The Inert Flow Path Story for GC and GC–MS: Eliminating the Weakest Links
LCGC North America
For the next improvements, we must look beyond the column.
Advances in gas chromatography (GC), especially gas chromatography–mass spectrometry (GC–MS) detector technologies, are pushing the levels of detection to parts-per-billion and parts-per-trillion levels. However, detectors can only measure the analytes that reach them. Because a typical column contributes a majority of the surface area, column inertness has received a great deal of attention in the GC community. One must think of the GC flow path as a chain of components linked together to separate the analytes and deliver them to the detector. This installment looks at contributions of the entire GC flow path, identifying the remaining links that contribute to surface activity. Improvements in GC flow path inertness are highlighted.
Gas chromatography–mass spectrometry (GC–MS) and GC–MS-MS systems have increased in sensitivity dramatically during the last two decades, especially over the last five years. In the early days of benchtop GC–MS, single-digit nanogram on-column amounts were barely detectable in full-scan mode — today sensitivities are in the picogram to femtogram range in full-scan mode, and even more so in single-ion mode (SIM) and multiple reaction monitoring (MRM) mode with GC–MS-MS. This increase in sensitivity has enabled analysts to reach lower and lower detection limits. So, what is the challenge? The MS detector can only measure analytes that reach it. The lower the amount of analyte injected, the greater the chance that this analyte can be lost to any chromatographically reactive parts of the GC flow path. The problem for laboratories chartered to detect and measure trace components is the risk of not detecting trace analytes if the GC flow path is chromatographically reactive (active). The purpose of this installment of "Column Watch" is to take a look at the entire GC flow path and identify those system elements that may contribute to analyte loss (or extremely bad tailing) and nondetectability. Furthermore, we discuss actions that have been taken to eliminate (or substantially decrease) the contributions of the weakest links in the GC flow path.
What Is the GC Flow Path?
The GC flow path is essentially every component that the injected sample potentially touches. To keep the scope of this column installment reasonable, the boundary begins at the sample inlet and ends at the mass spectrometer source (Figure 1). While other detection methods (such as electron-capture detection [ECD], pulsed flame photometric detection [PFPD], or sulfur chemiluminescence detection [SCD]) are frequently used in trace analysis, the focus on MS over other detection systems is deliberate because it is the most widely used GC detection method in trace analysis. The point-of-injection is when the sample leaves the syringe or sample loop and enters the carrier gas flow stream. After the separated components leave the gas chromatograph itself and enter the source of the MS system, ionization takes place and the path of the ions through the MS system is no longer considered to be part of the GC flow path.
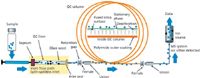
Figure 1: Gas flow path in a typical GCâMS system.
What Is Chromatographic Reactivity?
In any given chromatographic system at the molecular or atomic level, there is a finite number of active sites (for example, molecules or atoms) on the surfaces of the chromatographic flow path materials that may interact with analytes of interest. This number of active (or reactive) sites is generally fixed on a new or freshly maintained system. Here is an example to illustrate why this is important for trace analysis: If a system has 10 reactive sites and 100 reactive sample molecules are injected, there is the potential to lose 10% of the sample molecules, probably not significant in routine trace analysis. If only 10 sample molecules are injected on a system that has 10 reactive sites, 100% of them could be lost to the reactive sites in the system. The end result — even though reactive analytes of interest are injected, they will never be detected if they are lost on reactive sites in the GC flow path. The impact of "non-detects" can be profound. Our food, drinking water, environment, and commercial goods are less safe and are of poorer quality, criminals go free, and warfare agents go undetected.
Some examples of active sites in a typical flow stream could be
- silanols on the inner surface of a fused silica capillary column tubing, retention gaps or guard columns, and inlet liners
- metallic atoms or molecules (such as metal oxides) on the surface of stainless steel weldments or other fabricated components (such as gold seals or connecting tubing)
- silanols or other molecular sites on the glass wool often used in inlet liners
- ferrules and unions used to connect the column or other devices into the flow stream.
What Parts of the GC Flow Path Contribute to Chromatographic Reactivity?
There are a number of sources of activity that a given sample will encounter in the sample flow path. Fortunately, a basic understanding of chemical reaction kinetics enables us to understand the potential relative contribution of a given GC flow path component to the loss of a reactive analyte.
