GC×GC–TOF-MS and Comprehensive Fingerprinting of Volatiles in Food: Capturing the Signature of Quality
LCGC Asia Pacific
Chemical fingerprinting can provide evidence for quality differences resulting from botanical and geographical origins of primary food ingredients, post-harvest practices, production processes (such as traditional versus industrial processes), and the shelf-life evolution of finished products. This article discusses the strategic role and potential of comprehensive two-dimensional gas chromatography (GC×GC) combined with time-of-flight mass spectrometry (TOF-MS) and pattern recognition using template matching for data processing to unravel the quality traits of high-quality food products. Practical examples dealing with high-quality cocoa and extra-virgin olive oil are described.
Chemical fingerprinting can provide evidence for quality differences resulting from botanical and geographical origins of primary food ingredients, post-harvest practices, production processes (such as traditional versus industrial processes), and the shelf-life evolution of finished products. This article discusses the strategic role and potential of comprehensive two-dimensional gas chromatography (GC×GC) combined with time-of-flight mass spectrometry (TOF-MS) and pattern recognition using template matching for data processing to unravel the quality traits of high-quality food products. Practical examples dealing with high-quality cocoa and extra-virgin olive oil are described.

After the second industrial revolution the need for the standardization of quality for food production led to the introduction of periodic inspections, standardization of procedures, and quality controls (1,2). The International Organization for Standardization (ISO) 9000 is probably the best known modern international standard for quality management (3). Current European Union (EU) policy on food quality aims to protect products characterized by unique features, linked to their geographical origin as well as traditional expertise. With this policy, product names are granted by a geographical indication (GI), a protected geographical indication (PGI), or a protected designation of origin (PDO), if they have specificities strictly linked to the place where they are produced, including the compositional characteristics of raw materials, the climate, and the traditional processes of manufacturing and transformation (4).
An analytical platform or method capable of capturing the chemical traits of a food that are related to its perceived quality-mainly sensory quality, raw material authenticity, and processing impact-will contribute to the quality assessment process while providing a foundation for consumer-tailored strategies to improve a product’s acceptance and loyalty.
Chemical fingerprinting can provide evidence for quality differences arising from botanical and geographical origins of primary ingredients, postâharvest practices, production processes (traditional versus industrial), and shelf-life evolution of finished products. Illustrative examples on how comprehensive chemical fingerprinting processes can be strategically valuable for characterizing the quality traits of food from our recent research are highlighted here. This article focuses on the potentials of comprehensive two-dimensional gas chromatography coupled to time-of-flight mass spectrometry (GC×GC–TOF-MS) to generate highly informative fingerprints of chemicals from complex fractions, including potent odourants responsible for the aroma signature, and technological markers that indicate the impact of the production process. When complex two-dimensional (2D) patterns of chemicals are explored by dedicated pattern recognition algorithms, a high level of information about sensory profile, product authenticity, and technological impact is revealed (5,6).
The first example deals with high-quality cocoa, a food commodity of global economic interest, and the second example deals with extra-virgin olive oil, an important local commodity in many Mediterranean countries, including Spain, Italy, Greece, and Tunisia. Both of these commodities have an intrinsic “added value” related to their flavour profile and perceived quality that are 80–90% a result of aroma-active compounds (7).
Experimental Procedures
Chemicals and Samples: The mixture of n-alkanes (n-C9 to n-C25) for calibrating linear retention indices (IT) was from Sigma-Aldrich. The IT solution was prepared in cyclohexane at a concentration of 100 mg/L.
Cocoa samples were from Gobino srl and were selected on the basis of their peculiar sensory profile from high-quality productions of different geographic origins. Processing was performed in three replicated batches using time and temperature protocols from 100–130 °C between 20–40 min. Processing was optimized for each origin and driven by a desirable flavour development. Hot-air roasting was conducted in a vertical roaster designed by Bühler AG.
Cocoa samples were frozen in liquid nitrogen immediately after each step of processing and then stored at -80 °C. Before headspace analysis, samples were ground in a laboratory mill up to approximately 300 µm (Grindomix GM200, Retsch); particle size homogeneity was verified by visual inspection.
Extra-virgin olive (EVO) oil samples were collected within the Italian “Violin” Project (Valorization of Italian OLive products through INnovative analytical tools – AGER Fondazioni in rete per la Ricerca Agroalimentare) and selected for their sensory profile by an expert panel. Examples cited in this paper refer to a commercial EVO oil with a PGI quality label (Azienda Agricola Mori Concetta, PGI Toscano, olives Mariolo cultivar, San Casciano in Val di Pesa, Firenze, Italy) and a PDO product (Azienda Agricola Leone Sabino, Don Gioacchino Gran Cru, DOP Terra di Bari Castel del Monte, 100% Coratina olives cultivar, Canosa di Puglia, Italy).
