Immunosorbents for Selective Sample Preparation of Complex Mixtures
Selective sample preparation techniques are particularly attractive for the analysis of trace amounts of small molecules in complex matrices. In this month's instalment, columnist Ron Majors covers the field of immunoextraction, a technique that employs immobilized antibodies to selectively capture specific analytes using molecular recognition via antibody–antigen interactions. Recently, the introduction of commercial products for specific high-volume environmental and food safety applications should spur further applications of this technique.
Despite the advances that have been made in the sensitivity and selectivity of analytical instrumentation, for complex samples encountered in environmental and food matrices, some form of sample pretreatment is almost always required to isolate the analytes of interest. For liquid samples, the use of liquid–liquid extraction (LLE) and solid-phase extraction (SPE) have been the most popular approaches to pretreat these samples. Many methods have been published using one or both of these techniques to simplify complex mixtures before chromatographic or spectroscopic analysis. In some instances, LLE has given way to SPE because there has been a trend to eliminate or reduce the consumption of organic solvents, especially chlorinated solvents. Even though there have been many new SPE sorbents introduced that provide more selective interactions, workers still prefer to use nonselective hydrophobic sorbents for their sample preparation needs.1 Even in sorbents considered to be more selective, such as ion exchange SPE packings, analyte retention is often highly influenced by hydrophobic interactions with the organic portion of the sorbent surface. When these interactions occur, coextraction of other analytes and matrix interferences generally occurs, often a big problem when the analytes of interest are in trace concentrations. Then, additional clean-up procedures are required, increasing the chance of analyte loss and contamination by the introduction of other chemical species.
Thus, there is a need for clean-up methods that will result in more selective extraction of the analytes of interest, hopefully in a single step. In the past decade, there has been an increasing level of interest in the development of highly selective sorbents based upon immunoaffinity interactions. Such sorbents involve antigen–antibody interactions that are based upon molecular recognition and, thus, are very specific. The antibodies are immobilized onto solid supports such as silica or polymeric adsorbents that are classified as immunosorbents (ISs). Then, these sorbents are used in a manner similar to SPE cartridges. The analyte isolation process is termed immunoextraction or immunoaffinity extraction. Sometimes, when the experiment is performed in a column format, it is referred to as an immunoaffinity chromatography (IAC) but the process involved is not really a chromatographic mechanism.
There have been many papers published on the subject of ISs and immunoextraction but only recently have commercial products become available that can be applied conveniently to real world applications. This instalment of "Sample Prep Perspectives" will be devoted to an introduction to ISs and their potential applications for small molecules that might be encountered as pollutants in environmental and food samples. For those who would like in-depth coverage, there are a number of excellent review articles2–4 and most recently a textbook that has chapters devoted to immunoaffinity and other bioanalytical techniques.5
In an earlier article, immunoaffinity products that were designed to remove high-abundance proteins from biological samples were covered.6 In principle, these products are similar to those discussed here but are optimized for the immunoaffinity interactions for large molecules. In addition, an earlier article discussed molecular imprinted polymers (MIPS),7 that, in principle, mimic antibodies but involve the formation of a specific molecular imprint (cavity) on the surface of the sorbent formed by a template molecule while ISs are prepared by the immobilization of an antibody onto the surface of a solid support.
What Are ISs and How Are They Made?
In the medical sense, an antibody is an immunoglobulin, a specialized immune protein, produced by the body's immune system because of the introduction of a foreign agent called an antigen. The antibody possesses the remarkable ability to combine with the very antigen that triggered its production. The first step in the production of an IS is to design an antibody with the capability of molecular recognition of one analyte or for a group of analytes. However, compounds of low molecular weight such as environmental pollutants or pesticides are unable to evoke an immune response so they have to be modified by binding them to a large carrier molecule, such as bovine serum albumin (BSA). In order for the target analyte to couple to the carrier, sometimes a functional group must be introduced into its structure without affecting the analyte identity. When this functional group is introduced, the substance is referred to as a hapten, which itself does not induce antibody formation but is able to combine with a specific antibody. A mammal such as a rabbit or mouse is infected with this hapten carrier conjugate and over the course of time, its immune system produces a number of different IgG molecules directed against all foreign parts of the hapten carrier conjugate molecules. Samples of the mammal's blood are collected over time to obtain antibodies produced against the antigen. The polyclonal antibodies that are produced are a heterogeneous mixture of antibodies that bind to various sites on the original conjugate with a range of strengths. For this reason, polyclonal antibodies are somewhat difficult to reproduce. After purification by chromatography and other means, the antibodies are immobilized on the surface of a solid support, producing an IS.