Temperature: The higher the temperature at the time of analyte contact, the more likely the system component will be reactive.
Nature of Reactants: Of the compounds that are typically analyzed by GC, on one end of the spectrum are hydrocarbons: they are very nonreactive at normal GC temperatures (which is one reason why they are frequently used as internal standards for trace analysis methods). On the reactive end, there are polar analytes like phenols, amines, and thermally labile components (molecules that have complex structures with inherently strained bonds).
Catalysts: In the GC flow path there are really only three relevant general material types that are potentially catalytic:
Metals >> Glass >>> Organic coatings
Within these three subsets, there are varying degrees of activity. Certain metals may be more catalytic than others; sometimes special metal components are used in GC. Surface silanols on glass or fused-silica columns and glass wool vary in acidity (for example, isolated, vicinal, and hydrogen bonded). The organic coatings for wall-coated open tubular GC columns may have a fairly low degree of activity. A familiar culprit is the deactivation coating used on glass wool. When glass wool is deactivated outside of the injection liner and then a plug is inserted, often fibers are broken, which exposes active sites that can cause reactions with certain analytes. We will discuss this phenomenon in more detail later.
Physical State: One side of the physical state equation has been addressed in the above example of trace analysis. The other side of the equation is the contribution of the "physical state" by the GC system. One way to understand these physical contributions is to consider the "surface area" of each flow path component. The greater the surface area of catalytic material that a volatilized analyte is exposed to, the more it is a potential contributor to system reactivity. All of the exposed surfaces within the flow stream may not contribute equally to analyte–surface interactions, but for a first approximation, exposed surface area is a good place to start by comparing potential contributors to analyte loss or peak tailing.
GC Flow Path: A Chain of Components
When all the above elements are considered, it is relatively easy to understand which GC flow path components pose the most risk to analyte loss. Figure 1 provides an overview of some components that might be found in a typical GC flow path. Table I estimates the contributions of various flow path elements to potentially exposed surface area as well as the typical operating temperature that may enhance any chemical reactions or interactions that can occur.
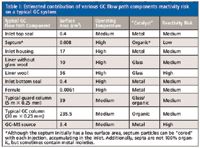
Table I: Estimated contribution of various GC flow path components reactivity risk on a typical GC system
We will discuss some of these contributions in the next section.
GC Capillary Columns
As depicted in Table I, the GC column itself is, by far, the biggest overall contributor to exposed surface area. More than a decade ago, even after chemically bonded and cross-linked GC capillary columns were introduced and being used, manufacturers started to address user problems associated with GC capillary columns employed with the increasing detection sensitivity, particularly with ECD and MS. Users were often baffled by the baseline drift during temperature programming because of stationary phase bleed, loss of analytes, and tailing of polar compounds, particularly at lower levels on state-of-the-art columns of the day. The first problem that was addressed was column bleed or baseline drift. Throughout the 1990s and into the 2000s, new columns with better bleed specifications were introduced with manufacturers "out-spec'ing" each other such as factors of four lower bleed and higher temperature limits. For examples, see the articles on GC columns introduced at Pittcon by Majors during that time period (1–4). Columns began to be specified with two upper temperature limits (for example, 225 °C and 250 °C). The first number was the upper temperature limit for isothermal operation and the second number was the upper temperature limit for programmed runs in which the column was not expected to perform for hours on end because it was normally cooled down to get ready for the next temperature-programmed run. New phase chemistries were developed that had the same or fairly similar selectivities to the popular phases at that time, such as 100% dimethylpolysiloxane and 5% phenyldimethylpolysiloxane. Arylene phases were introduced that had lower densities since the phenyl groups were more embedded within the polymer chain than their pendant phenyl counterparts, and thus, bleed mass was reduced for equivalent film thickness columns.
Indeed, most GC column manufacturers developed a second-generation family of columns that performed better than the columns that preceded them. Table II lists some typical column families that were introduced with improved specifications, most of them with an MS designation because of the growing popularity of this detection mode. Nowadays, ultrainert columns are the name-of-the-game with not only improved bleed specs, but better analyte recovery and lower tailing for polar compounds being observed. The variety of stationary-phase types offered with extremely inert characteristics has since expanded from apolar phases to more polar phases.
