The LCGC Blog: Investigating (and Solving) Robustness Issues in HPLC
Not every method is as we would hope it would be. Some methods come to us in imperfect form and we have to live with them, while others are difficult separations and, by necessity, need to be developed using close control of several variables such as eluent pH, eluotropic strength, ionic strength etc.
Not every method is as we would hope it would be. Some methods come to us in imperfect form and we have to live with them, while others are difficult separations and, by necessity, need to be developed using close control of several variables such as eluent pH, eluotropic strength, ionic strength, and so on.
In our analytical laboratory, we deal with methods such as this on a daily basis, and one particular example cropped up recently which highlights how we might go about investigating and solving problems with methods which are less robust.
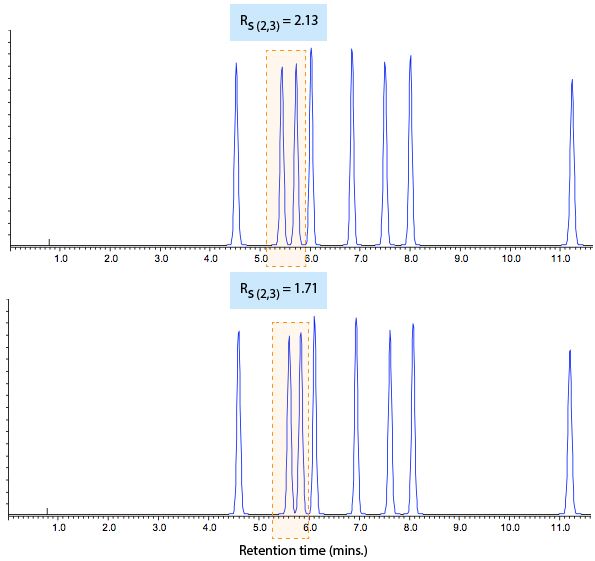
Figure 1 shows the issue.
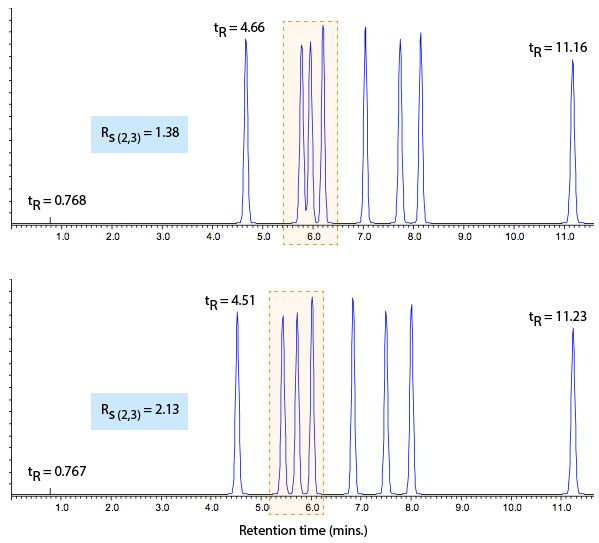
As you can see, the separation doesn’t look as it should in the reference chromatogram (bottom) and we needed to investigate why this is the case. The peaks within the highlighted area in Figure 1 have changed so that resolution is lost (selectivity altered?), but the rest of the separation looks pretty much the same. For subtle changes such as this, we may use a checklist approach to investigate the cause.
First, check the hold-up time (t0) value (you can do this with the baseline disturbance caused by the solvent) to see if there is any variation from the reference chromatogram. Any changes in t0 indicate that the flow rate (or column dimensions) may not be correct or consistent. Changes in these parameters should be easy to fix and, in our example above, the hold-up time is consistent with the reference chromatogram value. Note that when running a method on different systems, changes in hold-up time can also indicate differences in system extra column volume (VE) (see below).
