Recent Advances and Trends in Inductively Coupled Plasma–Mass Spectrometry and Applications
Special Issues
There have been exciting recent advances in ICP-MS instrumentation, such as the development of magnetic sector ICP-MS, multicollector ICP-MS, time-of-flight ICP-MS, and triple-quadrupole ICP-MS, as well as developments in the coupling of laser ablation (LA) and laser-induced breakdown spectroscopy (LIBS) to ICP-MS. This article surveys these developments and looks to the future.
Today, quadrupole inductively coupled plasma–mass spectrometry (ICP-MS) occupies an invaluable position in modern analytical laboratories worldwide because of its multielement and multi-isotope capability, high sensitivity, very limited interference effects, precision, and accuracy. Over the past three decades, however, there have been exciting advances in ICP-MS instrumentation, such as the development of high-resolution or magnetic sector ICP-MS, multicollector ICP-MS, time-of-flight ICP-MS, and triple-quadrupole ICP-MS. In addition, there have been important developments in the coupling of laser ablation (LA) and laser-induced breakdown spectroscopy (LIBS) to ICP-MS. This article surveys these developments and looks ahead to the future.
One of the most innovative developments in the area of analytical instrumentation during the past 50 years is the development of the quadrupole inductively coupled plasma–mass spectrometry (ICP-MS) and the subsequent developments of magnetic sector and time-of-flight (TOF) instruments (1–3). Over the past three decades, ICP-MS, with its excellent detection limits, multielement and multi-isotope capability, limited interferences, and a wide linear dynamic range, has become widely accepted as an established technique for trace- and ultratrace-element analysis, with thousands of instruments in use all over the world not only in the forefront of major areas such as earth, environment, ocean, and pharmaceutical sciences but also in emerging areas like proteomics and nanoparticle analysis (4–8). Recent developments in microelectronics and information processing have immensely contributed to making these instruments extremely smart, user friendly, and compact (9). The quest for interference-free determination and lower detection limits has driven numerous subsequent developments in recent years, which include coupling of laser-ablation sampling with laser-induced breakdown spectroscopy (LIBS), collision–reaction cells even in multicollector instruments, and multiple quadrupole MS arrangements. This article examines some of these recent developments in ICP-MS technology that have made considerable impact in science and technology.
ICP-Tandem Mass Spectrometry
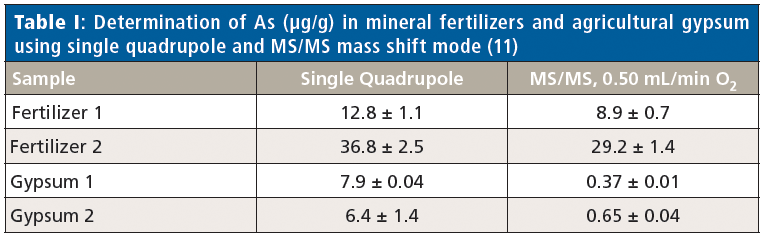
To overcome spectral interferences in quadrupole ICP-MS, features such as collision–reaction cells and kinetic energy discrimination (KED) have been introduced. Recently, two manufacturers have introduced ICP-MS/MS systems (or triple-quadrupole ICP-MS) systems, which further improve on the interference-removal capabilities of collision–reaction cells. In contrast to the traditional quadrupole-based ICP-MS systems equipped with a collision–reaction cell, ICP-MS/MS is characterized by the presence of two quadrupoles with a collision–reaction cell in between, which can be operated with a variety of cell gases, although one firm uses an octopole collision–reaction cell to do the similar function (Figure 1). The first quadrupole provides a mass-selection step before the cell, which gives better control over the ion–molecule reactions taking place in the cell by removing the matrix, followed by the mass selection by the second quadrupole. This double mass selection before and after the reaction cell, together with controlled ion–molecule chemistry, enables the determination of elements difficult to determine without this capability and delivers more accurate results for complex samples. It also improves detection limits. For