Headspace Solid-Phase Microextraction (SPME) Devices and Sampling Conditions: A divinylbenzene/carboxen/polydimethylsiloxane 1-cm SPME fibre from Supelco was used for HS-SPME sampling. The standard in-fibre procedure (8) was adopted to preload the IS (α-thujone) onto the fibre before sampling. A 5.0-µL solution of IS (α-thujone at 100 mg/L in diethyl phthalate) was placed into a 20-mL glass vial and subjected to HS-SPME at 50 °C for 5 min. After the IS loading step, the SPME device was exposed to 500 mg of cocoa in a headspace glass vial (20 mL) for 30 min at 50 °C or 100 mg of olive oil in a headspace glass vial (20 mL) for 60 min at 40 °C. Extracted analytes were recovered by thermal desorption of the fibre into the S/SL injection port of the GC system at 250 °C for 5 min.
GC×GC–TOF-MS Conditions: GC×GC analyses were performed on an Agilent 7890B GC system coupled with a Bench TOF-Select system (Markes International) featuring Tandem Ionization that provides variable-energy electron ionization. The ion source and transfer line were set at 270 °C. The MS optimization option was set to operate with a mass range between 40–300 m/z; data acquisition frequency was 50 Hz for each channel; filament voltage was set at 1.60 V. Electron ionization energies explored were 70 and 12 eV.
The system was equipped with a two-stage KT 2004 loop thermal modulator (Zoex Corporation) cooled with liquid nitrogen controlled by Optimode V.2 (SRA Instruments). The hot jet pulse time was set at 250 ms, modulation period (PM) was 4 s for cocoa and 3.5 s for olive oil, and cold-jet total flow was progressively reduced with a linear function from 40% of mass flow controller (MFC) at initial conditions to 8% at the end of the run.
GC×GC Columns and Settings: The column set was configured as follows: 1D SolGel-Wax column (100% polyethylene glycol; 30 m × 0.25 mm dc, 0.25-μm df) from SGE Analytical Science coupled with a 2D OV1701 column (86% polydimethylsiloxane, 7% phenyl, 7% cyanopropyl; 2 m × 0.1 mm dc, 0.10-μm df) from J&W (Agilent). SPME thermal desorption into the GC injector port was under the following conditions: split–splitless injector in split mode at 250 °C, split ratio 1:20. The carrier gas was helium at a constant flow of 1.3 mL/min. The oven temperature program was from 40 °C (2 min) to 240 °C at 3.5 °C/min (10 min).
The n-alkanes liquid sample solution for ITS determination was analyzed under the following conditions: split–splitless injector in split mode, split ratio: 1:50, injector temperature: 250 °C, and injection volume: 1 µL.
Raw Data Acquisition, 2D-Data Processing and Statistics: Data were acquired by TOF-DS software (Markes International) and processed using GC Image ver 2.8 (GC Image, LLC). Statistical analysis used XLStat (Addinsoft).
Results and Discussion
Cocoa Origin and its Distinctive Chemical Signature: Cocoa (Theobroma Cacao L. Malvaceae family) is the main raw ingredient for chocolate production (9). It is native to tropical forests of the South American continent, although recent statistics indicate that most of the production is concentrated in Africa with Cote d’Ivoire and Ghana covering about 56% of the global production, followed by Indonesia (12%), Nigeria (6%), and Cameroon (6%).
Cocoa and chocolate are considered comfort foods and are consumed worldwide for their pleasant sensory profile of unique and complex flavours. This complexity arises from multiple interconnected biochemical and chemical reactions occurring at post-harvest stage where pedoclimatic conditions and farming practices play a major role (10,11). Later in the processing chain, roasting, conching, and tempering develop the flavour profile and the distinctive sensory signature of chocolate (12–14).
Cocoa’s complex aroma is modulated by a series of potent odourants (15–17), whose specific qualiâquantitative distribution within the bulk of several hundreds of volatiles has been identified as a distinctive aroma signature also referred to as an aroma blueprint. From this perspective, GC×GC–MS would be the analytical technique of choice for an accurate and informative fingerprinting of such a complex fraction. Deeper insights on the quali-quantitative distribution of volatiles would help in delineating an origin-specific aroma blueprint, inform about the seasonal variations or the effect of climate changes, or help chocolate manufacturers in designing tailored blends evoking peculiar aroma notes.
GC×GC–MS exploits the potential of two separation dimensions with the additional orthogonal information provided by MS. This results in: (i) increased separation power, (ii) meaningful 2D patterns with analytes structurally ordered in the chromatographic space, and (iii) enhanced sensitivity as a result of band focusing in space obtained by cryogenic modulation (18–20).
In a recent study, we investigated the unique signatures of volatiles from commercial, high-quality cocoa intermediates, with a pattern recognition strategy based on GC×GC–MS that extends the investigation potential to both untargeted and targeted analytes (21). The approach is based on the template matching principle and is named untargeted/targeted (UT) fingerprinting (22,23). Samples were from different geographical provenience (Mexico, Ecuador, Venezuela, Colombia, Java, Trinidad, and Sao Tomè), selected by experts for their unique aroma profiles and were studied along early steps of processing as raw, roasted, steamed, and grinded nibs. For some origins, cocoa liquor was also included to evaluate its aroma signature for chocolate products design.
