Tosoh Bioscience – Characterization of Monoclonal Antibodies Using Native SEC–MS and Its Comparison to Denaturing RP–MS Technique
Therapeutic antibody technology currently dominates the biologics market, and the last decade has seen drugs based on monoclonal antibodies (mAbs) progressively overtake their small-molecule alternatives. mAbs have shown remarkable efficacy to treat a plethora of indications, including cancers, infections, autoimmune disorders, and cardiovascular and neurological diseases. Because the whole antibody therapeutics platform is regarded as one of the most promising classes of pharmaceutical technology to date, there is growing interest in developing integrated analytical methods to provide layered information on the structure, purity, and stability of mAbs.
The rise of proteomics has generated remarkable technological advances in mass spectrometry (MS), especially in the field of high-resolution MS (HRMS), leading to an increased impact of MS on the field of structural biology. Electrospray ionization (ESI) has proven to be a very useful technique for obtaining multiply charged ions of intact proteins, providing the biopharmaceutical industry with a unique opportunity to characterize and control mAb quality during clinical development and commercial production. U/HPLC methods that allow hyphenation with MS are of particular interest for this purpose, providing users with an additional dimension in their mAb characterization (molecular weight determination and structural information). Although direct infusion in the mass spectrometer remains the most straightforward approach to conduct intact mAb characterization, it is typically hampered by the various contaminants (such as salts, stabilizers or detergents) present in the matrix of recovered and purified mAbs. Sample preparation before MS analysis is therefore essential to ensure accurate mass and structural characterization.
With the use of MS-compatible eluents, reversed-phase (RP) chromatography is particularly well suited for protein desalting prior to MS analysis. Using a gradient from low to high organic content, the protein binds to the column while MS-interfering contaminants are eluted. Additional benefits to RP desalting include the increased mobile-phase volatility at the source for an improved ionization process. Nevertheless, the use of organic solvent in the mobile phase causes denaturation of the protein, which results in increased charge (z) values and often complicates MS data interpretation. In addition, mAb analysis by RP requires high temperatures (usually >50 °C) to reduce the strength of secondary interactions between mAb and the stationary phase, which can result in unwanted on-column fragments generation or loss of specific sugars from the glycan moieties.
Size-exclusion chromatography (SEC) remains the gold standard for determining the molecular weight (MW) distribution of mAbs expressed in mammalian cell culture. Obtaining structural information beyond the physical size (hydrodynamic volume) typically requires the combination of SEC with MS. Native ESI has proven particularly useful for generating multiply charged ions of intact proteins with lowered charge states, providing increased spectral resolution at higher m/z values. Still, the analysis remains challenging and involves biomolecule-specific optimization on both the chromatography and mass spectrometry side.
This application note describes the effective use of MS- compatible mobile-phase compositions in the analysis of mAbs using both RP (denaturing ESI) and SEC (native ESI) modes with in-line native ESI-MS detection on a hybrid Q-TOF instrument.


Results and Discussion
Denaturing vs. Native ESI-MS Analysis
The main difference between traditional denaturing and native ESI–MS is the composition and pH of the employed mobile phases (see Experimental Conditions). Denaturing solutions, containing organic solvents and non-neutral pH conditions, produce unfolded proteins by disrupting noncovalent interactions, causing molecules to unfold and expose sites of protonation. Conversely, native solutions at neutral pH (~7) preserve noncovalent interactions, as these molecules transition to the gas phase. Charge state and intensity during electrospray ionization are mainly determined by the number of ionizable sites. ESI–MS of denatured species typically produces a Gaussian distribution of highly charged ions in a wider charge state distribution, while native ESI produces ions with lower charge states and a narrower charge state distribution. This is largely due to the globular nature of folded proteins, limiting solvent accessible residues to only those on the surface of the protein. This is illustrated in Figure 1, which depicts the charge state envelopes obtained for the NIST mAb in both denaturing (RP-MS) (Figure 1B) and native conditions (SEC-MS) (Figure 1E).
Figure 1: Examples of the chromatographic elution profiles (A and D), the charge state envelopes (B and E), and the deconvoluted spectra (C and F) obtained under denaturing (A–C) and native (D-F) intact mass conditions for the analysis of the NIST mAb.