example, 129I in soil samples was determined using ICP-MS/MS, with the objective of investigating radioiodine released by the Fukushima Daiichi Nuclear Power Plant (FDNPP) accident. High background caused by 129Xe impurities in argon plasma gas and polyatomic ions such as 127IH2+ and 127ID+ usually make it difficult to determine this isotope using conventional ICP-MS instruments. Oxygen was used as a reaction gas to reduce the background intensity of m/z 129, principally by 129Xe. Accurate and extremely precise 129I/127I ratios obtained are consistent with those obtained by accelerator mass spectrometry (AMS) (10). In another interesting application, Machado and colleagues (11) have accurately determined arsenic using ICP-MS/MS by applying mass shift reaction in the collision–reaction cell. Accurate determination of 75As+ usually is a challenge because of the well known isobaric overlaps caused by polyatomic species, such as 40Ar35Cl+, originated from the presence of elements of the matrix in the argon plasma. In ICP-MS/MS, 75As+ species are converted to 75As16O+ ions by the addition of oxygen gas into the collision–reaction cell, which allows the accurate determination of As in samples with high contents of rare earth elements (REEs), with REE-related interfering species such as 150Nd2+ and 150Sm2+. The results presented in Table I show that in single-quadrupole mode concentrations are higher than those determined by MS/MS mass shift mode, and this difference is more pronounced in agricultural gypsum samples, where REE concentrations are up to 10-fold higher than those found in fertilizers. ICP-MS/MS also showed greater ability for the accurate determination of the fission products 135Cs, 137Cs, and 90Sr, which are radioactive and have detection limits as low as 0.01 pg/mL (12). There are several such examples in the recent literature where ICP-MS/MS has been very effectively used for the sensitive and interference-free determination of various elements in several matrices (13).
Tandem LA-LIBS Coupled to ICP-MS
Laser-ablation ICP-MS (LA-ICP-MS) is a powerful technique for direct solid analysis with unique capabilities such as microanalysis, depth profiling analysis, two-dimensional elemental or isotope mapping, and the analysis of conductive and nonconductive materials. Another emerging analytical technique for direct solid analysis that uses LA, laser-induced breakdown spectroscopy (LIBS), is an atomic emission technique. Recently, these two fundamentally different techniques have been incorporated into a single commercial instrument and the resulting LIBS–LA-ICP-MS setup has the benefits of both techniques. All three forms of ICP-MS, namely, quadrupole, time-of-flight, and magnetic sector instruments have been used for this kind of technique hyphenation. LIBS uses the characteristic photons generated during the relaxation of the excited atomic and ionic species to create a spectral signature of the elements, and the analysis can be performed under ambient conditions. Typical limits of detection of metallic elements reported for LIBS are in micrograms-per-gram to weight percent levels. LA-ICP-MS, on the other hand, acquires the mass-to-charge ratios characteristic of the elemental composition of the ejected particles resulting from the laser–material interaction with detection limits of different elements in the range of sub-microgram-per-gram levels. Because isotopic information can also be obtained by LA-ICP-MS, the combination of LIBS and LA-ICP-MS (Figure 2) can be used to obtain chemical information (both elemental and isotopic) over a wider concentration range. There are some exciting applications in the recent literature where simultaneous tandem LA-LIBS coupled to an ICP-MS system had an impact on qualitative and quantitative analyses applications (14,15).

Figure 1: Schematic diagram of a triple-quadrupole ICP-MS system with a collision–reaction cell located between two quadrupoles. (Courtesy of Agilent Technologies.)