Sometimes a monoclonal antibody is used for the IS. A monoclonal antibody is mass-produced in the laboratory from a specific lymphocyte cell clone and it produces only one type of IgG. Therefore, it recognizes only one antigen so it is generally more selective than a polyclonal antibody. Monoclonal antibodies are more expensive to make than polyclonal antibodies but once made, allows the large-scale production of identical antibodies fairly inexpensively. Thus, in general, monoclonal antibodies are more reproducible than polyclonal antibodies.
The antibodies can be covalently bonded to the surface, noncovalently bonded or adsorbed onto the surface, or can be encapsulated into the structure of the solid support. When bonded via amine groups, antibodies are linked by attaching reactive groups such as epoxide or aldehyde groups to the support or via surfaces activating agents such as cyanogen bromide or N-hydroxysuccinimide. Antibodies also can be linked via their carbohydrate groups located in the Fc region of the heavy chain.
Sorbents used as substrates should be inert, have hydrophilic surfaces, should be easily activated, be mechanically stable and have pores large enough to accommodate the antibody molecules that are in the 150 kDa range. They can be polymeric media such as agarose, cellulose, polystryene–divinylbenzene or polymethacrylate, or inorganic support such as silica gel or glass beads. Macroporous wide-pore silica gel makes an excellent medium for affinity columns. The high surface area, structural rigidity (pressure resistance), rapid mass transfer, variety of particle and pore sizes, and high purity are features that are conducive to making silica an ideal substrate. Of course, the silica surface must be treated appropriately so that nonspecific interactions are eliminated. A disadvantage of silica as an IS substrate is that its upper pH range is limited.
The production of an IS can be performed in the laboratory but most workers faced with solving a complex sample preparation problem for an environmental contaminant rarely have the time or the expertise to develop his or her own immunosorbent from scratch. Fortunately, commercial companies have responded to this need and ISs for specific analytes or analyte classes are available. Some of these products will be covered later.
How Are ISs Used?
Similar to SPE, ISs can be packed into disposable cartridges. They also can be packed into high performance liquid chromatography (HPLC) column hardware. The ISs can be used off-line, separately from the analysis experiment or can be incorporated into a flow system and used on-line. In the latter situation, the IS is connected in the flow steam before the HPLC or gas chromatography (GC) column and a valve is used to switch the effluent to waste or to a chromatographic column. The on-line approach is particularly attractive when additional sample preparation or chromatographic separation steps are required for ISs that extract analyte classes such as pharmaceuticals and their related metabolites. Precision is generally improved compared to the off-line approach. Some antibodies release the antigen very slowly so some form of reconcentration (for example, reversed-phase partition mechanism) might be required after the immunoextraction experiment.
In the use of ISs, there are three basic steps in the analyte (antigen) isolation process (see Figure 1). Step A shows a simple cartoon of a cartridge packed with an IS. Of course, the IS would be a particle rather than being bonded to the cartridge as the picture shows. The liquid sample containing the analyte antigen in a mixture of interfering compounds is loaded onto the cartridge (step B). By choosing the appropriate buffer and mobile-phase loading conditions, often in the area of physiological pH (pH 7.0–7.4), the antigen tightly binds to the antibody on the packing surface while the interferences are floating freely in solution. In reality, some of the interferences can bind to the sorbent by other mechanisms.
