An Inside Look at the Analytical Method Greenness Score in High Performance Liquid Chromatography Enantioseparations: An Earth Day Interview with Daniel Armstrong
Chromatographers are constantly thinking about how they can improve the environmental sustainability of separation techniques. This is particularly true when it comes to high-performance liquid chromatography (HPLC) enantioseparations. Recently, Daniel Armstrong, professor of chemistry and biochemistryat the University of Texas at Arlington, and his colleagues proposed mathematical strategies to minimize analytical method greenness scores (AMGS) in HPLC enantioseparations (1). They conducted a case study on the enantiomers of 38 amino acids and introduced a sustainable method using carbonated water-based eluents, which improved chromatographic performance while reducing environmental impact (1). Additionally, the study modified the AMGS formula to incorporate cycle time, providing a more accurate assessment of solvent waste generation (1). The team’s findings were published in Green Chemistry.
In celebration of Earth Day, LCGC International sat down with Armstrong to discuss his most recent paper and his team’s findings.
Daniel Armstrong at the University of Texas at Arlington. Photo Credit: © Daniel Armstrong

Can you explain the concept of analytical method greenness score (AMGS) and its significance in assessing separation methods?
The analytical method greenness score (AMGS) was invented in 2019 as part of the American Chemical Society’s Green Chemistry Institute (ACS-GCI). It is a simplified way to summarize or categorize an analytical method’s “environmental impact” with a single number (that is, the smaller the number, the greener the method). It is formulated in the way of an equation, which includes terms to account for variables such as instrumental power consumption, solvent hazard, and solvent cumulative energy demand (based on production and disposal). I expect that this approach will continue to evolve and become more accurate with future iterations and inputs.
What motivated your research to focus on minimizing AMGS in HPLC enantioseparations?
We wanted to look at the AMGS in general for chromatography and not just for enantioseparations. However, amino acids are an important class of compounds that provided a convenient test case. There is a long publication history of separating these compounds by achiral and chiral methods. This provided us with the opportunity of comparing what was done through previous decades with what is being done today, which tout claims of sustainability (1).
Could you describe the pragmatic and mathematical strategies you proposed to minimize AMGS in your study?
Our point of departure was the 2019 American Chemical Society’s Green Chemistry Institute study that suggested the original AMGS metric (1,2). For over a decade prior to this, we and several others had been doing chromatographic studies (including preparative chromatographic studies, which uses much more solvent and energy) that fall under the current popular moniker of “green chemistry,” but used other descriptive terms such as ultrafast, high throughput, peak processing, streamlined development, and stacked injections. The AMGS metric provided a straightforward way of comparing past, present, and future separations. However, we noted some inconsistencies in the original work and the omissions of factors, such as cycle times, that needed to be considered, which we did. This was not entirely surprising because, as I mentioned previously, this approach will continue to evolve and become more accurate.Also, there may be some issues with the “units”involving the original AMGS metric that may have to be resolved.
How did you generate and assess the case study involving the enantiomers of 38 proteo- and non-proteogenic amino acids?
First, by using a large number of analytes from this compound class, we were assured of having substantial overlap with all prior studies of this type of compound. This allowed us to determine a few things. For example, recent studies that claim “sustainability” actually had higher AMGSs than studies published over 25 years ago. Water and supercritical CO2 are currently considered the “greenest” solvents. However, the current AMGS rubric does not include factors such as having to derivatize a compound (amino acids in this case) or having to use mobile phase additives, such as trifluoroacetic acid, both of which would substantially increase the AMGS. Hence, our study focused on separating native (that is, underivatized) compounds using only water and small amounts of dissolved CO2. Additionally, we focused on ways to use as little solvent as possible, having fast analysis times and including cycle times which had not been considered previously.
Can you elaborate on the sustainable method you introduced for generating carbonated water-based eluents, and how it contributed to lowering the AMGS?
Past researchers have tried numerous methods for carbonating mobile phases (1). We tried several approaches, but we needed something that was simple, inexpensive, and would supply a consistent pressure of CO2 producing an aqueous mobile phase free from all atmospheric contaminants. A thick polypropylene ion chromatography eluent reservoir worked perfectly because it was able to be continually pressurized up to 25 psi and only a little CO2 was needed for these analyses (1).
