Ascentis Express 5 micron: A Fused-Core Particle HPLC Column for Faster HPLC Separations with No Backpressure Concerns
The Application Notebook
Ascentis Express 5 micron columns provide a new choice for improving the performance of traditional HPLC systems. Based on Fused-Core particle technology, Ascentis Express provides the benefits of high speed and high efficiencies without the concerns of smaller particle columns. Due to the high efficiencies at low backpressures, Ascentis Express 5 micron can benefit conventional HPLC users with no drawbacks.
Ascentis Express 5 micron columns provide a new choice for improving the performance of traditional HPLC systems. Based on Fused-Core particle technology, Ascentis Express provides the benefits of high speed and high efficiencies without the concerns of smaller particle columns. Due to the high efficiencies at low backpressures, Ascentis Express 5 micron can benefit conventional HPLC users with no drawbacks.
The new Fused-Core particle consists of a 3.3 μm solid core and a 0.6 μm porous shell. A major benefit of the Fused-Core particle is the small diffusion path (0.6 μm) compared to conventional fully porous particles. The short diffusion path reduces axial dispersion of solutes and minimizes peak broadening. In fact, Ascentis Express 5 micron columns are able to achieve greater speed and efficiency than any other 5 micron particle based column. This means that Ascentis Express 5 micron becomes the standard column for all of your 5 micron based methods.
Beyond the new standard column for all 5 micron based methods, Ascentis Express 5 micron is an excellent choice for bioanalytical LC–MS methods. The Ascentis Express 5 micron excels under high flow rates and the high throughput demands of these methods. Furthermore, the large particle format provides an extremely rugged HPLC column. Available in 17 dimensions from 20 mm × 2.1 mm i.d. to 250 mm × 4.6 mm i.d. and two introductory phases, C18 and F5, the conversion of current methods is seamless.

Table I: Summary of efficiency, backpressure, and speed for Figure 1
Experimental Conditions
Shown in Figure 1 are HPLC chromatograms of a mix of polar and non-polar analytes using both the new Ascentis Express 5 micron and a popular traditional 5 micron column. The chromatogram illustrates the high speed and high efficiency capabilities of the Ascentis Express at low back pressures. Table I provides a summary of the performance parameters of Figure 1.
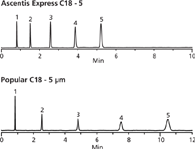
Figure 1: HPLC chromatogram of a mix of polar and non-polar analytes on Ascentis Express. Column: Ascentis Express C18, 15 cm à 4.6 mm i.d., 5 μm particles. Mobile Phase: 50:50 water:acetonitrile; temperature: 35 °C; flow rate: 1.50 mL/min; detection: UV, 250 nm; injection vol.: 5 μL. Peaks: 1. Uracil, 2. Acetophenone, 3. Benzene, 4. Toluene, and 5. Naphthalene.
Results and Discussion
Conventional HPLC users can obtain an immediate increase in efficiency with this "drop-in" replacement column. No need to change column dimensions, flow rates, mobile phase conditions, or sample prep. The Ascentis Express "5" can improve the performance of all of your HPLC systems.
Conclusion
Ascentis Express 5 micron with Fused-Core particles is an excellent choice for standardizing all 5 micron methods. The combination of low backpressures and high efficiencies allows for greater column length and flow rate flexibility than offered by any 5 micron column currently available.
Ascentis is a registered trademark of Sigma-Aldrich Co. LLC. Fused-Core is a registered trademark of Advanced Materials Technology, Inc.
Supelco/Sigma-Aldrich
595 North Harrison Road, Bellefonte, PA 16823
tel. (800) 359-3041, (814) 359-3041
Website: sigma-aldrich.com/analytical

Free Poster: NDSRI Risk Assessment and Trace-Level Analysis of N-Nitrosamines
April 25th 2025With increasing concern over genotoxic nitrosamine contaminants, regulatory bodies like the FDA and EMA have introduced strict guidelines following several high-profile drug recalls. This poster showcases a case study where LGC and Waters developed a UPLC/MS/MS method for quantifying trace levels of N-nitroso-sertraline in sertraline using Waters mass spectrometry and LGC reference standards.
New TRC Facility Accelerates Innovation and Delivery
April 25th 2025We’ve expanded our capabilities with a state-of-the-art, 200,000 sq ft TRC facility in Toronto, completed in 2024 and staffed by over 100 PhD- and MSc-level scientists. This investment enables the development of more innovative compounds, a broader catalogue and custom offering, and streamlined operations for faster delivery. • Our extensive range of over 100,000 high-quality research chemicals—including APIs, metabolites, and impurities in both native and stable isotope-labelled forms—provides essential tools for uncovering molecular disease mechanisms and exploring new opportunities for therapeutic intervention.
New Guide: Characterising Impurity Standards – What Defines “Good Enough?”
April 25th 2025Impurity reference standards (IRSs) are essential for accurately identifying and quantifying impurities in pharmaceutical development and manufacturing. Yet, with limited regulatory guidance on how much characterisation is truly required for different applications, selecting the right standard can be challenging. To help, LGC has developed a new interactive multimedia guide, packed with expert insights to support your decision-making and give you greater confidence when choosing the right IRS for your specific needs.
Using the Carcinogenic Potency Categorisation Approach (CPCA) to Classify N-nitrosamine Impurities
April 25th 2025Learn how to manage nitrosamine impurities in pharmaceuticals with our free infographic. Discover how the CPCA approach establishes acceptable intake limits and guides the selection of NDSRI reference samples. Stay compliant and ensure safety with our ISO-accredited standards.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)