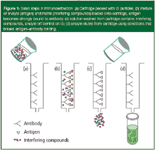
Figure 1
In step C, the particles are washed from the cartridge with additional buffer, leaving behind the analyte still tightly bound to the IS. In step D, the antigen is eluted from the cartridge by adjusting the eluting conditions to break the strong antibody–antigen binding that often involves a combination of electrostatic, H-bonding, van der Waals and other forces. The eluting solvent must be chosen to induce release of the antigen and involves using one's knowledge of chemistry in ensuring that these forces are overcome so that the analyte can be eluted in the smallest volume possible. Conditions such as the use of chaotropic compounds and other displacers (a compound that induces a stronger biospecific interaction with the antibody), pH changes, addition of organic solvent mixtures to break hydrophobic interactions, salt concentration and temperature changes can be used to accomplish this goal. Care must be exercised not to change conditions too dramatically such that the immobilized antibody is affected. After the step D, the IS must be regenerated so that it can be reused for another sample. Regeneration of the antibody-based ligand is accomplished by adding the initial application buffer to the cartridge.
Fortunately, for the commercial speciality IS products that are designed for specific small molecular weight analytes or analyte classes, the loading, elution and regeneration conditions are already worked out, making their applications more foolproof.
What Are Some Important Considerations in Using an IS?
The ISs are based upon molecular recognition using antibodies. The combination of high affinity and high selectivity of the antigen–antibody interaction, a high degree of molecular selectivity is displayed. The mechanism of affinity separations is sometimes described by a "lock and key" analogy. The retention of the ligand molecule (the key) to the specific binding site (the lock) is governed by a high affinity between the two — a perfect fit (Figure 2). Sometimes extraction, concentration and clean-up are achieved in a single step and other times, multiple sample prep techniques can be required for complete isolation of the analyte of interest.

Figure 2
Typical parameters that govern separation and retention in SPE and HPLC such as the effect of particle size and flow-rate on mass transfer, breakthrough volume, sample capacity, pressure stability and cycle time are equally important in affinity separations.
There are a number of important IS properties for their application that are somewhat unique. First, the binding capacity should be sufficient to bind all of the antigen analyte that is in the sample. The antibody binding density of the IS is related directly to the antigen binding capacity. The particular substrate used to immobilize the antibody has a direct influence on the binding density but there must be a balance between the surface area (dictates the number of binding sites) and the pore size (permits the high molecular weight antibodies to be loaded onto the sorbent). The bonding density is also related to the purity of the IgG antibodies bound to the sorbent. The higher the purity, the more antibody available for antigen binding.
Another important property is the structural availability of the immobilized active antibodies to interact with the target analyte (antigen). The antigen binding sites must be oriented from the surface without steric hindrance so that there is maximum contact with the antigen. This orientation is particularly important for the polyclonal antibodies that have lower numbers of target specific antibodies. The orientation of the antibodies can be controlled somewhat by the type of sorbent and antibody immobilization procedures that are used.
Just as in SPE, the analyte recovery is another important parameter in the application on an immunosorbent, especially when trace analyte isolation is desired. The recovery represents the fraction of desired analyte injected into the cartridge that can be eluted successfully after the binding experiment. Ideally, there should be 100% recovery but incomplete binding upon loading the sample or incomplete elution of strongly held antigen will reduce observed recovery. Nonspecific interactions with active sites on the substrate can affect antigen recovery.
The cross-reactivity of the immobilized antibodies is an important parameter in their application. The cross-reactivity is the ability of an antibody to react with similar antigenic sites on different molecules. Ideally, an antibody would be specific for a single target analyte. But, in reality, molecules with similar structures can interact with a given antibody. Thus, the IS can be an ideal selective phase for a class of compounds or for a target analyte and its metabolites because many of the latter might possess similar functionalities.
Another important consideration in the application of an IS is the sample volume that can be passed through the IS bed before there is breakthrough of analyte during the loading step. Of course, this volume is related to binding capacity that can potentially be affected by matrix components. The higher the affinity for the analyte, the greater the volume of sample that can be percolated through the bed. Breakthrough will represent a loss of analyte and low recovery. During method development, it is advisable to determine the breakthrough capacity, particularly if one is going to pass large volumes of water or other liquid samples through the IS. Just as in SPE, the elution step should use the smallest possible volume of solvent so as to concentrate the analyte of interest. If a large volume of elution solvent is required, another concentration step might be required, taking more time and inducing possible losses in recovery.
Regeneration is virtually a requirement of an IS. The cost of producing an antibody and bonding it to a substrate makes a typical IS a great deal more expensive than a typical SPE cartridge. Therefore, it is highly desirable to be able to regenerate the antibody multiple times after using it for an extraction. There are many factors that influence the number of times that an IS can be regenerated. Exposure to high percentages of organic solvent are not recommended as the life of an IS will be affected.