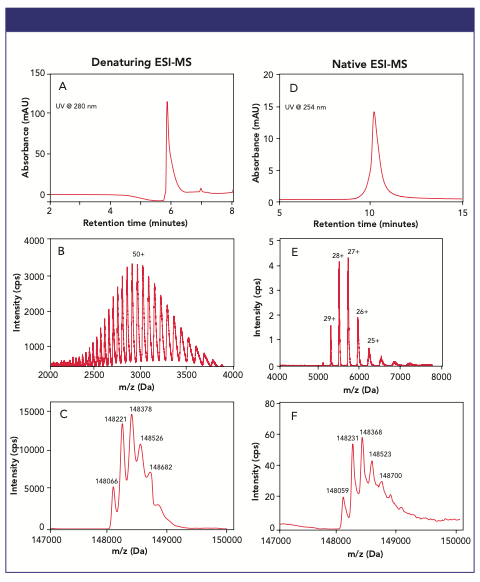
Both RP (on a TSKgel Protein C4-300 column) and SEC (on a TSKgel UP-SW3000 column) analysis of the ~150 kDa NIST mAb coupled to a HRMS QTOF mass spectrometer led to robust separation and detection of the mAb. The main mAb RP peak eluting at 6 min (Figure 1A) exhibits a mass spectrum (Figure 1B) made of a charge distribution ranging from ~40+ to ~60+, with the Gaussian distribution representative of its unfolded form centered around m/z ~2900. In contrast, the main mAb SEC peak eluting at 10.5 min (Figure 1D) exhibits a less complex mass spectrum made of a charge distribution ranging from 24+ to 30+, with the Gaussian distribution representative of its native, folded form centered around m/z ~5300 (Figure 1E). Intact protein mass values—also called the zero-charge state—from these pseudo-molecular ions can be determined using deconvolution of protein charge states. MS deconvolution results for the denatured and native NIST mAb are presented in Figures 1C and F, respectively, exhibiting low mass errors (average: <10 Da) and achievement of MS resolution for the major glycoforms of the NIST mAb.
Buffer Consideration for Intact Protein Analysis by LC–MS
Making your liquid chromatography (LC) method compatible with MS is a crucial first step in your method development or transfer. When using MS detection, your analytes first must be “transferred” from the liquid phase into the gas phase, which is quite different from most optical detectors classically hyphenated with LC (for example, UV and fluorescence detectors) where your sample is maintained in the liquid phase. Hyphenation with MS involves the process of ionization, which occurs in the ion source of your MS equipment, making your mobile phase (and all its additives) a crucial part of a successful method development and/or transfer.
RP is easily amenable to MS compatibility, with the focus being on the acidifier additive. Traditional RP-UV methods have used trifluoroacetic acid (TFA), which needs to be replaced by a volatile acidifier such as formic acid (FA) or difluoroacetic acid (DFA). In the case of SEC, although they are best suited to maintain the native folded structure of your mAb of interest, phosphate-based buffers cannot be used as they will crystallize in the ion source, suppress ionization, and ultimately contaminate your ion source. Switching to volatile buffers (such as ammonium acetate, ammonium formate or ammonium bicarbonate) is required. When doing so, considering your mobile phase from both the SEC and the MS point of view is crucial, as both LC performance (retention times, peak shape, resolution) and MS performance (ionization efficiency, maintenance of proteins’ native structures) can be affected by buffer type and buffer concentration.
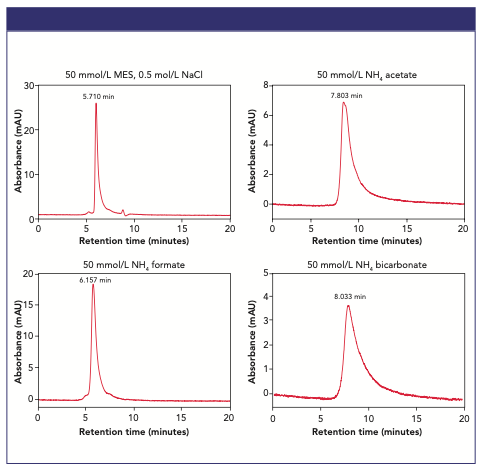
The effects of MS-compatible volatile SEC buffers on mAb elution profiles are illustrated in Figure 2, where the separation of Humira® biosimilar on the TSKgel UP-SW3000 column was conducted using four different buffers at 50 mmol/L concentration. A sharp monomer peak at 5.7 min was observed when using a non-volatile buffer (MES) containing 150 mmol/L NaCl. When testing three volatile, MS compatible buffers at 50 mmol/L, three different peak shapes and retention times were observed, with 50 mmol/L ammonium formate offering the best chromatographic performance.
Figure 2: SEC elution profiles of Humira biosimilar analyzed on TSKgel UP-SW3000 using four different mobile phase compositions.
Experimental Considerations for a Successful Intact mAb Characterization by LC–ESI–MS
Below are the three main steps of how to optimize a successful intact mAb analysis using LC–MS:
- Consider the various aspects of chromatographic separation and how to make them compatible with MS analysis.
- Electrospray ionization: Identify the various parameters that can influence ionization efficiency and desolvation when you hyphenate LC with MS.
- The MS detection and analysis: What parameters to consider on your MS instrument.
