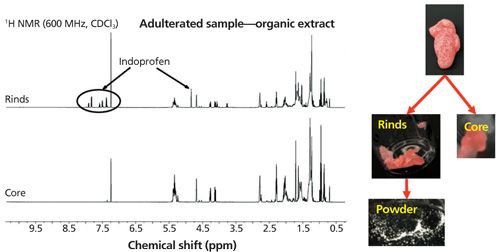
Figure 9: NMR confirmation that adulteration was only present on the sample surface. (Courtesy of R. Cohen.)
Further characterization by the group at Merck displayed a rather pleasing cyclical conclusion: They refined the morphological characterization they did at the beginning of the exercise by using scanning electron microscopy (SEM), as shown in Figure 10.

Figure 10: Refined scanning electron microscopy (SEM) of the adulterated samples and a control indicated changes in the surface were present. (Courtesy of R. Cohen.)
Because the scope of this narrative is necessarily limited, much of the elegant work done by the teams has been omitted here. Details about the work the teams did using conformational analysis and electronic circular dichroism (ECD) calculations, characterization of additional unknowns in the samples, spectral deconvolution of proton NMR using a complete reduction to amplitude-frequency table (CRAFT), quantitative summaries, and more, can be reviewed on the CoSMoS website (www.CoSMoScience.org). Links to each team’s presentation can be found in the right-most panel of the home page.

At the conclusion of this year’s Method Development Olympics, it was clear from the enthusiasm of the attendees, the range of questions, and the engagement, that these three teams represented the best of the best, making it hard to rank them. They all brought valuable experience to share with their colleagues. In the end, the bronze medal went to the Novartis team, the silver to the Catalent team, and the gold to the Merck team.
Conclusion: Big Benefits for Participants
It is clear from the comments made by the participants that they found the experience rewarding in many ways. Lucas Westling of the Novartis team noted that being chosen as a finalist offered an opportunity to present his team’s work to an audience in a relatively stress-free setting. “I say ‘relatively stress-free,’ because I am terrified of speaking in front of groups,” he admitted. “The CoSMoS conference offers a modestly sized, interested, and engaged audience that may help one feel more comfortable in a presentation setting.”
Wei Pan noted that his team’s silver medal has been viewed as a huge success within Catalent, and the story made headline news on the Catalent intranet. The team then held a special Lunch-and-Learn session about their experience. “The exercise demonstrated that when people with different training and skills work together as a team, amazing things can happen and science can be fun,” said Pan. After the news story ran on the company’s intranet, he and his teammates received questions from scientists at other Catalent sites about how to participate in the next competition. “There may be multiple Catalent teams competing in the CoSMoS Method Development Olympics next year,” he said.
The benefits of teamwork that Pan mentioned were also important to the Merck team. “The main reason Merck became involved was that it would foster better interaction between the specialty groups (NMR, MS, and high-throughput analysis) and the drug product analytical group,” said Ryan Cohen. “It’s being looked at as a huge success story internally, as a very good example of collaboration. I’m really glad that you guys are running this and we could be a part of it.”
Michael Balogh is a strategic technology development consultant. A former consulting principal scientist at Waters Corporation and Director of Strategic Relations, he has held the position of adjunct professor and visiting scientist at Roger Williams University and has been a reviewer for grant proposals for the National Science Foundation (NSF). Direct correspondence to: mpbalogh@gmail.com

Troubleshooting Everywhere! An Assortment of Topics from Pittcon 2025
April 5th 2025In this installment of “LC Troubleshooting,” Dwight Stoll touches on highlights from Pittcon 2025 talks, as well as troubleshooting advice distilled from a lifetime of work in separation science by LCGC Award winner Christopher Pohl.
Study Examines Impact of Zwitterionic Liquid Structures on Volatile Carboxylic Acid Separation in GC
March 28th 2025Iowa State University researchers evaluated imidazolium-based ZILs with sulfonate and triflimide anions to understand the influence of ZILs’ chemical structures on polar analyte separation.