Applications of IS
For over a decade, there have been many studies of immunoextraction applied to a wide variety of analytes primarily in the environmental, biopharmaceutical and food areas, many by academic institutions. The technique has been found to be a selective, simple extraction sample preparation method. Immunoextraction can eliminate multiple extraction and derivatization steps that might be required by the more conventional sample preparation methods. The on-line application has been shown to be quite useful with precision well within that of other sample preparation procedures (1–5% relative standard deviations).8
To illustrate the variety of analytes and matrices that have been investigated using immunoaffinity extraction as the sample preparation method, Table 1 provides some typical examples. For additional applications, readers can cite the review articles mentioned earlier.2–5

Table 1: Typical examples of the use of immunoextraction for sample preparation.
To illustrate one example of the practical use of an IS, I have chosen to show a food application, the determination of fluoroquinolones in chicken liver.10 The fluoroquinolones are antibiotics that are used in both humans and animals to treat bacterial infections. There has been some concern about using these antibiotics in food animals such as chickens because by constant human consumption of these antibiotics can allow bacteria to develop resistance to them.
Conventional methods to measure these antibiotics in meat products require extensive sample clean-up procedures. Early immunoassay procedures for fluoroquinolones showed significant cross-reactivity. To circumvent these problems, immunosorbents were used to capture residues of interest and by adjusting the binding and elution conditions, the authors were able to use a reversed-phase column with a switching valve to selectively analyse these antibiotics in chicken liver on-line.
To synthesize the IS, the authors used an anti-sarafloxacin monoclonal antibody (Sara-95) that was developed in their own laboratory. For preparation of the immunoaffinity column, the purified Sara-95 was covalently bonded to a Protein G POROS medium (Applied Biosystems, Foster City, California, USA) and then packed into a 30 mm × 2.1 mm PEEK cartridge. For sample preparation, the fluoroquinolone-free chicken livers were fortified with the four main fluoroquinolone antibiotics (ciprofloxacin, enrofloxacin, satafloxacin and difloxacin, see structures in Figure 3) at concentrations of 20, 50 or 100 ppb (ng/g). After performing some wet chemistry including addition of a small amount (5%) of methanol to provide effective extraction and minimal nonspecific binding to the IS, the final injection volume of 100 μL represented about 10 mg of tissue and 0.2, 0.5 and 1.0 ng/mL of each fluoroquinolone.
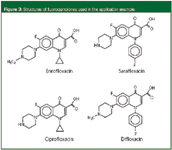
Figure 3
To capture the fluoroquinolone antibiotics, the mobile phase was a phosphate buffered saline (PBS, 100 mM sodium phosphate, 150 mM sodium chloride, pH 7.0) solution. The Sara-95 showed cross reactivity to all of the fluoroquinolone and, thus, retained all of them under this condition. After loading, the column was washed with 10 column volumes of PBS solution flushing the matrix components to waste followed by 0.1 M sodium dihydrogen phosphate buffer (pH 6.0) to lower the salt concentration. Then, the valve was switched to the reversed-phase column and the fluoroquinolones were eluted from the IS using 2% acetic acid, pH 2.2. The antibiotics were then separated isocratically (85:15 elution buffer–acetonitrile) using fluorescence detection. Before this operation, the immunoaffinity column was reequilibrated with PBS solution and was ready for the next sample.

Figure 4
Figure 4 shows the timing sequence of the previous operation and the separation of the standard mixture on the tandem column set-up. Peaks A (enrofloxacin and ciprofloxacin) and B (sarafloxacin and difloxacin) actually represent a separation of some of the fluoroquinolones on the IAC column because their relative affinities for the Sara-95 differ. Figure 5 depicts the chromatograms obtained for a liver blank and a liver extract fortified with 50 mg/g of each fluoroquinolone. Most of the liver matrix components were eluted immediately after injection. Although these liver matrix components did build up on the reversed-phase column and cause an increase in pressure, they did not interfere with the fluoroquinolone assay. The column was washed periodically and an in-line filter was installed. The IAC column was used over the course of a year for hundreds of samples and it continued to perform.

