Making it Crystal Clear: Detecting PPCPs in Water Using LC–MS
The Column
This article describes a simple, more convenient method than traditional solid-phase extraction (SPE)-based methods for highly sensitive PPCP detection using triple quadrupole liquid chromatography–mass spectrometry (LC–MS–MS).
Environmental regulatory bodies such as the United States Environmental Protection Agency (US EPA) monitor the composition of potable and environmental water to ensure its safety. Pharmaceutical and personal care products (PPCPs) can be found in water, having been released into wastewater from industrial sources and households. The presence of PPCPs in environmental and potable water is a widespread concern because of their potentially harmful effects to both humans and the environment. This article describes a simple, more convenient method than traditional solid-phase extraction (SPE)-based methods for highly sensitive PPCP detection using triple quadrupole liquid chromatography–mass spectrometry (LC–MS–MS).
(PHOTO CREDIT: LAUREN BURKE/GETTY IMAGES)

Pharmaceutical and personal care products (PPCPs) include any product used by individuals for health or cosmetic reasons, or by agribusiness to boost growth or health of livestock. This includes human and veterinary medicines, cosmetics, and household cleaning products. Wastewater from households and industrial sources contains multiple PPCPs of various combinations, concentrations, and relative toxicologies. PPCPs are bioactive, they do not dissolve easily or evaporate at normal temperatures or pressures, and therefore can easily enter soil and aquatic environments via sewage, biosolids, and irrigation. The presence of PPCPs in environmental and potable water is a concern because of the potentially harmful effects to both humans and the environment. There is also evidence to suggest PPCPs are linked to some ecological damage such as the delayed development in fish and delayed metamorphosis in frogs.1
To ensure the safety of water, PPCP concentrations are stringently monitored at trace concentration by environmental regulatory bodies such as the United States Environmental Protection Agency (US EPA).2 Detection is traditionally a complicated and laborious process because of the range of substances potentially present in clean water.
Conventional methods of PPCP detection have followed the defined US EPA 1694 "template" for analysis that requires the preconcentration of large volume water samples and solid-phase extraction (SPE) cleanup, followed by liquid chromatography–mass spectrometry (LC–MS) analysis to achieve low ng/L (ppt) level detection to comply with regulations.3 In this study a method was developed using ultrahigh-pressure liquid chromatography (UHPLC) with triple quadrupole LC–MS. PPCPs were detected at 1–2 ppt with a linear response up to 200 or 500 ppt. The incorporation of an on-line extraction module enabled method-driven on-line sample cleanup.
Method
Table 1: Test results for selected PPCPs in real water samples.
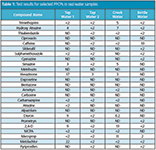
Standards and Samples: Several samples were collected from tap water, bottled water, and creek water (Table 1).
Instrument Set-Up: An EVOQ Elite triple quadrupole mass spectrometer (Bruker) was used for analysis. The set-up was as follows: HV 4000 V; cone gas flow: 15 units; cone gas temperature: 300 °C; heated probe gas flow: 40 units; heated probe temperature: 450 °C; nebulizer gas flow: 50 units; exhaust gas: On; Q2 pressure: 1.5 mTorr (Argon). A Bruker LC system was used and set-up as follows: Trap column: 35 mm × 2.0 mm, 3-µm YMC-Pack ODS-AQ; column temperature: 40 °C; injection volume: 400 µL; flow-rate: 400 µL/min. Solvent A: 2 mM ammonium formate, 0.1% FA in water. Solvent B: 2 mM ammonium formate, 0.1% FA in MeOH. Solvent C: 2 mM ammonium formate, 0.1% FA in water. Gradient conditions: 0.0 min, 10% B; 0.2 min, 10% B; 0.8 min, 25% B; 8.0 min, 95% B; 9.0 min, 95% B; 9.1 min, 10% B; 12.0 min, 10% B.
Results
Figure 1: Selected MRM chromatograms for PPCPs at 2 ppt.

Optimal conditions are achieved with the use of a more acidic third pump eluent for preconcentrating the water samples. Under these conditions, all of the PPCP species representing compounds were detected at 2 ppt or better with the injection of 0.4 mL samples with a linear response range up to 200 or 500 ppt. Replicate injections with 5 ppt level spiked in tap water demonstrated good robustness with relative standard deviation of 3–13%. The results for the analysis of tap, creek and bottled waters are shown in Table 1. Figures 1 and 2 show the excellent linear responses for the PPCPs at both positive and negative polarities in the same analytical segment.
Conclusion
Figure 2: Selected calibration curves.

The continual increase in the use and range of PPCPs in industries and households has necessitated more robust and sensitive screening techniques in wastewater effluents. The method described illustrates how UHPLC and LC–MS–MS systems can provide the sensitivity demanded by regulatory bodies, even with challenging ranges of samples at various concentrations. The technique also presents a more convenient and simpler approach to the analysis of PPCPs compared to traditional solid-phase extraction techniques. Together with more intelligent sample techniques, LC–MS provides convenient analysis methods in high-throughput laboratories.
References
1. K. Hirsch, Pharmaceuticals and Personal Care Products [Available from: http://serc.carleton.edu/NAGTWorkshops/health/case_studies/pharmaceutical.html](2013).
2. US EPA PPCPs Basic Information, [Available: http://www.epa.gov/ppcp/basic2.html] (2010).
3. US EPA, EPA Method 1694: Pharmaceuticals and Personal Care Products in Water, Soil, Sediment, and Biosolids by HPLC/MS/MS (2007).
Joe Anacleto is the VP for Bruker Daltonics Applied Markets Business Group.
Zicheng Yang is a senior field application specialist at Bruker Daltonics.
Helen (Qingyu) Sun is a product marketing specialist at Bruker Daltonics.
Kefei Wang was a global GC–MS product manager at Bruker Daltonics.
E-mail: cam-sales@bruker.com
Website: www.bruker.com/
This article is from The Column. The full issue can be found here: http://www.chromatographyonline.com/vol-10-no-16-column-september-04-2014-europe-and-asia-pdf

Determining the Effects of ‘Quantitative Marinating’ on Crayfish Meat with HS-GC-IMS
April 30th 2025A novel method called quantitative marinating (QM) was developed to reduce industrial waste during the processing of crayfish meat, with the taste, flavor, and aroma of crayfish meat processed by various techniques investigated. Headspace-gas chromatography-ion mobility spectrometry (HS-GC-IMS) was used to determine volatile compounds of meat examined.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)










