Gas Chromatographic Techniques for the Analysis of Chemical Warfare Agents
LCGC Europe
The complexity of the systems that are used vary from standard GC configurations to assess the effectiveness of protective clothing to trace analysis of biomedical samples with complex GC configuration.
Chemical warfare agents (CWAs) were first used on a large scale during World War I. They can be divided into different classes: choking agents (phosgene, chlorine, nitrogen oxides); blister or vesicants (mustard gas, lewisite, phosgene oxime); blood agents (cyanogen chloride, hydrogen cyanide); nerve agents (sarin, soman, tabun, VX); and incapacitating agents (tear gas, pepper spray).
This article will primarily focus on the analysis of mustard gas and nerve agents. Mustard gas [bis (2-chloroethyl) sulphide] is known to cause blisters on the skin and has devastating effects on the lungs and the eyes. The working mechanism behind the blister formation is still unknown. Low-level exposure of mustard gas also poses a risk because the compound has alkylating properties and can react with proteins and deoxyribonucleic acid (DNA), which could possibly induce cancer.
The toxicity of the nerve agents has been studied intensively. Organophosphates (OPs) inhibit the essential enzyme acetylcholinesterase (AChE), which leads to an accumulation of acetylcholine, results in neurological malfunctioning and can be lethal within a few minutes.
The use, storage and production of chemical weapons is prohibited by the chemical weapons convention (CWC) and maintained by the organization for the prohibition of chemical weapons (OPCW). However, several countries have not signed this treaty and terrorist groups that gain access to information on how to produce chemical weapons are a major threat.
Research on the defence against chemical warfare agents is very important and involves the development of protective clothing and gas masks, the detection of CWAs in environmental samples, the verification of exposure to individuals by analysing biomedical samples, and the development of medical countermeasures against CWAs.
GC is a tool that is used for the quantitative analysis of CWAs in these different research areas. The complexity of the systems that are used vary from standard GC configurations to assess the effectiveness of protective clothing to trace analysis of biomedical samples with complex GC configuration.
This article will focus on the various configurations of the instrumentation that is used to analyse CWAs. Sample preparation is also very important but is beyond the scope of this article.
Environmental Analysis
Environmental samples can be roughly categorized as air, water or soil samples. One of the tasks of the OPCW is to perform inspections to assess whether the state parties comply with the treaty. The inspectors are allowed to take environmental samples on their inspections or when investigating an alleged incident of use. For the analysis of environmental samples, it is necessary to be able to detect low concentrations of the CWA, which affects the choice of chromatography system used.
A good water analysis example was presented by Degenhardt et al.1 They described a large volume (200 μL) on-column injection technique to analyse a mixture of nerve agents: sarin, diisopropyl fluorophosphate (DFP), soman, tabun and VX. This mixture was extracted from a spiked water sample by using an Amberlite XAD–4 column (Sigma-Aldrich, St Louis, Missouri, USA) followed by extraction with a mixture of n-pentane/methanol (95:5 v/v). A thermal ionic detector (TID) was used to detect the typical phosphorus signal of the organophosphates. The nerve agents could be detected at the parts per trillion (ppt) level starting with a 10 mL water sample. Figure 1 shows a typical chromatogram.

Figure 1
Analysing nerve agents in air samples is mostly performed using a solid adsorbent, such as Tenax–TA (Alltech, Deerfield, Illinois, USA), followed by thermal desorption. Volatile CWAs can be detected at ppt levels. Air samples containing non-volatile compounds such as VX or lewisite are more problematic. Thermal desorption is more difficult here because of thermal degradation. Another disadvantage is that off-line analysis is not easily automated, which complicates continuous monitoring of the environment. This means that the addition of sampling time, time to transport the sampling tube to the analytical equipment and the analysis time are quite large, which prevents continuous monitoring of the environment.