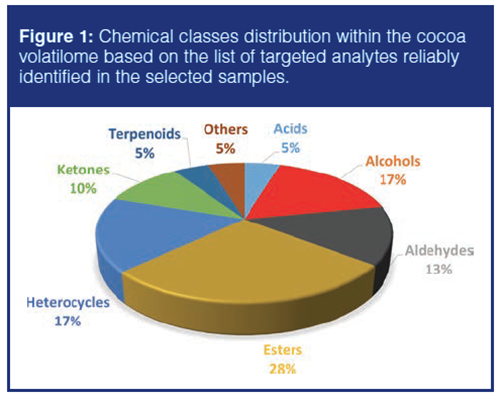
The fraction of volatiles was extracted by automated headspace solid-phase microextraction (HS-SPME) and on-line analyzed by GC×GC–MS in a system equipped with a loop-type thermal modulator. Within 595 detectable analytes, delineated by unique 2D peak-regions and covering most of the chemical dimensions of cocoa volatilome, about 200 compounds were tentatively identified on the basis of 1D Linear Retention Indexes (IT) and MS spectral similarity with authentic standards or with spectra collected in commercial (Wiley 7n and NIST 2015) and in-house databases (23). Figure 1 shows chemical classes distribution within the cocoa volatilome. Esters dominate the volatile fraction and, together with alcohols and acids, bring information about fermentation and its impact on primary metabolites (mainly sugars and amino acids). Within the heterocycles, the subset of alkyl pyrazines is of great relevance because they provide information about the technological impact on some precursors present in raw cocoa, and for the most odourâactive compounds, they bring the earthy/roasty notes to the global aroma. Alkyl pyrazines are formed from the early stages of processing during bean drying and later by roasting and steaming (9). Carbonyls (aldehydes and ketones) are formed mainly from fatty acids precursors by oxidative (chemical and enzymatic) reactions. The sub-group of Streker aldehydes (2- and 3-methylbutanal, methylpropanal, and phenylacetaldehyde) are fundamental for cocoa and chocolate flavour modulating the malty, buttery, and honey-like notes.
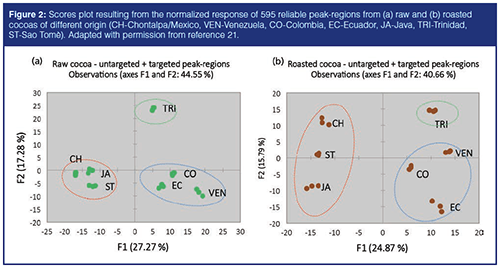
The quali-quantitative distribution of known and unknown volatiles, for example, the volatile metabolome fingerprint, is potentially informative, and helps in the discrimination and differentiation of geographical origin and manufacturing stage. As an example, unsupervised multivariate analysis, such as principal component analysis (PCA), can be applied to reveal the natural conformation (groups) of the analyzed samples and helps in localizing informative chemical features responsible for cocoa discrimination. Figures 2(a) and 2(b) show the scores plot resulting from the normalized response of 595 reliable peak-regions from raw (2[a]) and roasted (2[b]) cocoas of different origin (CH-Chontalpa/Mexico, VEN-Venezuela, CO-Colombia, EC-Ecuador, JA-Java, TR-Trinidad, ST-Sao Tomè). Samples are grouped into three main clusters: EcuadorâVenezuela-Colombia (blue circles), Chontalpa/Mexico-Sao Tomè-Java (red circles), and Trinidad (green circles) (21). The total explained variability represented by the first two principal components (F1 and F2) is about 44% for raw and 41% for roasted beans.
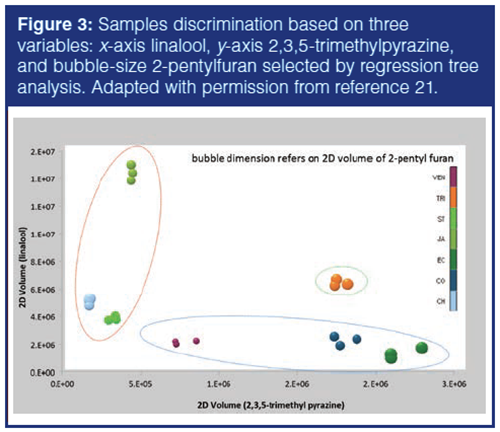
Supervised approaches, at this stage, may help in defining or selecting highly discriminating variables. A classification model based on three variables, 2-pentylfuran, 2,3,5-trimethylpyrazine, and linalool, is capable of discriminating between cocoa nib origins. Figure 3 shows how samples could be discriminated in the Cartesian space of three variables: x-axis linalool, y-axis 2,3,5-trimethylpyrazine, and bubble-size 2-pentylfuran (21). This model confirms what unsupervised exploration by PCA showed (Figure 2[b]). Samples from Ecuador and Colombia are aligned along the x-axis (low linalool content) together with the Venezuela samples. Java is characterized by a low pyrazines signature (2,3,5-trimethylpyrazine is one of the most origin sensitive), but clustered together with Chontalpa/Mexico and Sao Tomè for their lower amount of linalool. Trinidad has an intermediate position between the two groups, but with a relatively high amount of 2-pentylfuran and trimethyl pyrazine.