Figure 5
The average recovery of fluoroquinolones from fortified chicken liver samples at the 20–100 ng/g levels was over 85% with standard deviations in the 1–4% range. The limits of detection based upon a signal to noise of 5:1 ranged from 0.3 to 0.9 ng/g in liver. Sample volumes of up to 2000 μL could be used to increase the sensitivity even further.
This study is typical of many published results and shows that IS extraction is fairly easy to perform, saves time and manual labour and provides very selective extractions. Some development work to ensure maximum analyte capture–elution and minimal matrix capture–elution is required for a robust method.
Commercial Small Molecule IS Products
Because most chromatographers who need selective sample preparation procedures do not have the expertise, the desire, nor the time to devote to the development of an IS for their application, they turn to commercial companies to develop products that can be purchased. In the last few years, a number of companies who have developed immunoassay kits have turned to providing IS products for those analytes deemed to have a high enough volume to commercialize. Table 2 lists some typical products, analytes and suppliers who have IS columns or cartridges in their offerings. This list might not be all-inclusive but serves to depict typical products in this area. The main focus of these devices has been the area of mycotoxins in food products. Obviously, mycotoxins are of great interest to many of us because these toxins can be present in everyday foods (for example, nuts, grains, milk and meat).
Table 2: Typical commercial IS products.
To illustrate the use of one of these commercial products, I have chosen the isolation and determination of aflatoxins in coconut waste using the Venture AF Immunoaffinity column (Grace Vydac, Baltimore, Maryland, USA). Aflatoxins are highly-toxic natural substances and suspected carcinogens produced by mold that can be found on agricultural products such as grains, corn, peanuts and seeds. There are four aflatoxins that are commonly analysed: B1, B2, G1 and G2, each with different structures. The common analysis procedure is to perform LLE, SPE or column chromatography followed by HPLC analysis with fluorescence detection. Recently, a more selective straightforward clean-up was introduced using off-line immunoaffinity chromatography.24 Figure 6 depicts on-line immunoaffinity clean-up followed by reversed-phase chromatography and fluorescence detection. The Venture AF column contains an antiaflatoxin antibody immobilized on wide-pore silica and is selective for the compound class.

Figure 6
For sample preparation, a 25 g sample of coconut fibre was shaken with 5 g of sodium chloride and 100 mL of 80% (v/v) acetonitrile in water. A 1 mL volume of this extract was diluted 10-fold with 9 mL of PBS buffer consisting of 0.1 M phosphate plus 0.15 M sodium chloride, pH 7.0. A sample of the extract was injected directly onto the immunoaffinity column resulting in the chromatogram of Figure 6. There are a number of matrix peaks at the beginning of the chromatogram but the aflatoxin compounds are cleanly separated from the interferences. The limit of detection of the method was determined to be 2–4 pg (0.2–0.4 μg/kg depending upon the aflatoxin).
There are also other products for the immunoextraction of small molecules in addition to mycotoxins. For example, products for the isolation of food nutrients, endocrine disruptors, pesticides and steroid hormones are available. These products not only provide the cartridges themselves but also the detailed methodology for use for a variety of matrices.
Conclusion
Immunoaffinity extraction has been shown to be an effective sample preparation method for trace analysis. The technique is selective, simple to use and reduces the number of steps involved in sample preparation. The preparation of a suitable IS does involve specialized technology, so it has not received as much attention as techniques such as SPE. Most laboratories do not have the expertise, the desire nor the time to generate their own ISs. However, for high-volume assays for trace analysis in complex matrices such as those encountered in environmental and food safety applications, commercial products are being developed. As more of these commercial products become available, the use of immunoaffinity extraction will continue to grow.
