Developments in Green Chromatography
LCGC Europe
Why should you be concerned about mobile-phase degassing - it's all done automatically, isn't it?
Green analytical chemistry is a widely recognized concept that has led to the development of new analytical methods with reduced environmental impact and minimized analyst occupational exposure. Achievements include the development of microextraction, ultrasound-assisted extraction (UAE), and microwave-assisted extraction (MAE) techniques. Research towards greener separation processes focuses on the elimination of toxic solvents in liquid chromatography (LC) and the reduction of separation time. The recent developments in the labelling and categorization of analytical procedures are also presented. The National Environmental Methods Index (NEMI), Eco-Scale, and grouping with multivariate statistics are discussed together with their advantages and limitations. This article presents the most recent progress in the development of greener sample preparation and chromatographic separation techniques.
Green analytical chemistry is now a widely recognized term and is defined in the literature as clean, benign, or environmentally friendly analytical chemistry (1). Green analytical chemistry means performing reliable analysis while applying quality assurance/quality control (QA/QC) and minimizing any environmental impacts resulting from the analysis. When one thinks of chromatographic procedures that meet the standards of green analytical chemistry principles, green chromatography comes to mind first (2). Chromatographic procedures are usually a series of operations that involve sample collection, transportation, preparation, proper chromatographic separation, and analysis. Greening chromatography concerns all stages of analysis, but the steps with the greatest environmental impact are sample preparation and the chromatographic separation. These stages are considered to be the most environmentally unfriendly and therefore have a lot of potential for "greening" (3).

(PHOTO CREDIT: COMSTOCK/GETTY IMAGES)
Green chromatography originates from the 12 green chemistry principles developed to minimize the environmental impact of chemical synthesis. When compared to chemical synthesis methods, the amount of pollutant waste products generated during chromatographic analysis appears small. However, it has been estimated that one liquid chromatograph can generate one litre of liquid waste daily (2). Three years ago, a series of three articles were published in LCGC Europe addressing the different aspects of green chromatography. The first article provided an introduction to green chromatography, describing its origin and the potential of green liquid chromatography (LC) (4); the second article proved that gas chromatography (GC) is often beneficial compared to LC (5); and the third article reviewed the importance of sample preparation before chromatographic analysis and how green chromatography principles could be applied (6). This article summarizes the achievements and developments over the last three years that have advanced the field of green chromatography.

Sample Preparation
In a chromatographic analysis, sample preparation has the most detrimental effect on the environment and the question of whether or not it is really needed should always be asked. Unfortunately in most cases the answer is "yes" but there are chromatographic procedures that can omit this procedural step, as reviewed in reference 7. Direct procedures can be used to analyze samples with simple matrices such as water, petroleum products, and alcoholic beverages. Such approaches reduce environmental impact as well as saving money.
Figure 1 presents the options available to implement green sample preparation including miniaturization (8), solvent elimination (9), and application of extraction assisting or enhancing agents (such as ultrasounds or microwaves) (10). It can be difficult to automate microextraction techniques; however, advances have been made including the development of dispersive liquid–liquid microextraction (DLLME); liquid-phase microextraction (LPME), and single drop microextraction (SDME) (11).
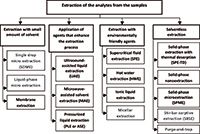
Figure 1: Solutions in sample preparation that meet green chromatography requirements.
In recent years, a lot of work has focused on new solutions and applications that improve the "greenness" of sample preparation techniques. One technique that has been intensively worked on is DLLME. DLLME utilizes a tiny amount of nonpolar solvent (usually below 100 µL) and approximately 1 mL of less toxic dispersive solvent. Procedures based on DLLME have been proposed to determine UV filters (12), antibiotics (13), polycyclic aromatic hydrocarbons (PAHs) (14), or oestrogens (15) in water samples; and pesticides in honey, after sample dissolution in water (16).
Performing DLLME extraction can be assisted with ultrasounds that eliminate or reduce the need for disperser solvent. The application of dispersive solvent increases the solubility of the extraction solvent. Without the dispersive solvent, extraction solvent volume can be reduced, because less of it is dissolved in water; and despite low initial solvent volume it is easy to recover enough extraction solvent volume to handle.
