The LCGC Blog: Potential for use of Stable Isotope–Labeled Internal Standards in Gas Chromatography–Vacuum Ultraviolet Spectroscopy Methods
Internal standards (IS) are commonly incorporated into quantitative methods to increase accuracy and precision. An IS is a compound that is different than the analyte of interest, has similar physicochemical properties to the analyte, and is added to samples, calibration standards, and quality control samples in a known quantity. It should not be present in the sample, it should be available in high purity, and it should be easily differentiable from the analyte of interest.
Internal standards (IS) are commonly incorporated into quantitative methods to increase accuracy and precision. An IS is a compound that is different than the analyte of interest, has similar physicochemical properties to the analyte, and is added to samples, calibration standards, and quality control samples in a known quantity. It should not be present in the sample, it should be available in high purity, and it should be easily differentiable from the analyte of interest.
As samples and standards are analyzed, the response of the analyte is normalized by the response of the known amount of IS. The IS is ideally subjected to the same discrimination processes throughout the method as the analyte; thus, by normalization, systematic errors are reduced. An IS should be chosen to properly ensure that all potential discrimination processes can be corrected. Random error is also generally reduced using an internal standard calibration method.
In mass spectrometry (MS)-based quantitative methods, the ideal IS a stable isotope–labeled version of the analyte. This is an analyte form where some or all of its stable (non-dissociable) hydrogens are switched to deuterium atoms, carbon-12 atoms are switched for carbon-13 atoms, or nitrogen-14 atoms are switched for nitrogen-15 atoms. With each one of these switches, a new compound is formed that has an incrementally higher molecular weight, but virtually the same physicochemical properties of the analyte. A stable isotope–labeled internal standard (SIL-IS), which is added to the sample before processing, will track together through the entire sample preparation and analysis process with the analyte, and then be differentially detected by the mass difference, evinced by MS.
Many gas chromatography–mass spectrometry (GC–MS) methods feature SIL-ISs. In our research using the vacuum ultraviolet detector for gas chromatography (GC–VUV) (1,2), it has been an unanswered question whether the use of SIL-IS methodology would be a viable thing to consider. Although GC–VUV can distinguish positional isomers (dimethylnaphthalenes) (3), constitutional isomers (designer drugs) (4), and stereoisomers (mono-, di-, and trisaccharides)(5) to a significant degree, it was unclear whether the addition of neutrons into atoms would affect significant changes to the electronic absorption spectra for the heavy forms of the molecules, relative to the natural form. We decided it was time to answer that question (6).
Before I discuss that research, I think it is important to address a couple of points. First, to largely evaluate the ability to distinguish two chemical compounds based on their gas-phase absorption profile in the 125–240 nm wavelength range, we can calculate a sum of squared residuals (SSR) value for any pair of spectra we are interested in comparing. The larger the SSR between them, the easier they are to distinguish from one another-the more dissimilar are the spectra.
If a pair of compounds with a high SSR between them (for example, SSR > 100) were coeluted chromatographically, then their individual contributions to the summed signal observed could still be easily determined (a process we call deconvolution), even if they were present at as much as three or more orders of magnitude difference in relative abundance (assuming similar absortivities and that the minor component was still above the detection limit). If two compounds were coeluted that have an SSR ~ 10 between them, then they could likely still be reliably deconvolved at a relative abundance of around 100:1. As spectral similarity increases (that is, SSR decreases), it becomes less possible to deconvolve two overlapping signals having different relative abundances. An SSR = 1 might accommodate a 10:1 difference. With an SSR < 0.1 it might be still possible to deconvolve two compounds overlapping in a 1:1 relative abundance, but not much more than that; additionally, at that level, if the spectra for two compounds are that similar, then a library search initiated from an experimental spectrum of one or the other compounds would certainly also return a high match.
To draw the lines here, remember that to use a SIL-IS, we need to incorporate it into all samples and standards at a single known amount. If the SSR value = 1 between the spectra of the analyte and its SIL-IS form, then the widest range of calibration you might expect to accommodate in your method is just two orders of magnitude (that is, if we prepare the samples so that the SIL-IS amount is in the middle of the calibration range, then we can expect to detect one order higher and lower from this with the analyte concentration).
Another important thing to keep in mind is that the VUV spectra are highly reproducible. This attribute is extremely important for library searching. If a high-quality library spectrum has been recorded into the library, and then another high-quality spectrum is generated experimentally and compared to the library spectrum, SSR values below 0.01 can be routinely observed. Since many of the isomer pairs we have tested exhibited SSR values above this level, the correct library match would be selected most of the time. However, if a noisy experimental spectrum was generated (for example, from a low-abundance compound), some ambiguity might be present in the library search results. But, even in that case, the search results will still more often than not indicate the correct class of compounds.
Figure 1 provides some additional data regarding reproducibility of normalized spectra for a series of fatty acid methyl ester compounds. The figure shows the normalized spectra recorded for three different compounds over a period of approximately five months, as well as the SSR values relative to the recorded library spectrum for each compound. In this case, some of these library spectra were recorded more than two or three years ago. Thus, we can see that gas-phase electronic absorption spectra are highly reproducible-peak maxima do not shift and ratios of absorptivities at different wavelengths remain fixed and constant.
