Quantitative Multi-Target Screening Using Library-Based Identification
This article presents a novel approach for toxicology and pharmacology laboratories that combines multi-compound screening followed by multi-compound determination in a single method. The multi-compound assay developed allows 90 molecules including benzodiazepines, cocaine and related stimulants, amphetamines, and opioids to be measured. The multi-targeted screening (MTS) method was developed using a spectra library containing over 1200 compounds. The approach was evaluated in a routine clinical toxicology laboratory to detect and quantify compounds in unknown samples.
Photo Credit: CA-SSIS/Shutterstock.com

Tiphaine Robin1,2, 1Shimadzu Corporation, Marne-la-Vallée, France, 2Department of Pharmacology and Toxicology, Limoges University Hospital, France
This article presents a novel approach for toxicology and pharmacology laboratories that combines multi-compound screening followed by multi-compound determination in a single method. The multi-compound assay developed allows 90 molecules including benzodiazepines, cocaine and related stimulants, amphetamines, and opioids to be measured. The multi-targeted screening (MTS) method was developed using a spectra library containing over 1200 compounds. The approach was evaluated in a routine clinical toxicology laboratory to detect and quantify compounds in unknown samples.
Screening has been the first analysis to be performed in toxicology when the nature or the presence of a drug is totally unknown, which is often the case in clinical and forensic toxicology (1). Usually, a screening precedes more specific analyses, allowing the quantitation of detected compounds. It is now possible to combine both tasks and to benefit from methods that allow both identification and quantitation.
In addition to reducing the possibility of false defect reporting without compromising accuracy, precision, or limits of detection, the actual liquid chromatography tandem mass spectrometry (LC–MS/MS) instruments could combine the sensitivity of multiple reaction monitoring (MRM) detection with the identification power of a full scan product ion spectrum.
Simple and rapid preparation procedures are also necessary. In this article, a generic sample preparation method based on QuEChERS salts was optimized. QuEChERS (an acronym for “quick, easy, cheap, effective, rugged, and safe”) are notably used in food safety and pesticide analysis. However, a few studies have used this approach in clinical toxicology (2,3,4,5).
The global strategy of this study was: i) to build an exhaustive spectra library of the most pertinent compounds; ii) to develop a generic sample preparation method based on QuEChERS salts; and iii) to test the identification and quantitation performance of an MS acquisition mode combining threshold triggered product ion scan and MRM detection.
Experimental
The chromatographic system consisted of two Shimadzu LC-30 AD pumps (Nexera X2), a CTO 20AC oven, and a SIL-30 AC-MP autosampler (all Shimadzu). Chromatographic separation was performed using a 100 × 2.1 mm, 2.7âµm Raptor biphenyl column (Restek). A Shimadzu LCMS-8060 triple quadrupole mass spectrometer was used.
Spectra Library: Each library spectrum was acquired by authentic standard flow injection at collision energies of 10–55 V. Compounds that ionized efficiently in more than one adduct state were saved resulting in 1507 library entries from 1207 compounds (1278 positive mode, 229 negative mode). Spectral library information was registered for three collision energies corresponding to CE ±10, ±35, and ±55 V. Optimized MRM transitions were also determined for all compounds together with retention time and peak area data ratio of response between quantifier and qualifier transitions. Retention time (RT) analysis included internal standard compounds for relative RT calculation. Compound metadata included: structure, retention time, CAS number, formula, synonyms, compound class or properties, ChemSpider URL and ID number, InChI, and InChIKey.
Sample Preparation Method: A 200âµL measure of acetonitrile was added to 100 µL of whole blood containing internal standard compounds. Samples were vortexed for ~5 s and then incubated at -20 °C for 10 min. A 40-mg measure of QuEChERS salts was added and samples were then vortexed for ~5 s. Samples were then centrifuged (16,000 g, 10 min) and 50-µL of supernatant was removed and added to 150-µL mobile phase A.