For example, ultrasound-assisted extraction with 13 µL of chlorobenzene can be applied to determine siloxanes in water samples (17). In LC analysis, ionic liquids are considered as green solvents and can be applied as extraction agents in DLLME.
An alternative solution to DLLME is solidification of floating organic drop (SFO). The extraction solvents used in SFO are higher alcohols such as nonanol, decanol, or undecanol. After extraction, the sample is subjected to a lower temperature and the solvent solidifies and can be easily mechanically separated from the sample matrix. The technique can be used in sample preparation before GC and LC analysis. It is suitable for the extraction of compounds with log kow in the range 3–7 (18). For example: Triazines in water samples can be determined using 10 µL of 1-undecanol and 100 µL of acetonitrile as dispersive solvent (19). A similar procedure was designed for the determination of amphetamines in urine samples (20), and for the determination of pesticides in vegetables and fruits after extraction with acetone (21). In 2013, Dong et al. applied DLLME to the analysis of soil samples (22). The authors extracted phytocides from soil with a water–methanol mixture and evaporated to dryness before the residue was redissolved in methanol. The samples were subjected to ionic liquid-based ultrasound-assisted DLLME before high performance liquid chromatography (HPLC) analysis.
A derivatization step can be easily combined with DLLME. It is particularly practical when the derivatizing agent easily hydrolyzes in the water sample (23). In 2014, Casado et al. performed acetylation with simultaneous extraction of benzotriazoles on a mixture of microvolumes of acetic anhydride, acetonitrile, and toluene (24). In 2013, Farajzadeh et al. applied butylchloroformate as a derivatizing agent in DLLME for the determination of amantadine, a pharmaceutical prescribed for influenza treatment, in human plasma samples (25).
The main trends in solid-phase microextraction (SPME) are automation, in situ sampling, development of in vivo applications, and the development of new coatings (non-bonded, bonded, or cross-linked polymers) (26). Polydimethylsiloxane (PDMS) has traditionally been used as a SPME fibre coating, but fibres based on polyacrylate or polyglycol polymers are now available for the extraction of polar analytes. The support for coatings has traditionally been fused-silica, but recently metal-based fibres have been developed for increased durability and improved mechanical properties. Increasing the coating film thickness to increase fibre capacity and therefore sensitivity is another trend; however, it can increase the extraction time required to obtain equilibration (27).
Ionic liquids have recently been demonstrated as fibre coatings, opening up the possibility to design and modify coatings to obtain greater selectivity towards analytes (28). In 2012, Feng et al. applied a polymerized ionic liquid 1-vinyl-3-octylimidazolium bromide as a coating in SPME to isolate alcohols from water (even methanol) and produced good recoveries (29). In 2012, Ho et al. extracted chlorophenols from water with 1-butyl-3-methylimidazolium hexafluorophosphate-coated SPME fibre (30).
Novel applications of SPME include the determination of contaminants or bioactive compounds directly in tissues of living organisms, or in their habitat, as demonstrated by Bessonneau et al. in the case of corals (31). In vivo SPME sampling has also been developed to determine emerging contaminants in fish (32), and volatile halogenated organic compounds in trees (33), insects, higher animals, and even humans (34).
Stir-bar sorbent extraction (SBSE) is another rapidly developing technique, where the majority of applications use PDMS coatings. Recently, alternative polar coatings have been introduced and intensively investigated for the extraction of analytes of a wide range of polarities including polyacrylate and polyglycol coatings (35). For the extraction of chlorophenols and chloroanizoles from wine samples, ethylene glycol with silicone in the form of a copolymer was found to be more effective than a polyacrylate coating (36). This is because it allows for simultaneous extraction and preconcentration of both polar phenols and nonpolar chloroanisoles. Another coating for the extraction of polar compounds from water samples is methacrylic acid and divinyl benzene copolymer (37). This SBSE coating gives much better recoveries (around 100% for the most of investigated compounds) for polar pharmaceuticals than commercial PDMS coating-based stir bar (less than 20%).
The stir bar has also been applied as a passive sampler for the in situ extraction of nitroorganic pesticides (38). Such an approach seems to be a promising technique for the collection of integrated samples. The in situ exposition and sample preparation steps are both straightforward. Selection of the most appropriate stir bar coating can result in sampling of polar analytes, which are usually not collected with more traditional passive samplers.