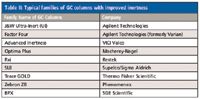
Table II: Typical families of GC columns with improved inertness
Enter Inert Flow Path Components
However, with regulatory agencies driving lower limits of detection for increasingly active and more complex samples, other parts of the GC flow system started to contribute adversely to the analytical results. Having to repeat or verify suspect trace analysis wasted the analysts' time and hindered productivity. Fortunately for the trace analysis community and the stakeholders served, leading instrument and consumables companies began to develop commercially available "inert upgrades" to the flow path components that have, for all practical purposes, made loss of analyte at trace levels negligible. This has enabled more accurate results for trace analysis.
The first area to be addressed was the injector port and, particularly, the inlet liner itself. Consisting of borosilicate glass often deactivated with silane reagents and other surface treatments to prevent analyte interaction, it was found that some of the lack of inertness was attributed to the hydrolysis of the deactivated liner surface, particularly when moisture was present in the sample and carrier gases were not treated with moisture traps to remove residual water. Because the liner is at a high temperature, any surface activity effects were accentuated. Oxygen present in the carrier gas had to be scrubbed via oxygen traps. Contamination of the liner with sample matrix or bits of septum material from "coring" because of needle design were other problems that had to be addressed. To combat the first problem, manufactures came up with faster methods to change liners (some automated) so that users didn't have to shut down their gas chromatographs to change hot inlet components. To address the second problem, new septa and injector needles were designed that minimized the contribution to flow path activity of cored septum fragments. The exposed surface area of the GC septum itself, which is considered part of the flow system, is very small (Table I) and over the years polymer formulations have been developed that cause little activity problems, even in splitless backflash situations. Nevertheless, the inlet liner itself still needed to be addressed and most recently the use of new organic chemical vapor deactivation processes for glass materials beyond the typical siloxane chemistry has allowed the introduction of new inert borosilicate liners for improved performance with trace polar analytes.
However, one problem area remained that seemed to be a continuing saga for gas chromatographers: the preferred use of glass wool packed into the inlet liner. Glass wool serves two purposes: to "catch" impurities, especially nonvolatiles, that could enter the GC column and cause further problems with the column or detector; and to serve as a mixing volume and a thermal mass aid and ensure vaporization of the sample and solvent in the injection port. It is worth noting that Table I illustrates the fairly high surface area that an inlet liner with glass wool has relative to the other components in the flow path. This is because of the very small fiber diameter of the wool. If the glass wool was properly deactivated and correctly placed in the inlet liner, then its contribution to surface activity would be fairly minimal. However, when glass wool was deactivated externally and the delicate fibers were then "wadded up" and carefully placed in the liner, not surprisingly, fragments of the underlying high activity surface glass wool were inadvertently created that contributed to undesirable effects, particularly for trace polar compounds. In fact, many pesticide chemists refused to use glass-wool-packed liners because their trace pesticides would certainly be lost and their analytical results would be compromised.
The solution to this problem was to develop high purity, high surface area glass wool, carefully place it in the glass liner and then deactivate it in situ. Again, new deactivation procedures using organic coatings were used that gave even better results than traditional silylation. In this manner, high-surface-area glass wool fragments were not created and the new deactivated glass-wool liners provided a high level of inertness. These dramatic advances in organic coatings on high-surface-area glass wool have enabled their use without compromising inertness, enabling analysts to enjoy the benefits of glass wool and all the associated reduced operational expenses coming from a longer-lived GC column.
With respect to other elements of the flow path, the application of more inert coatings over metal surfaces has enabled better trace analyte "fidelity" for inlet housings, inlet bottoms, and top seals. Bottom seals consist of stainless steel or gold-plated stainless steel. Even gold seals have been shown to provide active sites and also must be further deactivated to ensure an optimum surface for trace polar analytes. In many cases, in the absence of sample backflash, many of the surfaces in the injection port aren't entirely exposed to sample vapor and their surface areas don't contribute to major activity problems. On the detection side, proprietary metal deactivation procedures and special metal alloys are also used to cut down on surface activity; even detectors with a high metal content, such as the thermal conductivity detector, now contribute less to analyte adsorption than earlier models. For flame ionization detection (FID), if the capillary column is correctly positioned there is almost no exposure to hot metal surfaces that could cause potential problems. Nevertheless, deactivated FID housings are also available.