Key aroma compound signatures (15–17) are of particular interest for perceived quality. These signatures are buried within the bulk of the cocoa volatilome, but their information is strategic for the confectionery industry and can be used in new origin selection and blending. Key aroma compounds include alkyl pyrazines (2,3,5-trimethylpyrazine, 2-ethyl-3,5-dimethylpyrazine, and 3,5-diethyl-2-methylpyrazine), which are responsible for the earthy notes; and short-chain and branched fatty acids (acetic acid, butanoic acid, 2-methylpropanoic acid, and 3-methylbutanoic acid), whose presence at high concentrations can impart off-flavours as a result of their rancid, sour, and sweaty notes. Strecker aldehydes (2- and 3-methylbutanal), formed during fermentation and roasting, impress malty and buttery notes, and phenylacetaldehyde, derived from L-phenylalanine (L-Phe), imparts a pleasant honey-like note.
Other key-aromas are esters (ethyl-2-methylbutanoate: fruity; 2-phenylethyl acetate: flowery), linear alcohols (2-heptanol: citrusy), phenyl propanoids derivatives (2-phenylethanol: flowery), and sulphurous-derived compounds (dimethyl trisulfide).
Accurate quantitation of these analytes, performed by multiple headspace extraction SPME–GC–MS/flame ionization detection (FID) (24), are visualized as a heat map for a subset of origins in Figure 4. Odourants were quantified by external calibration and FID-predicted relative response factors. Amounts are reported for nibs and cocoa mass in Figure 4(a) and the odour activity value (OAV) is shown in Figure 4(b). OAV is computed as the coefficient of the concentration of an odourant (mg/kg) versus its odour threshold (mg/kg). OAV is a useful parameter for discriminating odourants from interfering components. Below an OAV of 1, which is generally used as a threshold value, it is assumed that an odourant does not play a role in eliciting its characteristic quality. However, several more parameters need to be considered to judge odour activity of volatiles. The capture of chemical complexity of volatile patterns by comprehensive approaches is currently one of the most effective strategies available (25).
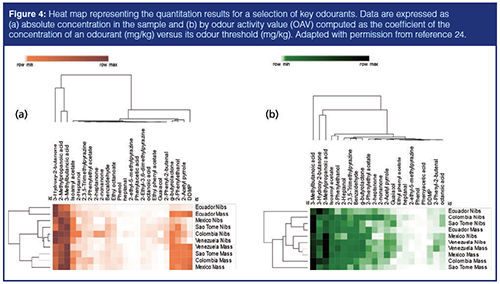
Hierarchical clustering based on Euclidean distances helps in sample discrimination based on their aroma profiles. Key odourants, such as 2-methylpropanic acid, 3-methylbutanoic acid, acetoin, and 2-phenylethanol, show a homogeneous trend across all samples. A similar behaviour is seen for other odourants, such as isoamyl acetate, γ-butyrolactone, and 2-acetyl pyrrole, that cluster independently from the others. As expected, 2,3-dihydro-3,5-dihydroxy-6-methyl(4H)-pyran-4-one (DDMP) is an effective marker of processing (21): its concentration in cocoa mass is, on average, two orders of magnitude higher than in cocoa nibs.
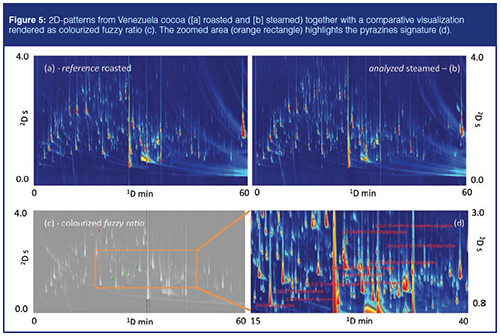
To confirm the great flexibility of GC×GC in cocoa volatilome fingerprinting, it is interesting to explore the effect of steaming on roasted cocoa beans with simple and intuitive tools. Steaming is conducted on shelled cocoa beans after roasting. It is an on-line process conducted with overheatedâsteam that lowers the bacterial charge of the beans. However, steaming impacts cocoa aroma and its process parameters have to be carefully tuned to avoid off-flavour formation or the loss of key odourants. From this perspective, comparative visualization based on datapoint features (26) could be of great help. This is a pointwise approach where chromatograms are compared pointâbyâpoint or pixelâbyâpixel. In a GC×GC–MS chromatogram, every datapoint corresponds to a detector event. With this approach, each datapoint is a feature and therefore datapoint features at the same retention times are implicitly matched. Figure 5 shows the 2D-patterns from Venezuela cocoa (Figure 5[a] roasted and Figure 5[b] steamed). Their comparative visualization is rendered as colourized fuzzy ratio (Figure 5[c]); within the zoomed area (orange rectangle), the pyrazines signature is visible (Figure 5[d]). This approach gives prompt information on analytes (relative) variations over the patterns and, by colour codes (green, red, and grey), informs about their higher or lower abundance in the samples. In this specific case, steaming has an impact on pyrazines quantitative distribution; those with short alkyl substituents are coloured by green pixels indicating that they were more abundant in the analyzed image (steamed beans). Conversely, red coloured pixels indicate analytes and chromatographic regions where the detector response was higher in the reference (roasted beans) image.