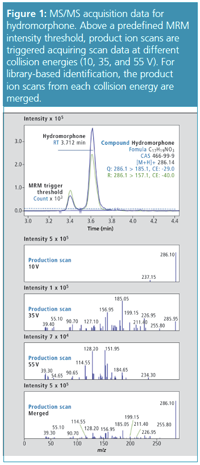
MS Acquisition: Two predefined MRM transitions for each compound were monitored. Once the MRM signals were above a set threshold intensity of 10,000 counts, a series of alternating product ion scans were acquired at three collision energies (10, 35, and 55 V) with an automatic exclusion for 3 s after two consecutive MS/MS scans. Product ion spectrum data from each collision energy were combined to create a fragment-rich single spectrum for library searching.
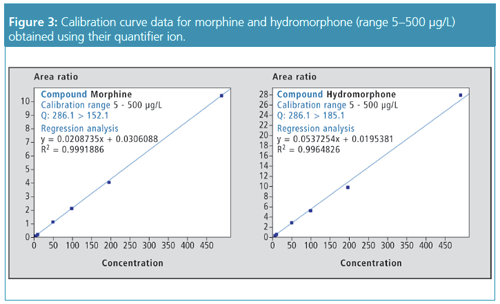
Applications of the Method: Whole blood samples were spiked with a panel of 90 commonly detected compounds reported in toxicological screening and normally measured in separate assays, namely, benzodiazepines, cocaine and related stimulants, amphetamines, and opioids; in addition, antipsychotic compounds were also screened. Calibration curves were prepared in the range 5 µg/L to 2000 µg/L for benzodiazepines, and 5 µg/L to 500 µg/L for cocaine and related stimulants, amphetamines, and opioids. The method performed well in identification of the 90 compounds, regardless of concentration levels: the similarity index (that is, similarity between the spectrum of a detected compound and its spectrum in the library) was above 75–80 in a majority of cases. After determining the concentration of the 90 compounds, performances for quantitation were recorded as accuracy in an 85–115% interval and r2 values above 0.99. Figures 2 and 3 illustrate these performances for two isobaric compounds: hydromorphone and morphine.
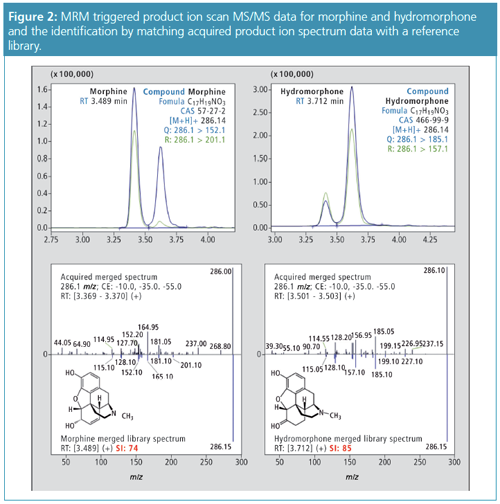
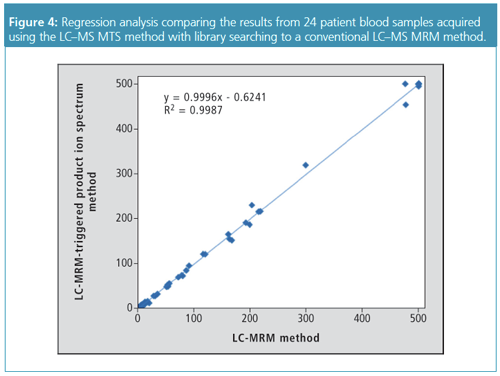
To test the viability of this approach and to quantify and identify targets in the different test panels, the MRM-triggered product ion spectrum acquisition method was applied to a series of patient blood samples and compared to a validated LC–MS/MS method using two MRMs for each target compound (2,3). The study concerned blood samples of 24 patients from a diverse range of contexts including overdoses, routine drug testing, driving under the influence of drugs (DUID), and samples provided by patients undergoing psychiatric treatment. For a number of patient samples more than one target compound was measured, resulting in 54 sample points in the regression analysis. Regression analysis showed a close agreement between both methods (slope = 0.9996; regression coefficient r2 > 0.99) (Figure 4).