The temperature of the separation can produce subtle changes in the selectivity of the separation and as this is relative easy to check, this is typically second on our list. For ionogenic compounds, the shifts in selectivity or resolution can be more pronounced. Check the column temperature setting in your data system as well as the actual column temperature (use a thermocouple digital thermometer).
If the reference chromatogram was obtained on a different system then it is important to check the gradient profile, particularly with respect to the dwell volume (VD) of the two systems. Differences in the time taken to form and deliver a change in the gradient composition can make a difference to the selectivity of the separation and this typically depends upon the type of pump and the extra column volume.
The details of this aspect are beyond our treatment here, however you can find a full treatment at:
http://www.chromatographyonline.com/gradient-elution-part-iv-dwell-volume-problems.
The take-home message is to ensure that the gradient has been programmed properly (check the data system), and that any differences in system dwell volume are accounted for, especially when transferring between HPLC and UHPLC, older and newer, binary or quaternary systems. In our example, the same system was being used, so this was not the cause of the problem.
Where peaks are very closely eluting, changes in peak shape (fronting and tailing) can radically alter the resolution between peak pairs. To this end, it’s important to carefully control the elutropic strength, pH and ionic strength of the sample diluent, to avoid problems of peak shape distortion on mixing with the eluent at the head of the analytical column. Adjusting the pH of the diluent can be important when the eluent pH is close to the pKa of any of the analytes or if the eluent pH is more than 1 pH unit away from the buffer pKa value. In short, if it’s possible to use the initial eluent composition for the diluent, then do so! Try to avoid lower ionic strength or higher eluotropic strength for the sample diluent and obviously the diluent composition is something to check when investigating method robustness issues. In our case, all was well with the sample diluent.
Once these quick checks have been made, the next step is to investigate the eluent composition, and the quickest way to identify the eluent as the causal factor, is to carefully make up a fresh batch of eluent or eluents if running in gradient mode. The selectivity of a method is usually affected most by the type and concentration of the organic modifier, so one should check that correct organic solvent is being used and is present in the correct ratio, if the method is isocratic. Note that to avoid volume inaccuracy due to the effects of latent heat of mixing, the volume of each solvent should be measured separately before they are combined.
The eluent pH can have a profound effect on the selectivity of the separation when ioniogenic analytes are involved. The method under investigation used a gradient in which the eluent is adjusted to pH 4.1 using a buffer system of 10mM of Ammomium Formate adjusted to pH using formic acid, but on checking the aqueous eluent component, the pH reading was 4.01. Some methods are very susceptible to small changes in pH and from experience, subtle changes in the selectivity of the separation such as we see here, often result from incorrect eluent pH vales. The closer the eluent pH is to the pKa value of the analyte, the more the retention will be affected as pH is altered, and while we strive to design methods in which we avoid having an eluent pH close to any of the analyte pKa values, this cannot always be avoided, especially where there are several ionogenic analytes that may be acidic, basic, zwitterionic or polyfunctional.
Figure 2 shows an illustrative example in which the eluent pH has a profound effect on the retention and selectivity of 2-chlorobenzoic acid.
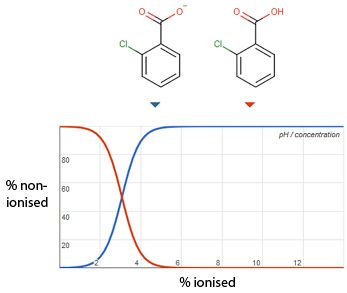
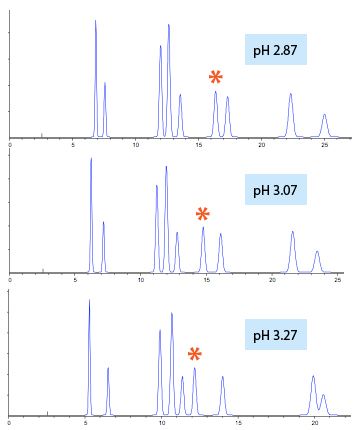
Figure 2: A separation of acidic species including 2-chlorobenzoic acid (*) which has a pKa value of 3.07 (illustrated above) and shifts retention time from 12.07 to 16.35 mins over the eluent pH range 2.87 to 3.27.