Stir bars can also be coated with molecularly imprinted polymers (MIPs) (39) or chiral imprinted polymers (40) for extraction of certain groups of analytes. The time needed to perform analysis is relatively long; however, the limit of detection (LOD) obtained are very low. For example, three hours of SBSE extraction followed by thermal desorption, two-dimensional GC separation and time-of-flight mass spectrometry (TOF-MS) allowed for the determination organochlorine pesticides at pg/L concentrations (41).
Separation Process
It is widely accepted that LC is less "green" than GC because LC mobile phases are usually solvents (unless water is used). One of the most commonly used toxic solvents in LC is acetonitrile. This solvent can be replaced with a propylene carbonate–ethanol–water mixture as demonstrated in the determination of bioactive compounds in human plasma (42). The "acetonitrile-less" procedure was characterized by good performance, similar to when using acetonitrile. Another green mobile phase that can be applied in LC is a ethyl acetate–ethanol mixture (43). One of the most common approaches to designing an appropriate mobile phase in reversed-phase high performance liquid chromatography (RP–HPLC) is to add small volumes of modifiers to water. Using a solution of 0.04% triethylamine in water as a mobile phase can give good separation of analytes of various polarities on a polyethylene glycol stationary phase (44). Another interesting solution to using toxic solvents is to perform high temperature liquid chromatography (HTLC). Water between 60 °C and 374 °C is used as the mobile phase, because at this temperature water has a lower viscosity and polarity, thereby shortening analysis time. For example, Terol et al. applied HTLC to determine arsenosugars in marine organisms (45).
An alternative option to remove the solvent factor is to perform capillary LC. Capillary LC, compared to packed-column LC, is characterized by low mobile phase flow rate and low consumption. Capillary LC methodology was developed for the determination of vitamin E in cosmetics by Plotka et al. and demonstrated by low mobile phase consumption, approximately 0.4 mL for each analysis (46).
Another way to apply a greener mobile phase is to perform supercritical fluid chromatography (SFC). High efficiency, safety, and short analysis time are the main advantages of SFC (47). SFC can replace LC for the analysis of polar drugs, using CO2 modified with methanol to increase polarity (48). Similarly, CO2 in the supercritical state can be modified with methanol and used as a mobile phase for separation of carotenoids (49).
Assessment of Procedures
One of the main problems of green analytical chemistry is the lack of procedure assessment systems. There needs to be a convenient method for delivering information regarding environmental impact, together with some quantitative information about the impact and a comparison with other procedures to aid an analyst in their decision. The first labelling system was based on National Environmental Methods Index (NEMI) symbols (50). The circle symbol has four quarters, each representing one method aspect that could have a possible environmental impact. Methodology that meets the standards of green analytical chemistry has all four fields filled green. If the procedure does not meet one of the requirements the respective quarter of the symbol is not coloured. The procedure does not meet the "greenness" requirements when: (i) Any of the chemicals used in the procedure is listed as persistent, bioaccumulative, or toxic; (ii) any of the chemicals used in the procedure is listed on the TRI (toxic release inventory) list or on any of the RCRA's (Resource Conservation and Recovery Act) lists as hazardous; (iii) the pH during analysis is below 2 or above 12; and (iv) the amount of waste generated is above 50 g. The main advantages of the NEMI symbol assessment are the simplicity of the assessment and reading the score. The main drawback is that it does not consider the amount of chemicals used in the analysis, and the waste generated is analyzed rather non-quantitatively. In addition, searching official lists for chemicals used during the procedure is inconvenient.
In recent years, other approaches for "greenness" assessment have been proposed, such as the Analytical Eco-Scale (51). Assessment is based on the calculation of a numerical score, where 100 is the ideal green procedure. From the basic 100 points, penalty points are subtracted for each chemical used in the procedure that are hazardous, generate waste, risk occupational exposure, or have high energy consumption. A score above 75 points indicates that the procedure is green, a score between 50 and 75 indicates that it is acceptably green, and a score below 50 indicates nongreen analysis. The amount of chemicals and waste are taken into consideration semi-quantitavely and the degree of each chemical hazard is proportional to penalty points. The main advantage of the analytical Eco-Scale method is semi-quantitative consideration of various methodological parameters, while at the same time being simple with well-defined evaluation criteria.