Discussion and Conclusion
A method was developed for clinical toxicology and forensic analysis using QuEChERS sample preparation, a single LC analysis, and an MRM triggered product ion spectrum acquisition method. The method was designed to meet the needs of a routine clinical toxicology and forensic laboratory by delivering a single approach to the analysis of a diverse range of target panels, and also reducing false defect reporting.
There are two clear advantages of acquiring MRM-triggered product ion spectrum data: (i) a higher confidence in compound identification as a result of library searching, for example, in situations where reference ion ratios can be outside method tolerances or if concentrations measured are below the lower limit of quantitation (LLOQ); (ii) quantitative data is identical to a conventional 2 MRM method.
This was implemented successfully to enable screening and quantitation by MTS for a broadly targeted range of drugs of abuse included in a spectra library containing more than 1200 compounds of interest. Further studies are ongoing to test the capacity of the tool to target this number of compounds in one run.
References
- D. Remane, D.K. Wissenbach, and F.T. Peters, Clin. Biochem.49(13–14), 1051–71 (2016).
- S. Dulaurent, S. El Balkhi, L. Poncelet, J.M. Gaulier, P. Marquet, and F. Saint-Marcoux, Analytical and Bioanalytical Chemistry408(5), 1467–1474 (2016).
- L. Poncelet, S. El Bakhi, S. Dulaurent, and F. Saint-Marcoux, Toxicologie Analytique et Clinique28(3), 201–210 (2016).
- K. Usui, Y. Hayashizaki, M. Hashiyada, and M. Funayama, Leg. Med. 14, 286–96 (2012).
- S. Matsuta, K. Nakanishi, A. Miki, K. Zaitsu, N. Shima, T. Kamata, et al., Forensic Sci. Int.232, 40–5 (2013).
Tiphaine Robin obtained a State Doctorate in pharmacy in 2017 (University of Limoges, France). She is currently a PhD student in analytical chemistry. The main objective of her thesis is to develop innovative LC–MS screening procedures for applications in clinical and forensic toxicology. These works are under the supervision of both the Department of Pharmacology and Toxicology of the Limoges University Hospital (Franck Saint-Marcoux and Souleiman El Balkhi) and Shimadzu Corporation (Stephane Moreau and Alban Huteau, Shimadzu France, Marne-la-Vallée, France). The Mass Spectrometry Business Unit (Overseas) of Shimadzu Corporation, with Neil Loftus and Alan Barnes, is also greatly involved in these developments.
E-mail:tro@shimadzu.fr
Website:www.shimadzu.eu

Accelerating Monoclonal Antibody Quality Control: The Role of LC–MS in Upstream Bioprocessing
This study highlights the promising potential of LC–MS as a powerful tool for mAb quality control within the context of upstream processing.
Using GC-MS to Measure Improvement Efforts to TNT-Contaminated Soil
April 29th 2025Researchers developing a plant microbial consortium that can repair in-situ high concentration TNT (1434 mg/kg) contaminated soil, as well as overcome the limitations of previous studies that only focused on simulated pollution, used untargeted metabolone gas chromatography-mass spectrometry (GC-MS) to measure their success.
Prioritizing Non-Target Screening in LC–HRMS Environmental Sample Analysis
April 28th 2025When analyzing samples using liquid chromatography–high-resolution mass spectrometry, there are various ways the processes can be improved. Researchers created new methods for prioritizing these strategies.
Potential Obstacles in Chromatographic Analyses Distinguishing Marijuana from Hemp
April 28th 2025LCGC International's April series for National Cannabis Awareness Month concludes with a discussion with Walter B. Wilson from the National Institute of Standard and Technology’s (NIST’s) Chemical Sciences Division regarding recent research his team conducted investigating chromatographic interferences that can potentially inflate the levels of Δ9-THC in Cannabis sativa plant samples, and possible solutions to avoid this problem.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)