What were the key findings regarding the use of carbonic acid (H2CO3*) additive in improving chromatographic figures of merit and reducing AMGS?
Compared to pure water, aqueous carbonic acid reduced retention and sharpened peaks. Furthermore, aqueous carbonic acid doesn’t absorb UV-C radiation significantly (down to 190 nm) and is mass spectrometry (MS) compatible. This allows sensitive detection of native amino acids using either low wavelength UV or MS.
How did the utilization of narrow diameter columns with superficially porous particles impact solvent waste generation compared to traditional analyses?
It is known that using “green” solvents, decreasing the amount of solvent used, and the instrumentation time are all key to lowering AMGS in chromatography. Smaller columns that utilize superficially porous packings (SPPs) that reduce retention times contribute to the latter two factors. However, the caveat remains that one must have sufficient retention and selectivity to obtain the desired analytical information. Otherwise, “going green” is useless.
Could you explain the modification made to the AMGS formula to incorporate the "cycle time" of the chromatograph, and its implications?
Note that we did this to address the following two questions: “What speed is optimal for the greenest possible conditions?” and “Is faster always greener for analytical separations?” (1). The time between the method ending and the next injection is known as the instrumental cycle time (tc) (1). First, we assume that an initial run time (ti) is known at one flow rate (Fi). Then, analysis time (ta) can be solved at any flow rate (F) with (1):
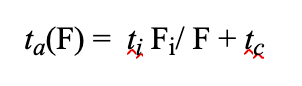
Upon substituting this equation into our previous AMGS equation, we can examine the effect of both cycle time and flow rate.The most interesting implication of neglecting cycle times is that higher flow rates rapidly and asymptotically approach a minimal AMGS, regardless of the solvent used. However, upon incorporating the cycle time, there is a flow rate that produces a minimum AMGS, and it is different for every solvent (1).
How did mathematical optimization principles help in minimizing AMGS with respect to flow rate, and what were the insights gained regarding ideal separation speeds?
By incorporating the abovementioned equation into the AMGS formula, one obtains a new AMGS equation that includes cycle time. Simply taking the derivative of that new equation with respect to flow rate and setting the derivative to zero (which is proper if there is a minimum or maximum) provides a simple relationship that allows one to calculate the flow rate that produces the smallest AMGS.
What are the potential implications of minimizing AMGS in future method development, particularly in fields such as drug development research and production reliant on separation sciences?
Most major pharmaceutical companies have hundreds to >1000 HPLCs and they do a lot of preparative HPLC/supercritical fluid chromatography (SFC) as well. In addition, sample preparation also was considered in the original AMGS work (2). Pharma could not only lower their environmental footprint, but it can also move to the forefront of “sustainable science,” serving as an example for other industries and academia as well. It is nice to have successful, good examples for others to see.
References
(1) Handlovic, T. T.; Wahab, M. F.; Glass, B. C.; Armstrong, D. W. Optimization of Analytical Method Greenness Scores: A Case Study of Amino Acid Enantioseparations with Carbonated Aqueous Systems. Green Chem. 2024, 26, 760–770. DOI: 10.1039/D3GC03005A
(2) Hicks, M. B.; Farrell, W.; Aurigemma, C.; et al. Making the Move Towards Modernized Greener Separations: Introduction of the Analytical Method Greenness Score Calculator. Green Chem. 2019, 21, 1816–1826. DOI: 10.1039/C8GC03875A
New Method Explored for the Detection of CECs in Crops Irrigated with Contaminated Water
April 30th 2025This new study presents a validated QuEChERS–LC-MS/MS method for detecting eight persistent, mobile, and toxic substances in escarole, tomatoes, and tomato leaves irrigated with contaminated water.
University of Tasmania Researchers Explore Haloacetic Acid Determiniation in Water with capLC–MS
April 29th 2025Haloacetic acid detection has become important when analyzing drinking and swimming pool water. University of Tasmania researchers have begun applying capillary liquid chromatography as a means of detecting these substances.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)