In a controlled experimental set-up it is possible to monitor the vapour concentration in air on a semi-continuous basis using GC. Vapour generation chambers are used to test detection devices and exposure experiments. The main challenge in these configurations is to create and monitor vapour concentrations at low levels (i.e., 0.1 μg/m3). A combination of a fixed volume injection loop and a short column allows a high cycle rate for the vapour analysis. A normal length chromatography column is connected with a Y-press fit to the vapour analysis column and are both directed to the same detector. Calibration of the vapour analysis is possible by analysis of standard solutions of the agents that are detected on the same detector as used for the vapour analysis.2
Because of the high degree of automation, the reproducibility of the analysis of the generated vapour was reasonable. The deviation at low concentrations (2.4 mg/m3 for soman vapour) was less than 4%. For the higher concentrations (208 mg/m3 for mustard gas) it was less than 1%. The detection limit (DL) for this configuration was approximately 0.5–1.0 mg/m3, depending on the agent and detector type.
For the analysis of very low levels of sarin vapour a cryogenic-fused silica capillary-cooled trap was used to focus the low amounts of sarin that were detected on the nitrogen phosphorous (NP) detector after a cycle time of 4 minutes. An example of the vapour analysis at the low ppt level is shown in Figure 2.3

Figure 2
Bioanalysis
Analysis of biomedical samples is performed to verify exposure to CWAs in instances of an alleged use or to measure the agent in toxicokinetic studies, in which the distribution and elimination of the agent is studied. In view of the high toxicity of CWAs the concentration of these agents in biomedical matrices, such as blood or urine, at toxicologically relevant levels is very low, which places high requirements on the analytical systems used.
The high reactivity of the agents means that the intact agent cannot be measured in the biological matrix, when the samples are taken hours or days after exposure. In most instances suitable biomarkers for exposure are metabolites or adducts to proteins (i.e., conjugate of the CWA to a protein such as albumin or butyrylcholinesterase).4
To be measured by GC, derivatization of the metabolites is necessary. Eventually these metabolites can be measured with standard gas chromatography–mass spectrometry (GC–MS) instruments. The advent of mass spectrometry–mass spectrometry (MS–MS) technology using triple-quad or ion-trap configurations further increased the selectivity and decreased limits of detection of pentafluorobenzyl-derivatized nerve agent metabolites.5
Adducts require a cleavage reaction from the protein (in some instances followed by derivatization) before they can be analysed by GC. A typical example of adduct analysis is the N-terminal valine adduct of mustard gas to haemoglobin, which can then be analysed by a standard gas chromatography–negative-ion chemical ionization–mass spectrometry (GC–NICI–MS) configuration.
Another example is the fluoride reactivation technique of protein-bound nerve agents. The nerve agent residue is regenerated as a phosphonofluoridate upon incubation with an excess of fluoride ions from proteins, mostly butyrylcholinesterase (BuChE). The phosphonofluoridate can be analysed sensitively with GC–MS with positive chemical ionization using ammonia as the reaction gas. The soft ionization mode allows the sensitive detection of the organophosphofluoridates while maintaining sufficient selectivity. Levels down to 5 pg/mL can be detected when used with the thermodesorption technique, which correspond with BuChE inhibition levels of 0.1%. This degree of inhibition can never be determined with a colourimetric enzyme-activity assay.
Toxicokinetic studies provide the quantitative basis for the development of medical countermeasures. In toxicokinetic studies the intact agent is measured in blood to study distribution and persistence of the toxicant. Unlike the analysis of nerve agents, which is possible with GC–MS and relatively soft chemical ionization, the analysis of intact mustard gas in blood required a stronger ionization technique such as electron impact ionization (EII).