The environmental impact of procedures can be compared using multivariate statistical techniques. The self-organizing maps procedure allows comparison of procedures in a multivariate manner to find statistical differences and similarities that allow for grouping of analytical procedures according to their "greenness" (52). It is easy to find discriminators between groups. Discriminators are factors that create differences between similar methods. In the majority of cases, discriminators are usually the environmental impact variables of an analysis. This means that the main differences between procedures lie in their "greenness," while the metrological parameters are similar. This evaluation procedure application is restricted to the created dataset and it is impossible to add a new procedure after the results have been generated. The only option is to add to the dataset the new procedure and repeat the analysis. Therefore, multivariate statistics is not suitable for routine assessment of procedures because the evaluation procedure is very time-consuming.
Another tool dedicated to the assessment of HPLC methodology "greenness" is the Environmental Assessment Tool (EAT) (53). A score is calculated based on the sum of safety, health, and environmental factors for a given amount of solvent applied during LC analysis: the lower the EAT value, the greener the procedure. EAT assessment focuses on solvent consumption, which seems to be the main problem in chromatographic analysis. EAT analysis is simple with a downloadable computer program.
The best approach to measure the environmental impact of an analytical procedure, although based on some assumptions, would be Life Cycle Assessment (LCA). This approach takes into consideration environmental impact "from cradle to grave". It covers the stages of production, disposal, and transportation of chemicals. Until now, analytical procedures have not been subjected to LCA analysis (54).
Conclusions
Significant developments have been observed to make chromatography more environmentally friendly. There has been a shift towards miniaturization and organic solvents have been substituted with water and other less toxic alternatives. What should be highlighted is that there are concerted efforts to qualitatively measure and compare the chromatographic procedures in terms of possible harm to the environment. Although this is a recent development, there are still a lot of issues, such as the amount and toxicity of solvents and reagents, to be improved upon to make the chromatography "greener".
Marek Tobiszewski obtained his PhD degree in 2012, and then began his research in the Department of Analytical Chemistry in Gdansk University of Technology. His research interests include environmental analytical chemistry, sample preparation before GC analysis, and the environmental fate of organic pollutants, in particular chlorinated solvents and PAHs. He is also interested in green analytical chemistry and the application of multivariate statistics in environmental monitoring.
Jacek Namieśnik obtained his PhD in 1978 and has been a professor since 1996. He was Dean of the Chemical Faculty at the Gdansk University of Technology from 1996 to 2002 and since 2005 he has been Head of the Department of Analytical Chemistry. He has also been Chairman of the Committee on Analytical Chemistry, Polish Academy of Sciences (PAS) since 2005. His major research interests include the development of new analytical procedures for determining trace and ultratrace constituents in samples with complex matrix compositions, and the design and testing of customized analytical units and measuring devices. He is the author of over 300 papers and 10 patents.
References
(1) S. Armenta, S. Garrigues, and M. de la Guardia, Trends Anal. Chem. 27(6), 497–511 (2008).
(2) C.J. Welch, N. Wu, M. Biba, R. Hartman, T. Brkovic, X. Gong, R. Helmy, W. Schafer, J. Cuff, Z. Pirzada, and L. Zhou, Trends Anal. Chem. 29(7), 667–680 (2010).
(3) J. Płotka, M. Tobiszewski, A.M. Sulej, M. Kupska, T. Górecki, and J. Namieśnik, J. Chromatogr. A 1307, 1–20 (2013).
(4) P. Sandra, G. Vanhoenacker, F. David, K. Sandra, and A. Pereira, LCGC Europe 23(5), 242–259 (2011).
(5) P. Sandra, A. Pereira, F. David, M. Dunkle, and C. Brunelli, LCGC Europe 23(8), 396–405 (2011).
(6) P. Sandra, B. Tienpont, and F. David, LCGC Europe 24(3), 120–133 (2012).
(7) M. Tobiszewski and J. Namieśnik, Trends Anal. Chem . 35, 67–73 (2012).
(8) F. Pena-Pereira, I. Lavilla, and C. Bendicho, Trends Anal. Chem. 29(7), 617–628 (2010).