Despite the tremendous advances in LA-ICP-MS and features such as high sensitivity and resolution, certain elements and isotopes remain largely unmeasurable by LA-ICP-MS. Recurring limiting factors include isobaric interferences, abundance sensitivity, and detector saturation that forces masking of specific element masses and reduced sensitivity as a result of insufficient ionization of high ionization potential elements in the argon ICP plasma. These parameters collectively make the detection of elements such as F, O, H, N, Si, Se, As, Ca, and S very challenging or even impossible with conventional LA-ICP-MS systems. As a result, other techniques such as X-ray fluorescence (XRF) spectroscopy are being used to obtain complementary information. However, cross-correlation between exact sample locations, excitation volumes, and resolution (spatial and depth) may vary substantially, leading to faulty results. But tandem LA-LIBS coupled to ICP-MS is used to successfully chemically correlate, image, and map major, minor, and trace elements and isotopes through two-dimensional (2D) layer-by-layer and 2D cross-sectional imaging as well as three-dimensional (3D) volume reconstruction of the elemental distribution in geological samples (16) (Figure 3). By adopting computed tomography imaging principles to visualize multiple elemental and isotopic distributions in bastnaesite mineral ore matrix, expanded elemental coverage from a single sampling event is possible. In a forensic application, Subedi and colleagues (17) used this combination technique to discriminate printing inks with information from the atomic or ionic emissions and isotopic composition (m/z) by using the fingerprint spectra of each ink sample. Such combination instrumentation is becoming very popular with several such examples having been seen in different areas of application.
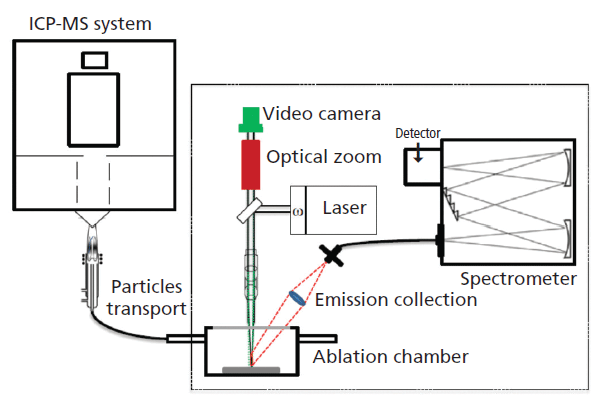
Figure 2: A schematic system of a tandem LIBS-LA-ICP-TOF-MS system (29).
Collision–Reaction Cell Multicollector ICP-MS
Nontraditional stable isotopes such as V, Cu, Mo, Ba, K, Pt, Pd, Ag, Ce, Er, and Si, with small variations in natural stable isotopic composition, have emerged as powerful tracers in geosciences, archaeology, nanotechnology, and environmental health studies. Mass differences in stable isotopes also give rise to fractionation during physical processes as a result of differences in the velocities of isotopic molecules of the same compounds. Information about the differences in these isotopes can help researchers understand physicochemical processes such as mass transfer and temperature changes. For example, magnesium is one of the most abundant elements on the Earth and has three isotopes (24Mg, 25Mg, and 26Mg) with relative mass difference between 24Mg and 26Mg of ~8%. Mg isotopes can be significantly fractionated during geochemical and biogeochemical processes. High-precision Mg isotope ratios allow us to understand geochemical processes in low-Mg rocks as well as many high-Mg rocks (18). Cu isotopic composition in the human body can help to identify the sources of Cu in the body as well as processes that are responsible for the movement of Cu in the body and to understand how reactions involving copper may be affected by disease. Until recently, measurement of these stable isotopic compositions has been carried out by thermal-ionization mass spectrometry (TIMS), and multicollector (MC)-ICP-MS (19). The current challenges in the measurement of nontraditional stable isotopes in complex samples by MC-ICP-MS lie in the identification and monitoring of spectral interferences, because certain molecular species can interfere directly with the atomic ions of the same nominal mass, leading to inaccurate isotope ratio determination. For example, 40Ar56Fe directly overlaps on 96Mo during molybdenum isotope ratio measurement, and the analysis of certain isotopes such as 40Ca is not possible on traditional MC-ICP-MS instruments because the 40Ar interference cannot be resolved. In addition, the low sensitivity offered in higher resolution mode is another problem because for every 10-fold increase in resolving power, there is a concomitant decrease in sensitivity. But recently one firm has introduced a collision–reaction cell MC-ICP-MS instrument with a novel design that incorporates both a "high energy" ion path (traditional MC-ICP-MS) and a separate "low energy" ion path (collision–reaction cell-MC-ICP-MS) (Figure 4) to enable accurate and precise measurement of these stable isotopes to meet the requirements of real applications. The instrument offers an ideal solution for the precise and accurate isotopic analysis of both the traditional isotope systems, and the nontraditional isotope systems.