Italian Extra-Virgin Olive Oil: How to Capture Volatiles Chemical Complexity
Consumers appreciate high-quality olive oil, whether virgin (VO) or EVO oil, for its health benefits and pleasant and distinctive flavour. The objective quality and purity of this product is regulated by international organizations, including the EU, International Oil Council (IOC), and Codex Alimentarius. If from one side, EVO oil adulteration can be assessed by characterizing the nonvolatile fraction, for example, fatty acid methyl esters (FAMEs) profiles, sterol and triterpene dialcohol composition, wax content, and presence of conjugated dienes and trienes, the aroma quality can be evaluated by panel testing without any objective analytical protocol supporting the sensory assessment.
Olive oil production is located mainly in Europe with Spain, Italy, and Greece as the key players. Italian production, which is estimated at approximately 50% of Spanish production, is characterized by a relatively high number of PDO oils, with 46 registered products compared with 31 for Spain (European Commission – DOOR database http://ec.europa.eu/agriculture/quality/door/list.html). Quality labelling gives consumers a sense of tradition while improving trust and confidence for a high-quality product. Although these products must comply for higher quality standards compared to ordinary production, the EU Parliament, through the resolution of 14 January 2014 on “food crisis, fraud in the food chain and the control thereof”, revealed that olive oil is, among others, one of the top foods subjected to adulteration. Consequently, the EU action plan focused on specific aspects related to quality, control, and promotion to counteract frauds and illicit operations.
Undoubtedly, quality control issues are partially related to the lack of powerful and informative analytical methods capable of supporting objective classification of olive oils, in particular for the distinction between VO and EVO oils based on their aroma signatures. Olive oil is currently the only food product with officially regulated sensory attributes; standardized protocols (27,28) for sensory assessment are established by law and highly trained panellists are involved in this task. Based on the presence or absence and the intensities of coded defects and, on the other side, of “fruity” perception, evaluated by smelling and tasting, virgin olive oil is then classified into three categories: EVO, VO, and lampante oil.
In this scenario, GC×GC–MS represents an analytical tool with great potential to describe the chemical signature of sensory quality markers, including potent odourants related to oil-coded defects, that is fusty/muddy sediment, musty/humid/earthy, winey/vinegary, or rancid. Previous studies by our research team explored and confirmed the possibility of delineating meaningful patterns of potent odourants eliciting the coded defects in olive oils (5). At the same time, thanks to the separation power and enhanced resolution achievable with a comprehensive 2D-GC analysis, additional chemical information can be collected and rationalized before correlating it with oil sensory attributes, origin (29,30), or ripening status of olives (6).
The volatile fraction of olive oil is complex and connoted by high chemical dimensionality (31), a parameter defined by Giddings to describe the degree of order or disorder in multidimensional separations. The presence of several chemical functionalities, also represented by homologous series, generated by the multiple chemical and biochemical reactions occuring to olives primary metabolites, results in complex 2D patterns that require, for accurate fingerprinting, high-resolution separations and orthogonal detection by mass spectrometry. MS is fundamental here to extract the information on analyte fragmentation patterns for a reliable identification. Figure 6(a) shows the 2D chromatogram of a PGI Toscano EVO oil. The number of detectable 2D-peaks with a signal-to-noise ratio (S/N) threshold of 100 is about 750 and, for 180 of them, reliable identification was possible by matching 1D IT and MS spectrum with those collected in commercial and in-house databases (5,6). Reference compounds were also adopted in the case of key odourants related to positive attributes or coded defects.