Consequently, the influence of the matrix also became more apparent, which means that the selectivity of the chromatographic separation became more important. The use of two-dimensional (2D) chromatography, using the heart-cutting method, makes this analysis possible (Figure 3). Glass sample tubes containing Tenax TA (Alltech) were loaded with a volume of the extract (in most instances ethyl acetate was used as the extraction solvent) followed by purging with nitrogen gas at ambient temperature. The samples were thermally desorbed using an Automatic Thermodesorption System (Gerstel, Mulheim an der Ruhr, Germany) and cryofocused onto a piece of GC column in a cooled injection system (CIS) (Gerstel) After the desorption process, the CIS was flash heated and the compounds were injected onto the first column. A small fraction of the eluate of the first column was selected in a second cold trap and cryofocused before it was injected onto a second column. Column switching was controlled by a multiple switch-able intelligent controller (MuSIC) system (Varian, formerly Chrompack, Middleburg, The Netherlands). Finally, the compounds of interest were detected on a mass spectrometer (Agilent MSD-5973, Agilent Technologies, Palo Alto, California, USA) running under electrospray ionization (EI) conditions. A flame photographic detector (FPD) was used to screen the separation on the first column and to focus on the window of interest. With this configuration a detection limit of 10 pg/mL of mustard gas in blood or tissue samples was achieved.6
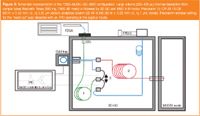
Figure 3
Nerve agents are chiral compounds and the stereoisomers are highly different in toxicity, which can be deduced from their binding constants with acetylcholinesterase and the rate of hydrolysis, which are both stereospecific.7 In toxicokinetic studies of nerve agents it is essential that the stereoisomers can be distinguished from each other because one is only interested in the toxicokinetics of the most toxic compound.
Nerve agents were extracted from the blood samples into ethyl acetate by means of solid-phase extraction (SPE) and analysed with a gas chromatography-nitrogen phosphorous detector (GC–NPD) using thermodesorption and 2D chromatography as described before for the mustard gas analysis. In this instance the second analytical column was replaced by a column with a stereoselective stationary phase.
The isomers of sarin could be separated on a Cyclodex column (Varian, formerly Chrompack) while the four isomers of soman were separated on a ChiraSil-Val-L column (synthesized at TNO-PML, Rijswijk, The Netherlands, column coating supplied by Varian, formerly Chrompack).8–10 The advantages of 2D chromatography are two-fold. Firstly, the 2D separation provides additional selectivity which is necessary because of the complicated sample matrix. Secondly, the fragile chiral stationary phase is protected from solvent and interfering components from the sample matrix. Nerve agent levels down to 10 pg/mL could be determined using the sensitive NPD. Figure 4 shows a typical separation of the four isomers of soman and a purified stereoisomer of deuterated soman (d13-soman) as the internal standard (IS).
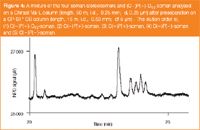
Figure 4
New Technologies
With the introduction of comprehensive gas chromatography (GC×GC) for the analysis of multi-component mixtures the possibilities for analysing CWAs have grown tremendously because of the increased selectivity. One of the biggest challenges in forensic science is finding "the needle in the haystack''. Despite the use of selective detectors, such as the NPD or an MS, it is very difficult to distinguish small traces of CWAs in complex matrices such as fuel samples. CWAs are relatively polar compounds while fuel consists of relatively non-polar compounds.
The multitude of components in fuel in a one dimensional (1D) separation would result in a continuous pattern of peaks. In a GC×GC configuration, the polar CWAs can be separated from the matrix on the second column because the non-polar components in fuels have not retained so much on the polar column. Figure 5 shows an example of the analysis of sarin and soman in gasoline.11 For this analysis a 15 m DB-1 (J&W, Folsom, California, USA) column with an internal diameter of 0.25 mm and a film thickness of 1 μm was connected to a 1 m DB–Wax (J&W) with an internal diameter of 0.1 mm and a film thickness of 0.1 μm. A Zoex loop-type modulator (Zoex Corp., Houston, Texas, USA) was installed in an Agilent GC (Agilent Technologies) and used for the modulation. Data acquisition was performed at a rate of 200 Hz; further data processing was performed with an early version of GC-Image software (Zoex Corp.).