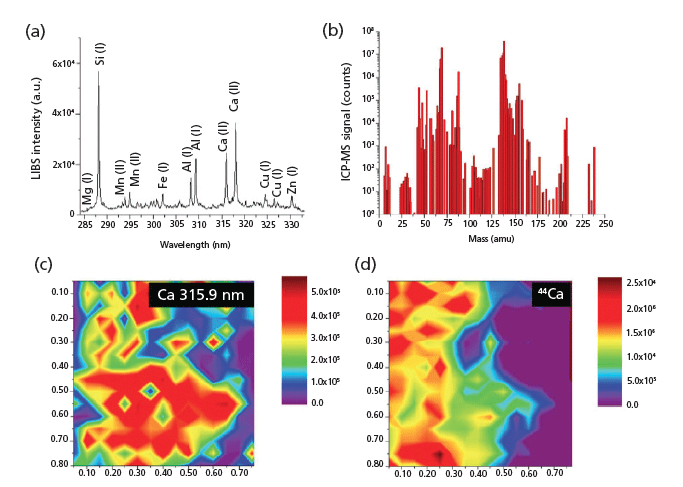
Figure 3: Representative LIBS (a) and LA-ICP-MS (b) spectra acquired from a single location of the sample. Surface (2D) distribution maps of Ca of the same 0.785 x 0.785 mm2 area of a bastnaesite rock sample obtained simultaneously by (c) LIBS and (d) LA-ICP-MS (16).
Other Significant Developments
Instrumental Improvements
The new-generation ICP-TOF-MS instruments are becoming more popular because of high-speed, high-spatial resolution, multielement and multi-isotope imaging that became possible as a result of advances in both LA and ICP-MS technologies. Though one firm has developed an ICP-simultaneous mass spectrometer that uses a complementary metal oxide semiconductor (CMOS) detector to simultaneously measure all masses from Li to U (20), for unknown reasons, the expected success has not been achieved so far. Recently, Obayashi and colleagues (21) have developed an LA-MC-ICP-MS system equipped with multiple-ion counting using three Daly detectors, which dramatically shortened the analysis time down to 1–5 s for in situ isotope ratio measurement over the conventional U–Pb age determinations using single-collector ICP-MS instruments. Conventional multiple collection using the Faraday detectors can be erroneous when the signal intensities of the analytes are unstable, particularly when the laser-ablation sampling technique is adopted. Deterioration in both the precision and accuracy of the isotope ratio measurements can be a result of the slow response of the Faraday amplifier. Daly detectors showed better long-term gain stability and wide dynamic range compared to conventional electron multipliers. Other improvements include the development of mini torch that consumes only two-thirds the argon compared to a conventional ICP torch, and improved spray-chamber systems with features such as faster temperature equilibration and extended temperature range (such as –25 °C to 80 °C with an accuracy of ±0.1 °C) for improving the performance of ICP-MS by effectively improving signal stability and reducing oxide interferences.
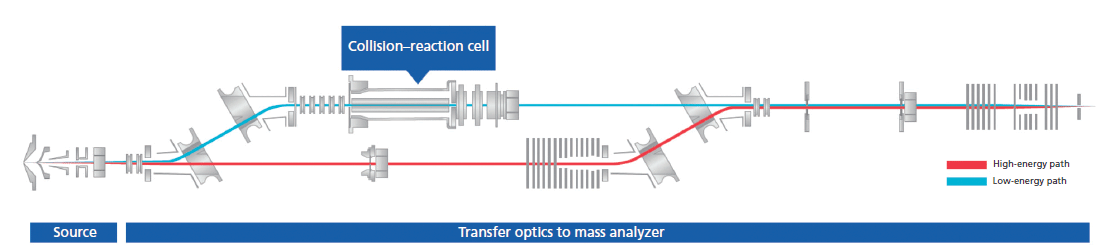
Figure 4: Schematic diagram of a collision-reaction cell MC-ICP-MS instrument with dual path transfer optics. (Adapted with permission from Nu Instruments.)