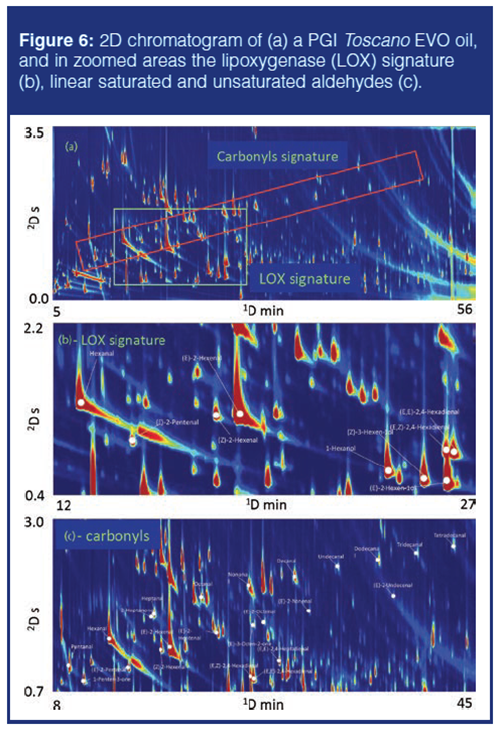
Within the separated volatiles, the lipoxygenase (LOX) signature (Figure 6[b]) is fundamental to define fresh-green and fruity notes, the positive attributes. C6 unsaturated alcohols and aldehydes, (Z)-2-hexenal, (E)-2-hexenal, hexanol, (Z)-3-hexenol, (E)-2-hexenol, (E,Z)-2,4-hexadienal, and (E,E)-2,4-hexadienal, are formed from linoleic and linolenic acids oxidative cleavage. Figure 6(c) illustrates the linear saturated and unsaturated aldehydes together with a few ketones likely representing hydroperoxides (that is, primary products of lipid oxidation) cleavage products. This last group of analytes informs generally about shelfâlife evolution, with increasing concentrations of potent odourants that bring rancid and fatty notes.
For an accurate and informative fingerprinting of EVO oil volatiles, an additional dimension at the detection level could be of help. Instruments capable of acquiring variableâenergy electron ionization spectra by timeâswitching between two ionization energies across every single analytical run represent an interesting option to extend method dimensionality. Research on variable-energy electron ionization spectra acquisition also showed benefits for the identification of large isomeric species in unresolved complex mixtures (UCMs) of motor oil samples (32) and for light volatile organic compounds (VOCs) from human blood (33). The operation of the ion source at low energies (12–16 eV) allowed enhanced intensity for structure-indicating fragments, which can improve method specificity.
Recently, Freyre et al. (34) proposed a tile-based Fisher ratio analysis and a discovery-based investigation strategy to detect exogenous analytes (spiked at 50 ppm) in diesel fuel samples. Their strategy was based on data processing after tandem signals fusion. Our research group explored the cocoa complex volatilome by GC×GC–TOF-MS and variableâenergy electron ionization proposing a novel approach based on the UT fingerprinting strategy but performed on fused data streams (hard and soft ionization energies together) for effective crossâcomparative analysis of samples (23).
Tandem data streams are complementary in terms of both spectral information/fragmentation pattern dissimilarity and absolute response. The first characteristic is of help when 70 eV spectra lack molecular ions and structurally informative fragments, while the differential response from the two channels opens new perspectives in terms of dynamic range and linearity. Although at lower energies (12–16 eV), the absolute number of ionized molecules is reduced, benefits are evident for background noise intensity and S/N. The latter benefit is accrued for analytes showing a reduced fragmentation at lower energies.
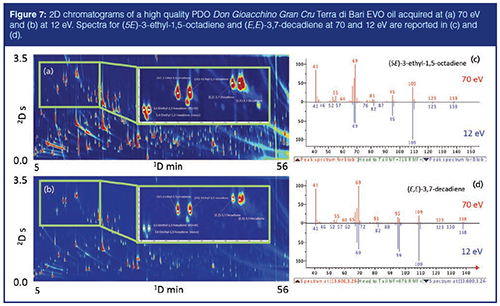
Figure 7 shows the volatile 2D pattern of a high-quality PDO Don Gioacchino Gran Cru Terra di Bari EVO oil acquired at 70 eV (Figure 7[a]) and at 12 eV (Figure 7[b]). The enlarged areas correspond to the elution region of unsaturated alkanes characteristic of early stages of olive ripening (35). They include: 3,4-diethyl-1,5-hexadiene (RS + SR), 3,4-diethyl-1,5-hexadiene (meso), (5Z) and (5E)-3-ethyl-1,5-octadiene, (E,Z)- and (E,E)-3,7-decadiene, and (E)-4,8-dimethyl- 1,3,7-nonatriene (6,35). Spectra for (5E)-3-Ethyl-1,5-octadiene and (E,E)-3,7-decadiene at 70 and 12 eV are reported in Figures 7(c) and 7(d).
Conclusions
The concept of quality of food is complex and connoted by different meanings. Food compositional complexity offers the opportunity, through informative and reliable analytical protocols, to objectify the most relevant characteristics of quality. Multidimensional analytical platforms, and in particular those implementing comprehensive twoâdimensional separations with mass spectrometry, have the intrinsic potential of delineating meaningful chemical fingerprints that can be explored for their targeted and untargeted feature distribution.
To achieve the higher level of information exploiting the quality concepts related to food origin, harvest practices, technological processes impact, and flavour profile, meaningful analyte patterns must be delineated. Data processing tools are therefore fundamental. “Flexibility” is a key characteristic for algorithms and workflows, and data patterns should be explored from different perspectives in relation to the objectives. If the full pattern-for example, the chemical signature-of untargeted and targeted analytes may help in describing chemical differences between samples of different origin, such as cocoa origins characterization, alternatively, datapoint features with visual comparisons may help in locating single analyte variations between sample pairs, such as cocoa processing.