(9) W. Wardencki, J. Curyło, and J. Namieśnik, J. Biochem. Biophys. Methods 70(2), 275–288 (2007).
(10) Y. Li, A.S. Fabiano-Tixier, M. Abert Vian, and F. Chemat, Trends Anal. Chem. 47, 1–11 (2013).
(11) L. Kocúrová, I.S. Balogh, and V. Andruch, Microchem. J . 110, 599–607 (2013).
(12) J.L. Benedé, A. Chisvert, A. Salvador, D. Sánchez-Quiles, and A. Tovar-Sánchez, Anal. Chim. Acta 812, 50–58 (2014).
(13) A.V. Herrera-Herrera, J. Hernández-Borges, T.M. Borges-Miquel, and M.Á. Rodríguez-Delgado, J. Pharm. Biomed. Anal . 75, 130–137 (2013).
(14) W.-C. Tseng, P.-S. Chen, and S.-D. Huang, Talanta 120, 425–432 (2014).
(15) D.L.D. Lima, C.P. Silva, M. Otero, and V.I. Esteves, Talanta 115, 980–985 (2013).
(16) M.W. Kujawski, E. Pinteaux, and J. Namieśnik, Eur. Food Res. Technol. 234, 223–230 (2012).
(17) C. Cortada, L. Costados Reis, L. Vidal, J. Llorca, and A. Canals, Talanta 120, 191–197 (2014).
(18) L.E. Vera-Avila, T. Rojo-Portillo, R. Covarrubias-Herrera, and A. Pena-Alvarez, Anal. Chim. Acta 805, 60–69 (2013).
(19) M.M. Sanagi, H.H. Abbas, W.A. Wan Ibrahim, and H.Y. Aboul-Enien, Food Chem. 133, 557–562 (2012).
(20) T. Ahmadi-Jouibari, N. Fattahi, and M. Shamsipur, J. Pharm. Biomed. Anal. 94, 145–151 (2014).
(21) M. Pirsaheb, N. Fattahi, and M. Shamsipur, Food Control 34, 378–385 (2013).
(22) S. Dong, Q. Hu, Z. Yang, R. Liu, G. Huang, and T. Huang, Microchem. J. 110, 221–226 (2013).
(23) M. Shamsipur, M. Taghi Naseri, and M. Babri, J. Pharm. Biomed. Anal. 81–82, 65–75 (2013).
(24) J. Casado, R. Nescatelli, I. Rodríguez, M. Ramil, F. Marini, and R. Cela, J. Chromatogr. A 1336, 1–9 (2014).
(25) M.A. Farajzadeh, N. Nouri, and A.A. Alizadeh Nabil, J. Chromatogr. B 940, 142–149 (2013).
(26) H. Kataoka and K. Saito, J. Pharm. Biomed. Anal . 54, 926–950 (2011).
(27) E.A. Souza Silva, S. Risticevic, and J. Pawliszyn, Trends Anal. Chem. 43, 24–36 (2013).
(28) H. Yu, T.D. Ho, and J.L. Anderson, Trends Anal. Chem. 45, 219–232 (2013).
(29) J. Feng, M. Sun, X. Wang, X. Liu, and S. Jiang, J. Chromatogr. A 1245, 32–38 (2012).
(30) T.-T. Ho, C.-Y. Chen, Z.-G. Li, T.C.-C. Yang, and M.-R. Lee, Anal. Chim. Acta, 712, 72–77 (2012).
(31) V. Bessonneau, B. Bojko, and J. Pawliszyn, J. Chromatogr. A 1328, 113–117 (2014).
(32) S. Wang, K.D. Oakes, L.M. Bragg, J. Pawliszyn, G. Dixon, and M.R. Servos, Chemosphere 85, 1472–1480 (2011).
(33) E.M. Sheehan, M.A.Limmer, P. Mayer, U.G. Karlson, and J.G. Bursken, Environ. Sci. Technol. 46, 3319–3325 (2012).
(34) G. Ouyang, D. Vuckovic, and J. Pawliszyn, Chem. Rev. 111, 2784–2814 (2011).
(35) N. Gilart, R.M. Marcé, F. Borrull, and N. Fontanals, Trends Anal. Chem. 54, 11–23 (2014).
(36) J.I. Cacho, N. Campillo, P. Viñas, and M. Hernández-Córdoba, Talanta 118, 30–36 (2014).
