Summary of FDA Method C-010.01: Determination of 16 Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) in Food Using Liquid Chromatography–Tandem Mass Spectrometry (LC–MS/MS)
Summary
Per- and polyfluoroalkyl substances (PFAS) are a diverse group of synthetic organofluorine compounds that have been widely used in industrial applications and consumer products, such as nonstick cookware, food packaging, fire-fighting foams, carpeting, apparels, and metal plating. PFAS are persistent in the environment and bio accumulative in humans and wildlife, and said to cause reproductive and developmental toxicity. This application note summarizes a validated procedure developed by the US Food and Drug Administration (FDA) for the measurement of 16 PFAS in food matrices using a QuEChERS sample preparation approach and LC–MS/MS analysis.

Sample Extraction
The edible portion of the food sample is collected and homogenized using an IKA tube mill with a disposable 100 mL polypropylene grinding chamber (an alternative homogenizer may also be used for this step). Samples are ground at 5000 rpm for approximately 2 min. The minimum sample size for analysis is 5 g.
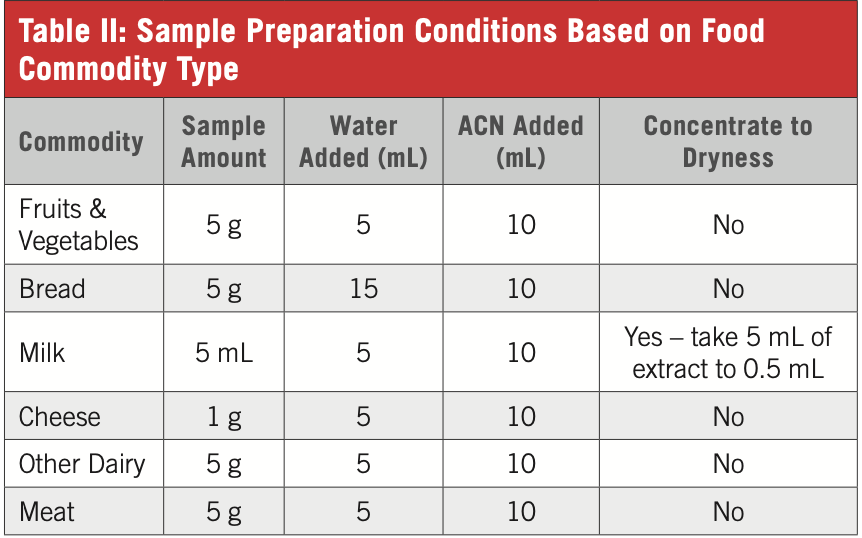
(1) Add amount of sample and LC–MS grade water based on Table II and commodity type to a 50 mL polypropylene (PP) centrifuge tube.
(2) Add 10 μL of 1 μg/mL isotopically labeled surrogate standard solution to the sample.
(3) Add 10 mL acetonitrile and 150 μL formic acid to the 50 mL PP conical centrifuge tube.
(4) Shake vigorously for 1 min.
(5) Add QuEChERS salt packet and shake for 5 min.
- Glas-Col® digital pulse vortexer at 1500 rpm with pulse set to 70
- Geno/Grinder® 2010 at 1500 rpm
(6) Centrifuge the samples at 10000 rcf for 5 min.
Sample Clean-up
(1) Transfer supernatant to the 15 mL dSPE tube
(2) Vortex/shake for 2 min.
(3) Centrifuge the samples at 10000 rcf for 5 min.
(4) Filter 5 mL of the extract with a 0.2 μm nylon syringe filter and transfer to a 15 mL conical centrifuge tube.
(5) Add internal standard: For samples that do not require nitrogen concentration:
- Add 5 μL of 1 μg/mL d5-N-EtFOSAA internal standard solution to the 5 mL extract to give a final concentration of 1 ng/mL. Surrogates will also have a final concentration of 1ng/mL in the final extract.
For samples that require nitrogen concentration:
- Concentrate to near dryness with nitrogen and reconstitute to 0.5 mL with methanol.
- Add 5 μL of the 1 μg/mL d5-N-EtFOSAA internal standard solution to give a final concentration of 10 ng/mL in solution. Surrogates will also have a final concentration of 10 ng/mL in the final extract.
(6) Briefly vortex and shake.
(7) Transfer sample to a polypropylene autosampler vial for analysis by LC–MS/MS.
Results
A level 2 single laboratory validation was conducted under the Guidelines for the Validation of Chemical Methods for the FDA FVM Program 2nd Ed. A total of four different types of foods and beverages were evaluated. These include produce, milk, fish, and bread. The method was validated at six concentrations (0.05, 0.15, 0.5, 1.5, 2, 5 ng/mL) in four food matrices. Acceptable recovery ranges for these compounds based on the FDA guidelines for the validation of chemical methods is 40–120% for concentrations spiked at 1 ng/mL. All compounds were within the acceptable range, except for 11Cl-PF3OUdS in bread samples which were on the lower side at 26–42% recovery.

UCT, Inc.
2731 Bartram Road, Bristol, PA 19007
Tel. (215) 781-9255, Fax (215) 785-1226
Website: www.unitedchem.com


.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)