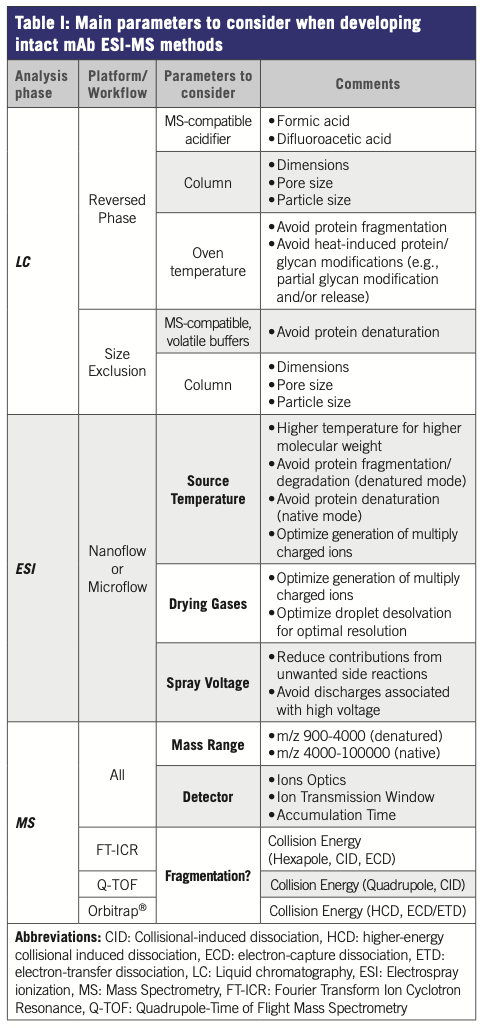
Table I presents a non-exhaustive summary of the various parameters and variables to consider when developing intact mAb ESI–MS experimental workflow, including MS platform-specific factors.
Conclusions
Monoclonal antibodies are highly complex biomolecules, requiring high resolution, precision, and dynamic range to fully characterize them with confidence. This application note illustrates an intact mAb analysis workflow solution integrating U/HPLC technologies, high-resolution mass spectrometry on a QTOF instrument and software for automatic data processing. The workflow permits rapid and accurate mass characterization of mAbs, using either denaturing ESI using reversed- phase chromatography or native ESI using size-exclusion chromatography, leading to excellent mass accuracy for glycoform distribution. Detailed information was obtained about the heterogeneous composition of mAb proteins, with minimal sample preparation involved.
TSKgel and Tosoh Bioscience are registered trademarks of Tosoh Corporation Nexera is a registered trademark of Shimadzu Corporation
Humira is a registered trademark of AbbVie Biotechnology Ltd.
Orbitrap is a registered trademark of Thermo Finnigan, LLC

Tosoh Bioscience LLC
3604 Horizon Drive, Suite 100, King of Prussia, PA 19406
Tel. (484) 805-1219, fax (484) 805-1277
Website: www.tosohbioscience.com
Free Poster: NDSRI Risk Assessment and Trace-Level Analysis of N-Nitrosamines
April 25th 2025With increasing concern over genotoxic nitrosamine contaminants, regulatory bodies like the FDA and EMA have introduced strict guidelines following several high-profile drug recalls. This poster showcases a case study where LGC and Waters developed a UPLC/MS/MS method for quantifying trace levels of N-nitroso-sertraline in sertraline using Waters mass spectrometry and LGC reference standards.
New TRC Facility Accelerates Innovation and Delivery
April 25th 2025We’ve expanded our capabilities with a state-of-the-art, 200,000 sq ft TRC facility in Toronto, completed in 2024 and staffed by over 100 PhD- and MSc-level scientists. This investment enables the development of more innovative compounds, a broader catalogue and custom offering, and streamlined operations for faster delivery. • Our extensive range of over 100,000 high-quality research chemicals—including APIs, metabolites, and impurities in both native and stable isotope-labelled forms—provides essential tools for uncovering molecular disease mechanisms and exploring new opportunities for therapeutic intervention.
New Guide: Characterising Impurity Standards – What Defines “Good Enough?”
April 25th 2025Impurity reference standards (IRSs) are essential for accurately identifying and quantifying impurities in pharmaceutical development and manufacturing. Yet, with limited regulatory guidance on how much characterisation is truly required for different applications, selecting the right standard can be challenging. To help, LGC has developed a new interactive multimedia guide, packed with expert insights to support your decision-making and give you greater confidence when choosing the right IRS for your specific needs.
Using the Carcinogenic Potency Categorisation Approach (CPCA) to Classify N-nitrosamine Impurities
April 25th 2025Learn how to manage nitrosamine impurities in pharmaceuticals with our free infographic. Discover how the CPCA approach establishes acceptable intake limits and guides the selection of NDSRI reference samples. Stay compliant and ensure safety with our ISO-accredited standards.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)