(37) D. Bratkowska, N. Fontanals, P.A.G. Cormack, F. Borrull, and R.M. Marce, J. Chromatogr. A 1225, 1–7 (2012).
(38) A. Assoumani, S. Lissalde, C. Margoum, N. Mazzella, and M. Coquery, Sci. Total Environ. 463–464, 829–835 (2013).
(39) A. Martin-Esteban, Trends Anal. Chem . 45, 169–181 (2013).
(40) A. Gomez-Caballero, A. Guerreiro, K. Karim, S. Piletsky, M. Aranzazu Goicolea, and R.J. Barrio, Biosensors Bioelectr. 28, 25– 32 (2011).
(41) N. Ochiai, T. Ieda, K. Sasamoto, Y. Takazawa, S. Hashimoto, A. Fushimi, and K. Tanabe, J. Chromatogr. A 1218, 6851–6860 (2011).
(42) M. Cheregi, F. Albua, S. Udrescu, N. Raducanu, and A. Medvedovici, J. Chromatogr. B 927, 124–132 (2013).
(43) N. Haq, M. Iqbal, F.K. Alanazi, I.A. Alsarra, and F. Shakeel, Arab. J. Chem. DOI: 10.1016/j.arabjc.2012.12.004 (2012).
(44) D. Šatínský, I Brabcová, A. Maroušková, P. Chocholouš, and P. Solich, Anal. Bioanal. Chem. 405, 6105–6115 (2013).
(45) A. Terol, F. Ardini, M. Grotti, and J.L. Todolí, J. Chromatogr. A 1262, 70–76 (2012).
(46) P. Viñas, M. Pastor-Belda, N. Campillo, M. Bravo-Bravo, and M. Hernández-Córdoba, J. Pharm. Biomed. Anal. DOI:10.1016/j.jpba.2014.02.001 (2014).
(47) J.M. Plotka, M. Biziuk, C. Morrison, and J. Namieśnik, Trends Anal. Chem. 56, 74–89 (2014).
(48) A. Dispas, P. Lebrun, P. Sassiat, E. Ziemons, D. Thiébaut, J. Vial, and P. Hubert, J. Chromatogr. A 1256, 253–260 (2012).
(49) V. Abrahamsson, I. Rodriguez-Meizoso, and C. Turner, J. Chromatogr. A 1250, 63–68 (2012).
(50) L.H. Keith, L.U. Gron, and J.L. Young, Chem. Rev. 107, 2695–2708 (2007).
(51) A. Galuszka, P. Konieczka, Z.M. Migaszewski, and J. Namieśnik, Trends Anal. Chem. 37, 61–72 (2012).
(52) M. Tobiszewski, S. Tsakovski, V. Simeonov, and J. Namieśnik, Green Chem. 15, 1615–1623 (2013).
(53) Y. Gaber, U. Törnvall, M.A. Kumar, M.A. Amin, and R. Hatti-Kaul, Green Chem. 13, 2021–2025 (2011).
(54) C. Turner, Pure Appl. Chem. 85, 2217–2229 (2013).

Thermodynamic Insights into Organic Solvent Extraction for Chemical Analysis of Medical Devices
April 16th 2025A new study, published by a researcher from Chemical Characterization Solutions in Minnesota, explored a new approach for sample preparation for the chemical characterization of medical devices.
Study Explores Thin-Film Extraction of Biogenic Amines via HPLC-MS/MS
March 27th 2025Scientists from Tabriz University and the University of Tabriz explored cellulose acetate-UiO-66-COOH as an affordable coating sorbent for thin film extraction of biogenic amines from cheese and alcohol-free beverages using HPLC-MS/MS.
Multi-Step Preparative LC–MS Workflow for Peptide Purification
March 21st 2025This article introduces a multi-step preparative purification workflow for synthetic peptides using liquid chromatography–mass spectrometry (LC–MS). The process involves optimizing separation conditions, scaling-up, fractionating, and confirming purity and recovery, using a single LC–MS system. High purity and recovery rates for synthetic peptides such as parathormone (PTH) are achieved. The method allows efficient purification and accurate confirmation of peptide synthesis and is suitable for handling complex preparative purification tasks.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)