Femtosecond Lasers for LA-ICP-MS
One of the most fundamental limitations of LA-ICP-MS has been a lack of reproducibility, which can create problems during quantitative analysis, especially in heterogeneous materials where difficulty in matrix matching between the sample and standard may introduce errors. However, LA-ICP-MS (all forms) with its high sensitivity, capability of isotope ratio measurements, and excellent spatial resolution has seen a surge in its applications in environmental and life sciences because of the introduction of femtosecond laser systems, which offer distinct improvements in terms of matrix effects and elemental fractionation. This performance improvement is also a result of the availability of a wide variety of certified reference materials (22). In addition, the reduction of the internal volume of the laser-ablation cell for faster washout time contributed to the successful application of LA-ICP-TOF-MS to high-speed and high-spatial resolution multielement analysis (23), and also for elemental characterization of diverse samples such as toners, inks, papers, gunshot residues, fragments of automotive paints, and samples of hair and bones in forensic studies.
Speciation Studies
Analysis of animal- and plant-based foods for toxic elements such as Pb, Cd, As, Se, and Hg is of increasing importance because of raised consumer awareness and the need to evaluate and establish regulatory guidelines for these toxic trace metals and metalloids. Common examples include As, Cd, and Hg speciation in rice and As speciation in foodstuffs. For such applications, chromatographic techniques such as liquid chromatography (LC) and high performance liquid chromatography (HPLC) coupled to all forms of ICP-MS continue to be the methods of choice (24).
Single-Particle Analysis
Recently, single-particle ICP-MS (spICP-MS) has become a proven analytical technique for the characterization of nanomaterials and to provide information pertaining to size, size distribution, particle number concentration, and major elemental composition with minimal sample perturbation (25).
Quantitative Proteomics
Although ICP-MS does not directly facilitate protein identification, the high-temperature process inside an inductively coupled plasma leads to complete fragmentation of all sample molecules, leaving only their detectable atomic constituents, namely metals, metalloids, or heteroatoms (such as Se or P), which can be used as surrogates to detect complex molecules such as proteins, nucleic acids, or even small organic molecules. In this emerging area, ICP-MS is able to solve many challenges in quantitative proteomics that are hard to address by other techniques, as clearly seen in the large number of recent publications (26).
Miniaturization
Another emerging trend in the world of analytical instrumentation is miniaturization (9). Because of tremendous advances in microelectronics, instrument designs, and computer technology, today the majority of analytical instruments have moved to the bench-top and, in some cases, are available in portable or handheld options. Among other benefits, miniaturization can cut costs and make techniques more environmentally friendly by considerably reducing power and reagent requirements. Although there is a considerable size reduction in current ICP-MS instruments compared to the first-generation instruments, no ICP-MS instrument has yet become handheld so far, but with the kind of rapid advancements taking place currently in microelectronics, chip technologies, and other allied areas, that possibility cannot be ruled out for the future.
Sample Preparation Methods
Despite rapid developments and innovations in ICP-MS, sample preparation remains the main bottleneck of elemental and isotope analysis. Although laser sampling has made it possible to conduct in situ analysis on solid samples directly, most applications still require acid digestion (4). Sample preparation devices with very high digestion efficiency such as high-pressure ashers and high-pressure vessels for inorganic substances (27) and techniques such as microwave-assisted decomposing procedures for organic substances (28) are becoming more and more popular.