Figure 5
Conclusion
GC is excellently suited to the analysis of CWAs. Many applications can be performed with standard GC configurations but some research topics require a more sophisticated configuration. The on-line monitoring of vapour concentration at low levels was only possible using on-line cryotrapping of the sample flow followed by detection with GC–NPD.
Trace analysis of nerve agents in biomedical matrices, such as plasma, was only possible after a large-volume sample introduction followed by detection with GC–MS using positive chemical ionization (PCI) with ammonia as the reaction gas. For toxicokinetic studies of nerve agents it is also essential that the stereoisomers of the OPs can be distinguished from each other; a requirement that could only be fulfilled by using 2D chromatography. It is anticipated that with the advent of more sophisticated detection methods, such as the multiple MS-modes used in triple quadrupole or ion trap instruments, the detection limits will decrease even further. Finally, the development of GC×GC chromatography allows the detection of CWAs in complex sample matrices such as gasoline.
Henk C. Trap is a chromatography expert at TNO Defence Safety and Security in Rijswijk, The Netherlands. He obtained his BSc in 1986 and has worked for more than 23 years with a wide range of chromatographic techniques for which he has developed several configurations over the years. His research interests include multidimensional separations, exposure techniques and toxicology. Marcel van der Schans is an analytical chemist at TNO Defence Safety and Security in Rijswijk, The Netherlands. He obtained his MSc (1992) and PhD (1997) in analytical chemistry from the Eindhoven University of Technology, The Netherlands. His research interests include toxicokinetics and diagnosis of exposure to chemical warfare agents. Column Editor, Robert Smits, is the president of the Koninklijke Vlaamse Chemische Vereniging (Royal Flemish Chemical Society). He is Chairman of the biennial "International Symposium on Hyphenated Techniques in Chromatography and Hyphenated Chromatographic Analysers" and a member of the Editorial Advisory Board of LCGC Europe.
References
1. C.E.A.M. Degenhardt-Langelaan and Ch.E. Kientz, J. Chrom. A., 723, 210–214 (1996).
2. H.C. Trap and J.P. Langenberg, J. High Resol. Chromatogr., 22, 153–158 (1999).
3. H.P.M. Van Helden et al., Toxicol. Appl. Pharmacol., 189, 170–179 (2003).
4. D. Noort, H.P. Benschop and R.M. Black, Toxicol. Appl. Pharmacol., 184, 116–126 (2002).
5. J. Riches et al., J. Chromatogr. B., 816, 251–258 (2005).
6. J.P. Oostdijk et al., J. Chrom. A., still in press.
7. H.P. Benschop et al., Toxicol. Appl. Pharmacol., 90, 490–500 (1987).
8. H.P. Benschop. and L.P.A De Jong, Neurosci. Behav. Rev., 15, 73–77 (1991).
9. W.E.T. Spruitet al., Toxicol. Appl. Pharmacol., 169, 249–254 (2000).
10. W.E.T Spruit et al., J. Anal. Toxicol., 25, 57–61 (2001).
11. S.E. Reichenbach et al., Chem. and Biol. Sensing, Proceeding of SPIE, 5085, 28–36 (2003).

Silvia Radenkovic on Building Connections in the Scientific Community
April 11th 2025In the second part of our conversation with Silvia Radenkovic, she shares insights into her involvement in scientific organizations and offers advice for young scientists looking to engage more in scientific organizations.
Regulatory Deadlines and Supply Chain Challenges Take Center Stage in Nitrosamine Discussion
April 10th 2025During an LCGC International peer exchange, Aloka Srinivasan, Mayank Bhanti, and Amber Burch discussed the regulatory deadlines and supply chain challenges that come with nitrosamine analysis.
Removing Double-Stranded RNA Impurities Using Chromatography
April 8th 2025Researchers from Agency for Science, Technology and Research in Singapore recently published a review article exploring how chromatography can be used to remove double-stranded RNA impurities during mRNA therapeutics production.