Algorithm flexibility should be accompanied by adequate tools for data alignment, with effective transform functions (36) that support re-alignment of datasets acquired across wide time ranges, in analytical batches affected by random instrumental fluctuations, or with multidimensional detectors, with variable-energy electron ionization TOF-MS, for example, for EVO oil volatile patterns.
In the scenario of linking meaningful chemical signatures to food quality concepts, the role of data processing software is central. Analysts should drive the exploration of complexity with simple and intuitive tools with an understanding of how data is pre-processed and treated along the steps of the data analysis workflow to fully understand and appropriately use results.
Interestingly, these research efforts have broken down barriers between academic and industrial research, indicating that these analytical tools and data mining concepts are not only academic exercises and speculations, but can also be concrete strategies to improve competitive advantages in a complex food market.
Acknowledgements
The research on cocoa was carried out thanks to the financial support of Firmenich S.A. Geneva, Switzerland. The research on olive oil was supported by Progetto Ager – Fondazioni in rete per la riscerca agroalimentare. Project acronym Violin - Valorization of Italian olive products through innovative analytical tools.
Referencesâ©
- J.C. Wood and M.C. Wood, Eds., Henry Ford: Critical Evaluations in Business and Management (Routledge, London, UK, and New York, USA, 2003).
- J. Papp, Quality Management in the Imaging Sciences (Elsevier Health Sciences, 2014), p. 372.
- www.iso.org
- https://ec.europa.eu/info/food-farming-fisheries/food-safety-and-quality/certification/quality-labels_en
- G. Purcaro, C. Cordero, E. Liberto, C. Bicchi, and L.S. Conte, J. Chromatogr. A1334, 101–111 (2014).
- F. Magagna, L. Valverde-Som, C. Ruíz-Samblás, L. CuadrosâRodríguez, S.E. Reichenbach, C. Bicchi, et al. Anal. Chim. Acta936, 245–258 (2016).
- C. Spence, Psychologist 23(9), 720–723 (2010).
- Y. Wang, J. O’Reilly, Y. Chen, and J. Pawliszyn, J. Chromatogr. A1072(1), 13–17 (2005).
- A.C. Aprotosoaie, S. Vlad Luca, and A. Miron, Compr. Rev. Food Sci. Food Saf.15(1), 73–91 (2016).
- N. Camu, T. De Winter, S.K. Addo, J.S. Takrama, H. Bernaert, and L. De Vuyst, J. Sci. Food Agric.88(13), 2288–2297 (2008).
- G.V. de M. Pereira, V.T. Soccol, and C.R. Soccol, Current Opinion in Food Science7, 50–57 (2016).
- J.E. Kongor, M. Hinneh, D. Van de Walle, E.O. Afoakwa, P. Boeckx, and K. Dewettinck, Food Res. Int.82, 44–52 (2015).
- R. Nazaruddin, H. Osman, S. Mamot, S. Wahid, and A. Nor, J. Food Process Preserv.30, 280–298 (2006).
- R. Saltini, R. Akkerman, and S. Frosch, Food Control 29(1), 167–187 (2013).
- P. Schnermann and P. Schieberle, J. Agric. Food Chem. 45(3), 867–872 (1997).
- F. Frauendorfer and P. Schieberle, J. Agric. Food Chem.54, 5521–5529 (2006).
- F. Frauendorfer and P. Schieberle, J. Agric. Food Chem.56, 10244–10251 (2008).
- H.J. Cortes, B. Winniford, J. Luong, and M. Pursch, J. Sep. Sci.32(5–6), 883–904 (2009).
- M.S. Klee, J. Cochran, M. Merrick, and L.M. Blumberg, J. Chromatogr. A1383, 151–159 (2015).
- M. Adahchour, J. Beens, and U.A.T. Brinkman, J. Chromatogr. A1186(1–2), 67–108 (2008).
- F. Magagna, A. Guglielmetti, E. Liberto, S.E. Reichenbach, E. Allegrucci, G. Gobino, et al., J. Agric. Food Chem.65(30), 6329–6341 (2017).
- S.E. Reichenbach, P.W. Carr, D.R. Stoll, and Q. Tao, J. Chromatogr. A 1216(16), 3458–3466 (2009).
- C. Cordero, A. Guglielmetti, C. Bicchi, E. Liberto, L. Baroux, P. Merle, et al., J. Chromatogr. A (in press) https://doi.org/10.1016/j.chroma.2019.03.025
- C. Cordero, A. Guglielmetti, B. Sgorbini, C. Bicchi, E. Allegrucci, G. Gobino, et al., Anal. Chim. Acta1052, 190–201 (2019).
- C. Cordero, J. Kiefl, S.E. Reichenbach, and C. Bicchi, TrAC - Trends in Analytical Chemistry113, 364–378 (2019).
- S.E. Reichenbach, X. Tian, C. Cordero, and Q. Tao, J. Chromatogr. A1226, 140–148 (2012).