Figure 1: Demonstration of the reproducibility of three fatty acids over a period of five months. SSR values are relative to the recorded library spectrum for each analyte.
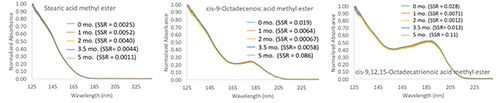
CLICK FIGURE TO ENLARGE
It is only because of this reproducibility, stability, and resolution that we can even consider that it might be possible to distinguish the absorption spectrum for the natural form of an analyte from its deuterated counterpart.
We systematically evaluated the spectral similarity between different labeled and unlabeled analytes (6). For benzene, we evaluated both spectral similarity and absolute absorptivity, but we focus here only on the former (pair-wise SSR values), and what it potentially means for creating a SIL-IS–based quantitation method using GC–VUV.
We found that as you add deuterium atoms sequentially to benzene, a linear increase in SSR value (from 0.4 for benzene/benzene-d1 to 1.7 for benzene/benzene-d6). Interestingly, the spectrum for an all-carbon-13 form of benzene was very similar to the natural form (SSR = 0.06). For other more diverse compound types, the change in SSR with degree of deuteration was less predictable (and only one deuterated form for each of the analyte/SIL-IS pair was obtained and evaluated). A general increasing trend in SSR value was found as the degree of deuteration increased, relative to the size of the molecule. For codeine/codeine-d3, SSR = 0.06, a very high degree of spectral similarity. For benzo[a]pyrene/benzo[a]pyrene-d12, SSR = 2.4, which was the highest degree of dissimilarity we measured in the study.
Again, to place some perspective, searching an experimental spectrum for codeine against the library could very possibly return codeine-d3 as the top hit. On the contrary, searching a good-quality benzo[a]pyrene spectrum against the library would very rarely if ever return the isotopologue as the top hit. Even more importantly, for benzo[a]pyrene and its d12-isotopologue, it would be conceivable to create a SIL-IS–based quantitative GC–VUV method, which would span close to three orders of magnitude. Of course, that is just a guess based on the measured SSR value; it would have to be determined experimentally.
In summary, through the lens of spectral similarity (that is, SSR values), we can evaluate the ability for GC–VUV to distinguish and correctly identify various isomer and isotopologue forms of an analyte. If a pair of isomers or isotopologues are coeluted, then spectral similarity will control over what range of relative abundance it would be possible to determine the individual compounds. For SIL-IS–based GC–VUV quantitative methods, the spectral similarity would control the calibration range, which could be possibly accommodated. Overall, the strategy would seem to be to try to use the most highly deuterated form of the analyte possible as the SIL-IS, if this was necessary. That said, most GC detection methods are not generally subject to a large degree of matrix effects. It may be fine to just simply use another similar compound, which can be chromatographically resolved as an IS. Even so, it was still an interesting question to evaluate.
References
- K.A. Schug, I. Sawicki, D.D. Carlton Jr., H. Fan, H.M. McNair, J.P. Nimmo, P. Kroll, J. Smuts, P. Walsh, and D. Harrison, Anal. Chem.86, 8329–8335 (2014).
- I.C. Santos and K.A. Schug, J. Sep. Sci.40, 138–151 (2017).
- J. Schenk, J.X. Mao, J. Smuts, P. Walsh, P. Kroll, and K.A. Schug, Anal. Chim. Acta945, 1–8 (2016).
- L. Skultety, P. Frycak, C. Qiu, J. Smuts, L. Shear-Laude, K. Lemr, J.X. Mao, P. Kroll, K.A. Schug, A. Szewczak, C. Vaught, I. Lurie, and V. Havlicek, Anal. Chim. Acta971, 55–67 (2017).
- J. Schenk, G. Nagy, N. Pohl, A. Leghissa, J. Smuts, and K.A. Schug, J. Chromatogr. A1513, 210–221 (2017).
- C. Weston, J. Smuts, J.X. Mao, and K.A. Schug, Talanta, in press (2017) https://doi.org/10.1016/j.talanta.2017.09.033.

Kevin A. Schug is a Full Professor and Shimadzu Distinguished Professor of Analytical Chemistry in the Department of Chemistry & Biochemistry at The University of Texas (UT) at Arlington. He joined the faculty at UT Arlington in 2005 after completing a Ph.D. in Chemistry at Virginia Tech under the direction of Prof. Harold M. McNair and a post-doctoral fellowship at the University of Vienna under Prof. Wolfgang Lindner. Research in the Schug group spans fundamental and applied areas of separation science and mass spectrometry. Schug was named the LCGC Emerging Leader in Chromatography in 2009 and the 2012 American Chemical Society Division of Analytical Chemistry Young Investigator in Separation Science. He is a fellow of both the U.T. Arlington and U.T. System-Wide Academies of Distinguished Teachers.
The LCGC Blog: Historical (Analytical) Chemistry Landmarks
November 1st 2024The American Chemical Society’s National Historic Chemical Landmarks program highlights sites and people that are important to the field of chemistry. How are analytical chemistry and separation science recognized within this program?