Quality Control
In all the measurements discussed here, there is a great need for stringent quality control measures. Because most analytical techniques, including ICP-MS, are comparative, a sample of known composition (reference material) is required for accurate calibration. One of the biggest challenges is the availability of calibrated elemental and isotopic reference materials for every application. However, international organizations like the National Institute of Standards and Technology (NIST) in the United States and the Federal Institute for Materials Research and Testing (BAM) in Germany support accurate and compatible measurements by certifying and providing hundreds of certified reference materials (CRMs) for many applications.
Future Challenges and Opportunities
As indicated by the broad range of examples discussed here, exciting developments have taken place in ICP-MS in recent years. ICP-MS/MS instruments further improve on the interference-removal capabilities of collision–reaction cells and also improve detection limits for several elements. Tandem LA-LIBS coupled to ICP-MS can provide unique images and can map major, minor, and trace element and isotope distribution information in different materials from a single sampling event. Collision–reaction cells greatly enhance the analytical capability of MC-ICP-MS, and this analytical tool is set to make an indelible mark in the areas of environmental, biological, geochemical, medical, and even in the emerging nanotechnology fields with its outstanding performance for the interference-free measurement of some of the most challenging, nontraditional stable isotope systems, and thus can provide a wealth of information. Procedural simplicity and larger throughputs make collision–reaction cell MC-ICP-MS a better choice than TIMS.
Though considerable advances have taken place in ICP-MS technology and instrumentation in recent years, challenges still exist as a result of the complex and variable matrices of different types of samples. All forms of ICP-MS instrumentation including recently emerged systems can be used for quantification only when suitable international CRMs are available for every kind of application. Thus, the availability of a range of CRMs, and perfect dissolution procedures, are extremely important for the success of these technologies. Novel application requirements continue to pop up from time to time, and bring challenges to instrument manufacturers.
References
(1) R.S. Houk, V.A. Fassel, G.D. Flesch, H.J. Svec, A.L. Gray and C.E. Taylor, Anal. Chem. 52, 2283–2289 (1980).
(2) N. Bradshaw, E.F.H. Hall, and N.E. Sanderson, J. Anal. At. Spectrom. 4, 801–803 (1989).
(3) P. Myers, G. Li, P. Yang, and G.M. Hieftje, J. Am. Soc. Mass Spectrom. 5, 1008–1016 (1994).
(4) M. Satyanarayanan, V. Balaram, S.S. Sawant, K.S.V. Subramanyam, V. Krishna, B. Dasaram, and C. Manikyamba, Atomic Spectrosc. 39(1), 1–15 (2018).
(5) U. Rambabu, V. Balaram, R. Ratheesh, S. Chatterjee, M.K. Babu, and N.R. Munirathnam, J. Testing and Evaluation 46(5) (2018). https://doi.org/10.1520/JTE20160645.
(6) V. Balaram, Trends Anal. Chem. 80, 83–95 (2016).
(7) L. Bush, Spectroscopy 30(6), 74–73 (2015).
(8) C.T. Kamala, V. Balaram, M. Satyanarayanan, A. K. Kumar, and K.S.V Subramanyam, Archives Environ. Contami. Toxicol. 68, 421–431 (2015).
(9) V. Balaram, Spectroscopy 31(10), 40–44 (2016).
(10) T. Ohno, Y. Muramatsu, Y. Shikamori, C. Toyama, N. Okabe, and H. Matsuzaki, J. Anal. At. Spectrom. 28, 1283–1287 (2013).
11) R. Machado, C. A. Virgilio, C. D. B. Amaral, D. Schiavo, J. A. Nóbrega, and A.R.A. Nogueira, J. Braz. Chem. Soc. 27(11), 2076–2081 (2016).
(12) W. Bu, J. Zheng, X. Liu, K. Long, S. Hu, and S. Uchida, Spectrochim. Acta Part B 119, 65–75 (2016).
(13) D. Kutscher, S. Lofthouse, S. Nelms, and S.M. Ducos, Spectroscopy 32(10), 38–41 (2017).