- Commission of the European Communities, Commission Regulation (Eec) No 2568/91 (Official Journal of the European Communities. 1991), pp. 1–83.
- International Oil Council, COI/T.20/DOC.15/Rev.10 Sensory Analysis of Olive Oil - Method for the Organoleptic Assessment of Virgin Olive Oil (2018).
- I. LukiÄ, S. Carlin, I. Horvat, and U. Vrhovsek, Food Chem.270, 403–414 (2019).
- L.T. Vaz-Freire, M.D.R.G. da Silva, and A.M.C. Freitas, Anal. Chim. Acta 633(2), 263–270 (2009).
- J.C. Giddings, J. Chromatogr. A703(1–2), 3–15 (1995).
- M.S. Alam, C. Stark, and R.M. Harrison, Anal. Chem.88(8), 4211–4220 (2016).
- L.M. Dubois, K.A. Perrault, P.H. Stefanuto, S. Koschinski, M. Edwards, L. McGregor, et al., J. Chromatogr. A1501, 117–127 (2017).
- C.E. Freye, N.R. Moore, and R.E. Synovec, J. Chromatogr. A1537, 99–108 (2018).
- F. Angerosa, L. Camera, N. D’Alessandro, and G. Mellerio, J. Agric. Food Chem.46(2), 648–653 (1998).
- D.W. Rempe, S.E. Reichenbach, Q. Tao, C. Cordero, W.E. Rathbun, and C.A. Zini, Anal. Chem. 88(20), 10028–10035 (2016).
Federico Stilo is a PhD student at the University of Turin, in Turin, Italy. Federico’s project deals with Italian extra-virgin olive oil volatilome with particular emphasis on sensory active volatiles responsible for positive and negative attributes. The toolbox to achieve project objectives includes: comprehensive two-dimensional gas chromatography coupled to time-of-flight mass spectrometry, automated headspace sampling, and dedicated data processing tools for effective chemical fingerprinting and profiling.
Erica Liberto is Associate Professor of food chemistry at the University of Turin. Her research interests cover food volatilome and sensometabolome (coffee, cocoa, olive oil, and vegetal matrices) and their chemical characterization by gas chromatography coupled to mass spectrometry, MS-nose platforms, and headspace sampling by miniaturized approaches and devices. Data elaboration by different chemometric approaches is also a field of active research.
Carlo Bicchi is Full Professor of pharmaceutical biology at the University of Turin. He has dedicated most of his research to the development of innovative analytical approaches and techniques, mainly based on gas chromatography, for the characterization of the complex volatile fraction of plants and food of vegetal origin. Research interests also cover the development of new GC stationary phases, and innovative approaches for headspace sampling based on solvent-free and fully automatic solutions.
Stephen E. Reichenbach is a Professor of computer science and engineering at the University of Nebraska, in Lincoln, Nebraska, USA. His research develops chemometrics and cheminformatics methods and tools for multidimensional data. He also serves as Director for GC Image, LLC (Lincoln, Nebraska).
Chiara Cordero is Associate Professor of food chemistry at the University of Turin. Her research interests cover food volatilome and sensometabolome (hazelnuts, cocoa, olive oil, and high-quality food of vegetal origin) and their chemical characterization by multidimensional gas chromatographic platforms, headspace sampling by miniaturized approaches and devices, and GC×GC data processing by advanced fingerprinting algorithms.

Analytical Challenges in Measuring Migration from Food Contact Materials
November 2nd 2015Food contact materials contain low molecular weight additives and processing aids which can migrate into foods leading to trace levels of contamination. Food safety is ensured through regulations, comprising compositional controls and migration limits, which present a significant analytical challenge to the food industry to ensure compliance and demonstrate due diligence. Of the various analytical approaches, LC-MS/MS has proved to be an essential tool in monitoring migration of target compounds into foods, and more sophisticated approaches such as LC-high resolution MS (Orbitrap) are being increasingly used for untargeted analysis to monitor non-intentionally added substances. This podcast will provide an overview to this area, illustrated with various applications showing current approaches being employed.
New Study Reviews Chromatography Methods for Flavonoid Analysis
April 21st 2025Flavonoids are widely used metabolites that carry out various functions in different industries, such as food and cosmetics. Detecting, separating, and quantifying them in fruit species can be a complicated process.
University of Rouen-Normandy Scientists Explore Eco-Friendly Sampling Approach for GC-HRMS
April 17th 2025Root exudates—substances secreted by living plant roots—are challenging to sample, as they are typically extracted using artificial devices and can vary widely in both quantity and composition across plant species.
Sorbonne Researchers Develop Miniaturized GC Detector for VOC Analysis
April 16th 2025A team of scientists from the Paris university developed and optimized MAVERIC, a miniaturized and autonomous gas chromatography (GC) system coupled to a nano-gravimetric detector (NGD) based on a NEMS (nano-electromechanical-system) resonator.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)









