The LCGC Blog
In this months technical tip we answer some often asked questions and dispel some myths regarding HPLC column abuse and reconditioning
In this months technical tip we answer some often asked questions and dispel some myths regarding HPLC column abuse and reconditioning. Please don’t forget that you can e-mail or call our technical team to get advice and recommendations on column problems - call 01357 522961 and ask for the technical team or e-mail [email protected]
Question 1 – Does air kill columns?
In the early days of HPLC, air in the column could be a problem because of the susceptibility of the silica particles to undergo ‘pneumatic mechanical shock’.
These days silica particles are much less susceptible. Also – it’s pretty difficult to introduce an appreciable amount of air into the column. Running the mobile phase dry usually results in a system under pressure and automatic shut-down. Columns should always be end-capped when not in use as if they are not, and are stored with an large amount of organic solvent, this solvent, over time, will evaporate. The secret here is to re-introduce the mobile phase slowly – flow rates of 0.1mL/min with an increasingly organic solvent composition usually works well. We typically recommend 20 column volumes – remember you can easily calculate the column volume using the equation below:
Vc = π x (diameter / 2)2 x length x 0.68
(keep all dimensions in mm and the result will be in μl)
Question 2 – You can’t reverse the direction of columns……right?
Wrong!
As a result of the column packing process, columns must have ‘directionality’ – that is, when the column is packed, the stationary phase particles at the column exit may be slightly more compressed than those at the entrance. However, column manufacturers work VERY hard to make sure the density of material across the column is as uniform as possible. Reversing column direction MAY lead to a slight settling in the reverse direction but often this doesn’t significantly impact on the column performance. In older columns, where a ‘void’ may have formed at the column head (usually because of chemical or mechanical issues) the reversed column may take a little time to ‘re-settle’ in the reverse direction, but usually one can get enough injections to see you through until your new column (from Crawford Scientific, of course) arrives. Reverse flushing of columns is often used to remove particulates from the frit on the top of the column – this is perfectly acceptable and usually a good way to clear the frit. Typically we recommend flowing at a low flow rate with relatively low viscosity solvent (80% MeCN (aq) is good) with the column decoupled from the detector, for 10 or more column volumes.
Symptoms of column voids / blocked frits include shouldered and split peaks.
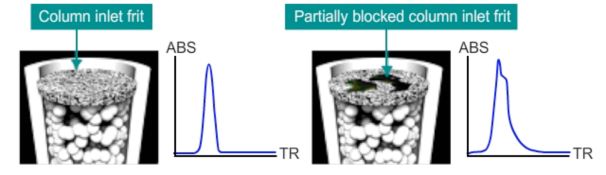
Question 3 – You can’t recondition a reversed-phase column which has been exposed to 100% water
Not true.
Although it’s really not to be recommended – running reversed-phase HPLC columns (especially C8 or C18 columns) doesn’t necessarily mean the end of the road for the column.
We find running the column in pure acetonitrile at high pressure and around 60oC will usually ‘displace’ the water from the inner pore structure of the particles in 50 column volumes.
Although it’s really not to be recommended – running reversed-phase HPLC columns (especially C8 or C18 columns) doesn’t necessarily mean the end of the road for the column.
We find running the column in pure acetonitrile at high pressure and around 60oC will usually ‘displace’ the water from the inner pore structure of the particles in 50 column volumes.
Question 4 – What’s the best storage solvent for a column?
This is somewhat dependent upon the column and it’s usage.
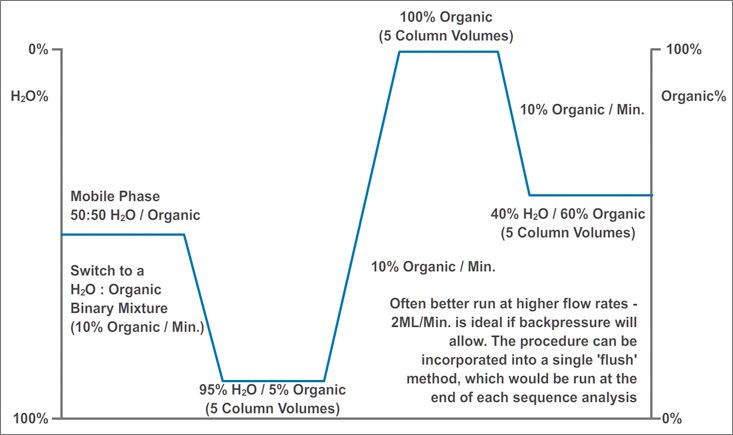
For reversed-phase columns used with standard buffers we run a gradient which is described below;
The first part of the gradient removes any buffers (the solid ones risk precipitation if the organic composition of the flushing solvent is ramped quickly) and the latter part ensures that the risk of microbial growth during storage is minimized.
We have these flushing gradients programmed into all of our instruments so it’s simply a matter of invoking the method at the end of a sequence and both the system and the column are ready for at least a weekend of inactivity. The column can then be removed and end caps fitted for linger term storage.
Columns used with trifluoroacetic acid (TFA) take a much longer time to re-equilibrate (TFA is an ion pairing reagent which will interact with the stationary phase) – it’s often better, for columns used with ‘official’ or unintentional ion pairing reagents to be dedicated to use with ion pairing reagents only and stored in the mobile phase including the ion pairing reagents but with >50% organic concentration.
Question 5 – I want to ‘regenerate’ a column, but don’t want to have the hassle of changing between multiple solvents – have you got a ‘quick fix’ easy regeneration method?
Phew – tough customer – but one who obviously lives in the real world.
Unfortunately, at least one exotic solvent will be necessary in order to remove components from the stationary phase that are not removed by typical reversed-phase organic solvents.
We typically use methylene chloride as the highly eluotropic solvent as it has good solubility with typical reversed-phase solvents. So our ‘quick fix’ protocol would be:
- 20 column volumes acetonitrile
- 20 column volumes methylene chloride
- 20 column volumes acetonitrile
- 20 column volumes mobile phase
For more information – contact either
Bev ([email protected]) or Colin ([email protected]).
For more tutorials on LC, GC, or MS, or to try a free LC or GC troubleshooting tool, please visit www.chromacademy.com
The LCGC Blog: Historical (Analytical) Chemistry Landmarks
November 1st 2024The American Chemical Society’s National Historic Chemical Landmarks program highlights sites and people that are important to the field of chemistry. How are analytical chemistry and separation science recognized within this program?