Although not discussed here, Figure 1 also shows some additional parts such as retention gaps, ferrules, and unions that can also contribute to the overall system performance. Indeed, in modern gas chromatographs, these components also require the same deactivation treatments that will render them harmless. As with all products, inertness claims, proof of inertness performance, and actual inertness performance may vary among manufacturers.
Putting It All Together
Figure 2 shows the comparison of a trace injection of drugs of abuse into a standard GC system versus a GC system with optimized components for ultrainert performance: an ultrainert capillary column, an ultrainert glass liner with deactivated glass wool, and all metal parts deactivated using proprietary procedures. Not only do the drug peaks in the ultrainert system give much stronger signals (lower detection limits) and better peak shape, but for temazepam a peak that was "lost" on the standard system, gives a nontailing peak on the high performance GC system. More definitive information can be found in reference 5.

Figure 2: Analysis of drugs of abuse at the 500 ppb level: inert flow path (green trace) vs. a standard flow path in a GCâMS (5). Instrument: Agilent 7890 GC system with 5975 C MSD detector; column: 30 m à 0.25 mm, 0.25-μm df J&W HP-5ms UI (Ultra-Inert); gas purifier: Gas Clean GCâMS, 1/8-in. kit; temperature program: 100 °C (4-min hold); 100â280 °C at 10 °C/min, 280â300 °C at 6 °C/min (4.67 min hold); carrier: helium at 52.7 cm/s (2 mL/min), set at 100 °C with EPC-Constant flow; inlet: pulsed splitless, 1 μL, 35-psi pulse until 0.73 min, 0.75-min purge at 50 mL/min, gas saver 20 mL/min at 2 min; inlet liner: Ultra Inert with wool (inert flow path), standard single taper liner with wool (standard flow path); seal: UI Gold seal (inert flow path), standard Gold seal (standard flow path); detector: MSD scan mode 40â450 m/z; source temperature: 230 °C; quadrupole temperature: 150 °C; transfer line temperature: 310 °C. Peaks: 1 = dronabinol, 2 = oxycodone, 3 = temazepam, 4 = flunitrazepam, 5 = diacetyl morphine.
Conclusions
With respect to trace analysis, the GC and GC–MS instruments of today outperform those produced only a few years ago. As the manufacturers of GC instruments and consumables continue to improve GC flow path parts exposed to the precious samples, users performing trace analysis in these ultrainert systems will have the same degree of confidence in their analytical results that general users have had for years in GC laboratories around the globe. Even simple procedures such as "touchless" packaging can bring major advantages to trace analysts in the prevention of accidentally contaminating frequently replaced flow path components, such as septa and inlet liners. Undoubtedly, as regulatory bodies or other user demands require lower limits of detection, challenges to further increase system inertness will continue to be confronted.
References
(1) R.E. Majors, LCGC North Am. 21(4), 332–349 (2003).
(2) R.E. Majors, LCGC North Am. 25(4), 340–358 (2007).
(3) R.E. Majors, LCGC North Am. 28(4), 274–290 (2010).
(4) R.E. Majors, LCGC North Am. 31(5), 364–381 (2013).
(5) K. Lynam, Agilent Application Note 5991-1859EN, Feb. 2013.
Mitch Hastings is a manager with the chemistry and supplies division of Agilent Technologies located in Folsom, California. He learned the majority of his chromatography from GC Column pioneer Dr. Walter Jennings and from experts within Agilent and the former J&W Scientific. Outside of work, he enjoys cycling and watching his son play sports.

Mitch Hastings
Ronald E. Majors "Column Watch" Editor Ronald E. Majors is a Senior Scientist in the Columns and Supplies Division at Agilent Technologies (Wilmington, Delaware), and is a member of LCGC's editorial advisory board. Direct correspondence about this column to lcgcedit@lcgcmag.com.

Ronald E. Majors

Perspectives in Hydrophobic Interaction Temperature- Responsive Liquid Chromatography (TRLC)
TRLC can obtain separations similar to those of reversed-phase LC while using only water as the mobile phase.