Ronald E. Majors is a senior chemist, Columns and Supplies Division, Life Sciences and Chemical Analysis Group, Agilent Technologies, Wilmington, Delaware, USA and is a member of the Editorial Advisory Board of LCGC Europe. Direct correspondence about this column to LCGC Europe, Advanstar House, Park West, Sealand Road, Chester CH1 4RN, UK, e-mail: dhills@advanstar.com
References
1. R.E. Majors, LCGC Eur., 16(2), 71–81 (2003).
2. V. Pichon et al., TrAC Trends in Anal. Chem., 18(3), 219–235 (1999).
3. D. Stevenson, J. Chromatogr. B, 745(1), 39–48 (2000).
4. N. Dalaunay, V. Pichon and M.-C. Hennion, J. Chromatogr. B, 745(1) 15–37 (2000).
5. Immunoassay and Other Bioanalytical Techniques, J.M. Van Emon, Ed. (CRC Press, Boca Raton, Florida, USA, 2007).
6. N. Zolotarjova, P. Mrozinjski and R.E. Majors, LCGC N. Am., 25(2), 118–140 (2007).
7. K. Ensing, C. Berggren and R.E. Majors, LCGC Eur., 15(1), 16–25 (2002).
8. A. Moser, M.A. Nelson and D.S. Hage, "Environmental Applications of Immunoaffinity Chromatography" in Immunoassay and Other Bioanalytical Techniques, J.M. Van Emon, Ed. (CRC Press, Boca Raton, Florida, 2007), p. 341.
9. J.C. Chuang et al., Anal. Chim. Acta, 583, 32–39 (2007).
10. C.K. Holtzapple, S.A. Buckley and L.H. Stanker, J. Agric. Food Chem., 47, 2963–2968 (1999).
11. P.A. Burdaspal, T.M. Legarda and J. Gibert, J. AOAC-Int., 84(5), 1445–1452 (2001).
12. P.B. Carrasco et al., J. Chromatogr. A., 909(1), 61–72 (2001).
13. A. Visconti, M. Solfrizzo and A. de-Girolamo, J.A.O AC-Int., 84(6), 1828–1837 (2001).
14. M.M. Rhemerv-Boom et al., J. Pharm. Biomed. Anal., 24(5–6), 825–833 (2001).
15. P.L. Ferguson et al., Anal. Chem., 73(16), 3890–3895 (2001).
16. S.D. Thomas and Q.X. Li, Environ-Sci-Technol., 34(12), 2649–2654 (2000).
17. A.Farjam et al., Int. J. Environ-Anal. Chem., 44(3), 175–184 (1991).
18. R. Schuhmacher et al., J. Anal. Chem., 359(6), 510–515 (1997).
19. M.A. Nelson et al., Anal. Chem., 76(3), 805–813 (2004).
20. G.A.Grant, S.L. Frison and P. Sporns, J. Agric. Food Chem., 51(18), 5367–5375 (2003).
21. V. Pichon, N. Delaunay and M.C. Hennion, Spectra-Anal., 28(211), 13–18 (1999).
22. N. Delaunay et al., Anal. Chim. Acta., 407(1–2), 173–186 (2000).
23. M. Bouzige et al., J. Chromatogr. A., 846(1–2), 317–329 (1999).
Thermodynamic Insights into Organic Solvent Extraction for Chemical Analysis of Medical Devices
April 16th 2025A new study, published by a researcher from Chemical Characterization Solutions in Minnesota, explored a new approach for sample preparation for the chemical characterization of medical devices.
Sorbonne Researchers Develop Miniaturized GC Detector for VOC Analysis
April 16th 2025A team of scientists from the Paris university developed and optimized MAVERIC, a miniaturized and autonomous gas chromatography (GC) system coupled to a nano-gravimetric detector (NGD) based on a NEMS (nano-electromechanical-system) resonator.
Common Challenges in Nitrosamine Analysis: An LCGC International Peer Exchange
April 15th 2025A recent roundtable discussion featuring Aloka Srinivasan of Raaha, Mayank Bhanti of the United States Pharmacopeia (USP), and Amber Burch of Purisys discussed the challenges surrounding nitrosamine analysis in pharmaceuticals.
Extracting Estrogenic Hormones Using Rotating Disk and Modified Clays
April 14th 2025University of Caldas and University of Chile researchers extracted estrogenic hormones from wastewater samples using rotating disk sorption extraction. After extraction, the concentrated analytes were measured using liquid chromatography coupled with photodiode array detection (HPLC-PDA).
Silvia Radenkovic on Building Connections in the Scientific Community
April 11th 2025In the second part of our conversation with Silvia Radenkovic, she shares insights into her involvement in scientific organizations and offers advice for young scientists looking to engage more in scientific organizations.