(14) B.T. Manard, C.D. Quarles, E.M. Wylie, and N. Xu, J. Anal. At. Spectrom. 32, 1680–1687 (2017)
(15) O. Syta. B. Wagner, E. Bulska, D. Zielinska, G.Z. Zukowska, J. Gonzalez, and R. Russo, Talanta 179, 784–791 (2018)
(16) J. R. Chirinos, D. D. Oropeza, J. J. Gonzalez, H. Hou, M. Morey, V. Zorba, and R.E. Russo, J. Anal. At. Spectrom. 29, 1292–1298 (2014).
(17) K. Subedi, T. Trejos, and J. Almirall, Spectrochim. Acta, Part B 118, 103–104 (2015).
(18) Y. An, F. Wu, Y. Xiang, X. Nan, X. Yu, J. Yang, H. Yu, L. Xie, and F. Huang, Chem. Geol. 390, 9–21 (2014).
(19) D. Lu, T. Zhang, X Yang, P. Su, Q. Liu, and G. Jiang, J. Anal. At. Spectrom. 32, 1848–1861 (2017).
(20) E. Carsten, Anal. Bioanal. Chem. 399(1), 213–219 (2011).
(21) H. Obayashi, M. Tanaka, K. Hattori, S. Sakata, and T. Hirata, J. Anal. At. Spectrom. 32, 686–692 (2017).
(22) A. Limbeck, P. Galler, M. Bonta, G. Bauer, W. Nischkauer, and F. Vanhaecke, Anal. Bioanal. Chem. 407, 6593–6617 (2015).
(23) Y. Bussweiler, O. Borovinskaya, and M. Tanner, Spectroscopy 32(5), 14–20 (2017).
(24) B. P. Jackson and T. Punshon, Curr. Envir. Health Report 2, 15–24 (2015).
(25) M.D. Montaño, J.W. Olesik, A.G. Barber, K.T Challis, and J.F. Ranville, Anal. Bioanal. Chem. 408(19), 5053–5074 (2016).
(26) O. Chahrour and J. Malone, Protein Peptide Letters 24, 1–14 (2017).
(27) S. Chen, X. Wang, Y. Niu, P. Sun, M. Duan, Y. Xiao, P. Guo, H. Gong, G. Wang, and Q. Xue, Science Bulletin 62(4), 277–289 (2017).
(28) C.A. Bizzi, M.F. Pedrotti, J.S. Silva, J.S. Barin, J.A. Nóbrega, and E.M. M. Flores, J. Anal. At. Spectrom. 32, 1448–1466 (2017).
(29) M. Dong, D. Oropeza, J. Chirinos, J.J. González, J. Lu, X. Ma, and R.E. Russo, Spectrochim. Acta Part B 109, 44–50 (2015).
V. Balaram, PhD, is an Emeritus Scientist and former Chief Scientist and Head of the Geochemistry Division at the CSIR–National Geophysical Research Institute (NGRI) in Hyderabad, India. Direct correspondence to: balaram1951@yahoo.com

Determining Enhanced Sensitivity to Odors due to Anxiety-Associated Chemosignals with GC
May 8th 2025Based on their hypothesis that smelling anxiety chemosignals can, like visual anxiety induction, lead to an increase in odor sensitivity, a joint study between the University of Erlangen-Nuremberg (Erlangen, Germany) and the Fraunhofer Institute for Process Engineering and Packaging (Freising, Germany) combined behavioral experiments, odor profile analysis by a trained panel, and instrumental analysis of odorants (gas chromatography-olfactometry) and volatiles (gas chromatography-mass spectrometry).
Investigating 3D-Printable Stationary Phases in Liquid Chromatography
May 7th 20253D printing technology has potential in chromatography, but a major challenge is developing materials with both high porosity and robust mechanical properties. Recently, scientists compared the separation performances of eight different 3D printable stationary phases.
Detecting Hyper-Fast Chromatographic Peaks Using Ion Mobility Spectrometry
May 6th 2025Ion mobility spectrometers can detect trace compounds quickly, though they can face various issues with detecting certain peaks. University of Hannover scientists created a new system for resolving hyper-fast gas chromatography (GC) peaks.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)