We checked the pH meter used to prepare the eluent for the separation in Figure 1 and found it to be accurate and properly calibrated and subsequently re-adjusted the eluent pH to 4.1. After proper equilibration (approx. 10 column volumes + system dwell volume), the separation was repeated and found to be restored to match the original reference chromatogram.
In our method, a large change in selectivity and resolution was induced by a very small change in pH, and this is indicates that pH control using a pH meter to monitor pH while dripping in a dilute solution of formic acid into constantly stirred solution may not be sufficiently accurate. Actually, the issue in this case was found to be that the operator had not measured the final pH of the solution on standing (for example, with no stirring). In such circumstances, it may be better to produce the eluent either volumetrically or gravimetrically, which will produce a more accurately controlled pH.
For more detail on how this can be accomplished, please follow this link:
http://www.chromatographyonline.com/acid-test-more-useful-calculations-hplc-eluent-preparation
When methods are susceptible to changes in eluent composition, the change (typically reduction) in ionic strength during the gradient may bring about changes in the separation, and to this end it is sometimes necessary to achieve a constant ionic strength gradient. This is often achieved by premixing organic and buffered aqueous components in different ratios (80:20 and 20:80 is typical) and adjusting the gradient proportions accordingly.
Figure 3 shows the effects of changes in ionic strength on selectivity of polar neutral compounds.
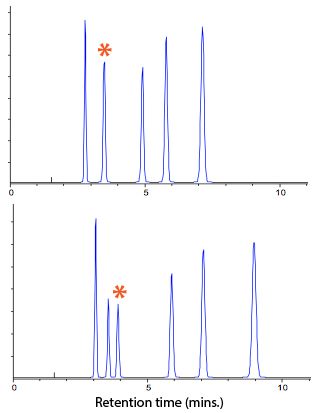
Figure 3: Effects on selectivity of polar neutral species at constant eluotropic strength with 20 mM ammonium formate buffer (top) and 25 mM ammonium formate buffer (bottom).
When methods are less robust than is desirable, then one needs to mitigate against changes in eluent pH during the campaign of analysis. Typically, eluents can become more acidic due to the ingress of CO2 on prolonged standing and pH can also change due to loss of volatile acids or bases.
Figure 4 shows the first and last injection of our current method which highlights the changes in selectivity and resolution due to a reduction in pH caused by ingress of CO2 into the eluent over the course of a long campaign of analyses. One can avoid this phenomenon by ensuring the eluent reservoirs are well capped and in extreme circumstances the headspace in the eluent reservoir may be replaced by a blanket of nitrogen.
In this case, one might consider increasing the buffer concentration from 10mM to avoid changes in pH due to CO2 ingress.
One final word. Make sure that you have a test mix which represents the types of analysis carried out in your department which you can go to as a refuge measurement. That is, when all else is changing, and you really don’t know where the issues are arising, you can inject this test mix to investigate issues with the instrument and HPLC column and rule out (or rule in!) instrument contributions.

Tony Taylor is the technical director of Crawford Scientific and ChromAcademy. He comes from a pharmaceutical background and has many years of research and development experience in small molecule analysis and bioanalysis using LC, GC, and hyphenated MS techniques. Taylor is actively involved in method development within the analytical services laboratory at Crawford Scientific and continues to conduct research in LC-MS and GC-MS methods for structural characterization. As the technical director of the ChromAcademy, Taylor has spent the past 12 years as a trainer and developing online education materials in analytical chemistry techniques.
The LCGC Blog: Historical (Analytical) Chemistry Landmarks
November 1st 2024The American Chemical Society’s National Historic Chemical Landmarks program highlights sites and people that are important to the field of chemistry. How are analytical chemistry and separation science recognized within this program?

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)